Particle in a Box - 1D (14.3)
• A simple problem that demonstrates the principles of quantum mechanics
is the particle in a box (PIB)
– Inside the box of a given length (a), the potential is zero; outside the box the potential is
infinite
– The wavefunction must be zero outside the box since the PE can’t be infinite
• The solution to the SE for the PIB is an oscillating function
– It is sinusoidal since the wavefunction must be zero at the ends of the box
– An arbitrary integer (n) is in the solution and is referred to as a quantum number
n x
2 nx
sin
a a
n 1, 2, 3, ...
• The energy of the
PIB is also dependent on the quantum number
– Quantum number means the energy is quantized (only has certain values)
– Energy also depends on the length of the box
h 2n 2
En
8ma 2
Properties of PIB Wavefunctions (14.3-14.4)
• Each solution of the PIB SE represents a state of the system
– The lower the state (smaller n) the lower the energy of the system
– First state is referred to as the ground state; higher energy states are called excited
states
• Some interesting features arise from the solution of the PIB SE
– As the quantum number increases, so does the number of values of x for which the PIB
wavefunction equals zero (nodes)
– The PIB probability densities show a number of interesting characteristics (non-uniform
probabilities, regions of zero probability within the box)
• PIB for higher dimensions (2D, 3D) show very similar behavior
– Wavefunction is a product of 1D PIB wavefunctions
– Energy is a sum of 1D PIB energies
– Some energy levels are degenerate (have same energies)
n n x, y n x n x
x y
x
y
2
h 2 n x2 n y
E
8m a 2 b 2
Excited States and Spectroscopy (18.1-18.2)
• In order for a system to go from one state to another, it must absorb or
emit a quantized amount of energy (radiation)
– Light is often used to promote particles to higher states (absorption)
– Light is often emitted when excited states go back to lower lying states (emission)
h E i1 E i
i 1, 2, 3, ...
• Spectroscopy is the use of light to probe states in matter
– Type of light needed depends on the physical problem one is interested in (i.e., the
potential energy function)
– Selection rules dictate whether two states can be connected by shining light on it
(depends on the nature of the wavefunctions of the two states)
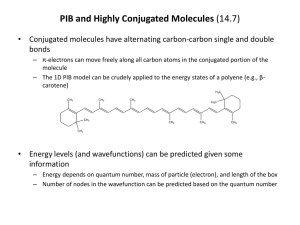
• Does PIB model any real physical phenomena?
– The spectroscopy of highly conjugated molecules (e.g., polyenes) can be explained quite
well by PIB model
Particle in a Box
PIB Wavefunctions
PIB Probability Densities
Absorption and Emission of Radiation
 0
0