Advanced Non-Small Cell
Lung Cancer:
State of the Art
Primo N. Lara, Jr., MD
Professor of Medicine
University of California Davis Cancer Center
Frontline therapy of advanced NSCLC:
“Back in the day” dogma (ie, 2009)
Prognostic factors for survival (PS, Weight Loss,
Gender) are known and clinically apparent
Platinum-based chemotherapy results in
prolongation of life, symptom control, & superior
QOL compared with supportive care alone
– Four to six cycles are sufficient
Inhibitors of epidermal growth factor and
angiogenesis pathways improve outcomes in
select patient subsets
ECOG 1594: Platinum-based doublets
have similar survival outcomes
Cis/Gem
Control arm: Cisplatin/paclitaxel
Schiller JH, et al. N Engl J Med. 2002; 346:92–98
Cis/Docetaxel
Carbo/Paclitaxel
Duration of chemotherapy:
4-6 cycles are sufficient
Author
Smith
JCO 2001
Socinski
JCO 2002
Eberhardt W, et al. (2007) ASCO
Study design
Patients
Median survival
(months)
MVP x 3
155
6
P value
.2
MVP x 6
153
7
Carbo/Pac x 4
114
6.6
.63
Carbo/Pac till PD
116
8.5
Bevacizumab in NSCLC (E4599)
Proportion surviving
1.0
12 mo 24 mo Median
PC
44.4% 15.4% 10.3 mo
BV/PC 51.0% 22.0% 12.3 mo
0.8
Median
12.3 mo
0.6
HR: 0.80, P = .013
Median
10.3 mo
0.4
0.2
0.0
0
6
12
18
24
30
36
42
48
PC
444
318
190
104
36
9
5
1
0
BV/PC
434
340
216
127
54
25
8
3
0
Patients at risk
Months
Non-squamous histology, no hemoptysis, no CNS mets, no anticoagulation
Sandler A, et al. N Engl J Med. 2006; 356:2542–2550
Cisplatin/vinorelbine + cetuximab (FLEX) in
EGFR-positive* (by IHC) NSCLC
Patients surviving, %
▬ Cetuximab +CT
▬ CT
HR = 0.871, 0.762–0.996
Log-rank P =.044
11.3
10.1
1-year OS
47% vs 42%
Months
Pirker R, et al. (2008) ASCO
* One or more IHC positive cell(s)
Metastatic NSCLC:
The New Issues (i.e., October 2010)
Maintenance therapy
– After completing platinum-based chemo, should
all patients receive maintenance therapy?
Tumor histology
– Should we routinely use histology to select
therapy?
Frontline biologics
– Can we abandon frontline platinum therapy in
favor of single agent EGFR TKI?
Maintenance therapy:
Considerations
Continuing same chemotherapy past 4–6
cycles yields no clear benefit
E4599 and FLEX trials included maintenance
therapy of bevacizumab and cetuximab,
respectively
Maintenance therapy with non-cross
resistant agents may be more efficacious
Maintenance pemetrexed
Double-blind, Placebo-controlled, Multicenter, Phase III Trial
Stage IIIB/IV NSCLC
Pemetrexed 500 mg/m2
(d1,q21d) + BSC (N = 441)*
PS 0-1
4 prior cycles of gem,
doc, or tax + cis or carb,
with CR, PR, or SD
Randomization factors:
gender
PS
stage
best tumor response to
induction
non-platinum induction
drug
brain mets
2:1
Randomization
Primary Endpoint = PFS
Placebo (d1, q21d) + BSC
(N = 222)*
*B12, folate, and dexamethasone given in both arms
Ciuleanu TE, et al. (2008) ASCO; Belani CP, et al. (2009) ASCO
Drug-related adverse events
Pemetrexed
n = 441
Placebo
n = 222
P value
Drug-related deaths*
0.0%
0.0%
1.000
≥ 1 serious AE (SAE)
4.3%
0.0%
.001
14.3%
3.6%
< .001
≥ 1 grade 3/4 AE
Drug-related CTCAE grade 3/4 toxicity (≥ 2% of pts)
Anemia
2.7%
0.5%
.070
Neutropenia
2.7%
0.0%
.011
Fatigue
4.3%
0.5%
.004
* on-study or within 30 days post-study
Ciuleanu TE, et al. (2008) ASCO
Progression-free
Survival
Progression-free
Probability
JMEN: “Maintenance” Pemetrexed vs
Placebo After Platinum-based Chemotherapy
1.0
0.9
0.8
0.7
0.6
0.5
0.4
0.3
0.2
0.1
0.0
HR = 0.60 (95% CI: 0.49-0.73)
P <0.00001
Pemetrexed 4.0 mos
Placebo 2.0 mos
Overall
Survival
Belani CP, et al. ASCO 2009. CRA8000.
Survival
Probability
0
1.0
0.9
0.8
0.7
0.6
0.5
0.4
0.3
0.2
0.1
0.0
3
6
9
12
15
Time (months)
18
21
24
HR = 0.79 (95% CI: 0.65-0.95)
P = 0.012
Pemetrexed 13.4 mos
Placebo 10.6 mos
0
3
6
9 12 15 18 21 24 27 30 33 36 39 42 45 48
Time (months)
JMEN: “Maintenance” Pemetrexed vs
Placebo After Platinum-based Chemotherapy
Pemetrexed
N = 309
Placebo
N = 178
Any systemic therapy
51%
67%
Docetaxel
22%
29%
Erlotinib
22%
21%
Gefitinib
13%
10%
1%
19%
Pemetrexed
•
•
•
•
Post-study therapy was not balanced between the arms
51% of patients on the pemetrexed arm received further therapy
(equates to third-line therapy)
Only 19% of placebo-arm patients received pemetrexed
Would survival benefits have been preserved if more patients on
the placebo arm received pemetrexed?
Belani CP, et al. ASCO 2009. CRA8000.
JMEN: “Maintenance” Pemetrexed vs
Placebo: Survival by Histology
Non-squamous (n = 481)
Squamous (n = 182)
Survival Probability
HR = 0.70 (95% CI: 0.56-0.88);
P = 0.002
HR = 1.07 (95% CI: 0.49–0.73);
P = 0.678
1.0
1.0
0.9
0.9
0.8
0.8
0.7
0.7
0.6
0.6
Pemetrexed 15.5 mos
0.5
0.4
Pemetrexed 9.9 mos
0.5
0.4
0.3
0.3
Placebo
10.3 mos
0.2
0.1
0.1
0.0
0.0
0
3
6
9 12 15 18 21 24 27 30 33 36 39 42 45 48
Time (months)
Belani CP, et al. ASCO 2009. CRA8000.
Placebo
10.8 mos
0.2
0
3
6
9 12 15 18 21 24 27 30 33 36 39 42 45 48
Time (months)
Maintenance chemotherapy:
Points to Ponder
• Definition of “maintenance” varies
• Benefit for pemetrexed confined to nonsquamous histology
• Many patients never received secondline therapy!
– Survival in control arms may have diminished due
to lack of life-prolonging second line therapy
• Difficult to reconcile with maintenance
bevacizumab, cetuximab, and erlotinib
Maintenance chemotherapy:
Is it time for routine clinical use?
• Issues of cost, convenience, toxicity, and
QOL need to be weighed against (modest)
PFS benefit in the palliative care setting
– Many patients desire a “drug holiday”
• Maintenance chemotherapy can be
considered only for selected, highly
motivated patients
– Bottom line: It’s not for everyone
Maintenance biologics
SATURN trial (N = 889)
– Phase III erlotinib vs. placebo following initial
treatment with platinum-based chemotherapy
ATLAS trial (N = 1157)
– Phase III bevacizumab ± erlotinib following initial
treatment with chemo + bevacizumab
– Brain mets, non-centrally located squamous,
anticoagulation allowed
1. Cappuzzo F, et al. (2009) ASCO; 2. Miller VA, et al. (2009) ASCO
SATURN study design
Cappuzzo et al, ASCO 2009, # 8001
Erlotinib
150mg/day
Chemonaïve
advanced
NSCLC
n=1,949
4 cycles of firstline platinum
doublet
chemotherapy*
Non-PD
n=889
PD
1:1
Placebo
PD
Mandatory tumour
sampling
Co-primary endpoints:
Stratification factors:
• PFS in all patients
• PFS in patients with EGFR IHC+
tumours
• EGFR IHC (positive vs negative vs
indeterminate)
• Stage (IIIB vs IV)
Secondary endpoints:
• ECOG PS (0 vs 1)
• OS in all patients and those with
• CT regimen (cis/gem vs carbo/doc vs
EGFR IHC+ tumours, OS and PFS in
others)
EGFR IHC– tumours; biomarker
analyses; safety; time to symptom
• Smoking history (current vs former vs
progression; QoL
never)
*Cisplatin/paclitaxel; cisplatin/gemcitabine; cisplatin/docetaxel cisplatin/vinorelbine;
• Region
carboplatin/gemcitabine; carboplatin/docetaxel carboplatin/paclitaxel
SATURN Trial: Efficacy Summary
Erlotinib
(n = 438)
Placebo
(n = 451)
(n = 437)
(n = 447)
PFS at 12 weeks
53%
40%
PFS at 24 weeks
31%
17%
12.3 weeks
11.1 weeks
12 months
11 months
(n = 436)
(n = 445)
ORR
12%
DCR
61%
PFS
Median PFS
Median OS
HR
P Value
0.71
< .0001
0.81
.0088
5%
NA
.0006
51%
NA
.0035
• PFS benefit with erlotinib observed regardless of gender, race, histology or smoking history
Cappuzzo et al. J Thorac Oncol 2009; 4(suppl 1):S289 (abstract A2.1).
PFS in EGFR wild-type tumors
PFS probability
1.0
Erlotinib (n=199)
Placebo (n=189)
0.8
0.6
HR=0.78 (0.63–0.96)
Log-rank p=0.0185
0.4
0.2
0
0
8
Capuzzo, ASCO 2009
16
24
32
40 48 56 64
Time (weeks)
72
80
88
96
PFS in EGFR mutation+ tumors*
PFS probability
1.0
Erlotinib (n=22)
Placebo (n=27)
0.8
HR=0.10 (0.04–0.25)
Log-rank p<0.0001
0.6
0.4
0.2
0
0
8
Capuzzo, ASCO 2009
16
24
32
40 48 56 64
Time (weeks)
72
80
88
96
*60% censored
SATURN: Post-study Treatment
Erlotinib*
(n = 438)
Placebo*
(n = 451)
All classes
55%
64%
Taxanes (including docetaxel)
26%
27%
Antimetabolites (including pemetrexed)
18%
20%
Antineoplastic agents
11%
15%
Tyrosine-kinase inhibitors
5%
16%
Platinum compounds
8%
11%
*% receiving ≥1 treatment
Cappuzzo F, et al. ASCO 2009. Abstract 8001.
ATLAS Phase III Study Design
Miller et al, ASCO 2009, #8002
Bevacizumab (15mg/kg) +
erlotinib (150mg) to PD
Chemo-naïve
advanced
NSCLC
N=1,160
4 cycles of
1st-line
chemotherapy*
+ bevacizumab
Non-PD
n=768 (66%)
1:1
Unblind
at PD
Bevacizumab +
placebo
to PD
Eligibility
Primary endpoint
• Stage IIIB**/IV NSCLC
• PFS in all randomized pts
• ECOG performance status 0-1
Secondary endpoints
Stratification factors
• Overall survival
• Gender
• Safety
• Smoking history (never vs
former/current)
Exploratory endpoints
• ECOG performance status (0 v >1)
Post progression
therapy
• Biomarker analyses (IHC, FISH, EGFR &
K-Ras mutation)
• Chemotherapy regimen
*Carbo/paclitaxel; cis/vinorelbine; carbo or cis/gemcitabine; carbo or cis/docetaxel.
**IIIB with pleural effusion
Progression-Free Survival
Proportion Without Event
1.0
Bev + Placebo
(n=373)
Bev + Erlotinib
(n=370)
0.8
0.6
HR=0.722 (0.592-0.881)
Log-rank P=0.0012
0.4
0.2
0.0
0
3
6
9
12
15
18
21
Progression-Free Survival (months)
Miller et al, ASCO 2009, #8002
ATLAS: Toxicity
Bev + Placebo, n (%)
(n=368)
Bev + Erlotinib, n (%)
(n=367)
Any Grade AE
313 (85.1%)
349 (95.1%)
Grade 3–4 AE
112 (30.4%)
162 (44.1%)
4 (1.1%)
8 (2.2%)
60 (16.3%)
84 (22.9%)
Grade 5 AE
SAE
The most common adverse events were rash and diarrhea
Miller et al, ASCO 2009, #8002
Formal statistical comparison testing between treatment arms was not done.
Take Home Points:
SATURN and ATLAS trials
• Trials confirm that erlotinib is an active agent in
NSCLC (duh!)
• Patients on maintenance erlotinib had more
toxicities than those on placebo
• Only a minority (16% in SATURN, 40% in ATLAS) of
placebo patients ever received subsequent EGFR
TKI therapy!!!
• As expected, PFS benefit was best in patients with
EGFR-mutated tumors
• Need to balance consequences of increased toxicity
and cost in the context of modest PFS benefit
Metastatic NSCLC:
The New Issues (i.e., October 2010)
Maintenance therapy
– After completing platinum-based chemo, should
all patients receive maintenance therapy?
Tumor histology
– Should we routinely use histology to select
therapy?
Frontline biologics
– Can we abandon frontline platinum therapy in
favor of single agent EGFR TKI?
Complexities of lung cancer pathogenesis
result in diverse histologic subtypes
Sun S, et al. Nat Rev Cancer. 2007; 7:778–790
SCLC
(~15%)
Squamous Cell Ca
(~25%)
Adenocarcinoma
(~45%)
BAC
(~5-10%)
Large Cell
(~5-10%)
NOS
(Not Otherwise
Specified
(~10-30%)
NSCLC histology:
Treatment Considerations
Bevacizumab is FDA-approved for non-squamous
histology due to SAFETY issues
– Severe hemoptysis
Pemetrexed is FDA-approved for non-squamous
histology due to EFFICACY issues
– PFS and OS benefit confined to non-squamous
EGFR TKIs traditionally viewed as more efficacious
in adenocarcinoma
– Likely due to higher rate of EGFR mutants in adeno
JMDB trial: Cisplatin/pemextexed (CP) vs
cisplatin/gemcitabine (CG) in Adv NSCLC
Randomization
Factors
•
•
•
•
•
Stage
PS
Gender
Histo vs cyto dx
Brain mets hx
Cisplatin 75 mg/m2 day 1 plus
Pemetrexed 500 mg/m2 day 1
R
Cisplatin 75 mg/m2 day 1 plus
Gemcitabine 1250 mg/m2 days 1 & 8
Vitamin B12, folate, and dexamethasone given in both arms
Scagliotti GV, et al. J Clin Oncol. 2008; 26:3543–3551
Cisplatin/pemetrexed (CP) vs
cisplatin/gemcitabine (CG) in NSCLC
No difference in
PFS or OS
CP improves survival over CG
in non-SCCA (HR 0.81, P = .005)
CG improves survival over CP
in SCCA (HR 1.23, P = .05)
Scagliotti GV, et al. J Clin Oncol. 2008; 26:3543–3551
Pemetrexed: Influence of histology on
efficacy
In two other phase III trials
(docetaxel vs. pemetrexed;
maintenance pemetrexed vs.
placebo), benefit was confined to
patients with non-squamous
histology
Scagliotti GV, et al. J Clin Oncol. 2008; 26:3543–3551
Take Home Points:
Histology-based therapy
NSCLC histology is now a consideration in
therapeutic selection for chemotherapy
This is but one (primitive) step in the
direction of “personalized therapy”
Still unclear how “NSCLC NOS” or cytologic
diagnoses (by FNA) should be treated
True personalized therapy will rely on
molecular profiling rather than histology
Metastatic NSCLC:
The New Issues (i.e., October 2010)
Maintenance therapy
– After completing platinum-based chemo, should
all patients receive maintenance therapy?
Tumor histology
– Should we routinely use histology to select
therapy?
Frontline biologics
– Can we abandon frontline platinum therapy in
favor of single agent EGFR TKI?
IPASS Study design
Conducted in China, Japan, Thailand, Taiwan, Indonesia, Malaysia, Philippines,
Hong Kong and Singapore
Randomization period: March 2006 – October 2007
Patients
•
•
•
•
•
•
•
Chemo-naïve
Age ≥18 years
Adenocarcinoma
histology
Never or ex-light
smokers*
Life expectancy
≥12 weeks
WHO PS 0-2
Measurable stage
IIIB / IV disease
Gefitinib
(250 mg / day)
End points
Primary
•
Progression-free survival
(non-inferiority)
Secondary
1:1 randomization
Carboplatin
(AUC 5 or 6) /
paclitaxel
(200 mg / m2)
3 weekly#
•
•
•
•
•
Objective response rate
Overall survival
Quality of life
Disease-related symptoms
Safety and tolerability
Exploratory
•
Biomarkers
─ EGFR mutation
─ EGFR-gene-copy number
─ EGFR protein expression
*Never smokers, <100 cigarettes in lifetime; ex-light smokers, stopped 15 years ago and smoked 10 pack years;
#limited to a maximum of 6 cycles. Carboplatin/paclitaxel was offered to gefitinib patients upon progression
WHO, World Health Organization; PS, performance status; AUC, area under the curve; EGFR, epidermal growth
factor receptor
Mok et al 2008
Progression-free survival
Probability 1.0
of PFS
N
Events
0.8
Gefitinib
Carboplatin /
609
453 (74.4%)
paclitaxel
608
497 (81.7%)
HR (95% CI) = 0.74 (0.65, 0.85) p<0.0001
0.6
5.8
74%
48%
7%
Median PFS (months)
5.7
4 months progression-free
61%
6 months progression-free
48%
12 months progression-free 25%
0.4
Gefitinib demonstrated superiority relative to
carboplatin/paclitaxel in terms of PFS
0.2
0.0
Patients at risk : 0
Gefitinib 609
C/P
608
4
8
12
16
20
24 Months
363
412
212
118
76
22
24
3
5
1
0
0
Primary Cox analysis with covariates; HR <1 implies a lower risk of progression on gefitinib; ITT population
PFS, progression-free survival; ITT, intent-to-treat; HR, hazard ratio; CI, confidence interval; C/P, carboplatin/paclitaxel
Mok et al 2008
IPASS: EGFR mutation positive status and clinical
characteristics
% of
samples
EGFR
mutation
positive
Overall EGFR mutation positive rate = 59.7% (261 / 437)
68.5
63.0
60.0
57.1
49.0
Male
60.7
57.8
60.2
56.7
46.9
Female
PS 0/1
PS 0/2
Never Light ex- Locally Metastatic Age
Age
smoked smoker advanced
<65 yrs >65 yrs
Progression-free survival in EGFR mutation positive and
negative patients
EGFR mutation positive
EGFR mutation negative
Probability of progression-free
survival
1.0
0.8
0.6
0.4
Gefitinib (n=91)
Carboplatin/paclitaxel (n=85)
HR (95% CI) = 2.85 (2.05, 3.98)
p<0.0001
No. events gefitinib , 88 (96.7%)
No. events C/P, 70 (82.4%)
Median PFS G, 1.5 months
Median PFS C/P, 5.5 months
1.0
Probability of progression-free
survival
Gefitinib (n=132)
Carboplatin/paclitaxel (n=129)
HR (95% CI) = 0.48 (0.36, 0.64)
p<0.0001
No. events gefitinib, 97 (73.5%)
No. events C/P, 111 (86.0%)
Median PFS G, 9.5 months
Median PFS C/P, 6.3 months
0.8
0.6
0.4
0.2
0.2
0.0
0.0
0
Patients at risk :
Gefitinib 132
C/P
129
4
8
12
16
20
24
0
4
8
Months
108
103
71
37
31
7
12
16
20
24
1
0
0
0
0
0
Months
11
2
3
1
0
0
91
85
21
58
4
14
2
1
Treatment by EGFR mutation status interaction test, p<0.0001
Cox analysis with covariates; HR <1 implies a lower risk of progression on gefitinib; ITT population
Mok et al 2008
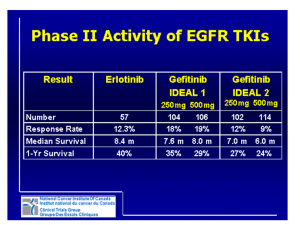
First line gefinitib vs. chemotherapy
in EGFR mutated NSCLC
NSCLC with
sensitive EGFR
mutations
Stage IIIb/ IV
No prior chemo.
PS 0-1
age of 20-75 y.o
R
balanced :
Institution
sex
stage
Gefitinib
n=160
Primary endpoint
PFS
2ndary endpoints
OS
Response
CBDCA+TXL
n=160
Side-effects
QOL
• The sample size was calculated to be 320 in total (alpha=5%,
power=80%) to confirm the superiority of Arm A (hazard ratio = 0.69).
• An interim analysis to investigate PFS was planned 4 months after
200 pts were entered .
North East Japan (NEJ) Gefitinib Study Group
PFS
OS
Memondo, NEJM 2010
Memondo, NEJM 2010
Potential Oncogenic “Drivers” in
Non-small Cell Lung Cancer (NSCLC)
Adenocarcinoma
K-ras
EGFR
B-raf
Her2
Other
PIK3CA
ALK
MET
Other
ALK (~5%)
ALK = anaplastic lymphoma kinase; EGFR = epidermal growth
factor receptor; Her2 = human epidermal growth factor receptor 2;
PIK3CA = phosphoinositide-3-kinase, catalytic, alpha polypeptide
Massachusetts General Hospital, data on file.
[AT Shaw, personal communication]
Tumor Responses to Crizotinib for
Patients
with
ALK-positive
NSCLC
(N=82)
60
Maximum change in tumor size (%)
Progressive disease
Stable disease
40
Confirmed partial response
Confirmed complete response
20
0
–20
–30%
–40
–60
–80
–100
*
*Partial response patients with 100% change have non-target disease present
77% of Patients with ALK-positive NSCLC
Remain on Crizotinib Treatment
Individual patients
• Duration of treatment
(median: 5.7 months)
0–3 mo
13 pts
>3–6 mo
29 pts
>6–9 mo
24 pts
>9–12 mo
9 pts
>12–18 mo
4 pts
>18 mo
3 pts
0
3
6
9
12
15
Treatment duration (months)
N=82; red bars represent discontinued patients
18
• Reasons for discontinuation
– Related AEs
1
– Non-related AEs
1
– Unrelated death
2
– Other
2
– Progression
13
21
•
Clinical Activity of Crizotinib in
Patients
with
NSCLC
Objective
response
rateALK-positive
(ORR): 57% (95%
CI: 46, 68%)
– 63% including 5 as yet unconfirmed PRs
– 57% (8/14) for patients with performance status 2
or 3
0
ORR
% (n/N)
80 (4/5)
1
52 (14/27)
2
67 (10/15)
≥3
56 (19/34)
No. prior regimens*
* Unknown for 1 patient
● Response duration: 1 to 15 months
● DCR† (CR/PR/SD at 8 weeks): 87% (95% CI: 77, 93%)
†Disease
control rate
Progression-free survival probability
Median PFS has Not been Reached
70% of Patients in Follow-up for PFS
1.00
PFS probability at 6 months:
72% (95% CI: 61, 83%)
0.75
0.50
0.25
0.00
Median follow-up for PFS: 6.4 months
(25–75% percentile: 3.5–10 months)
0
2.5
95% Hall–Wellner confidence bands
5.0
7.5
10.0
12.5
15.0
Progression-free survival (months)
17.5
Treatment-related Adverse Events in
ALK-positive NSCLC (≥10%)
Adverse event
Grade 1
n (%)
Grade 2
n (%)
Grade 3
n (%)
Grade 4
n (%)
Total
n (%)
Nausea
43 (52)
1 (1)
0
0
44 (54)
Diarrhea
38 (46)
1 (1)
0
0
39 (48)
Vomiting
35 (43)
1 (1)
0
0
36 (44)
Visual disturbance*
34 (42)
0
0
0
34 (42)
Constipation
18 (22)
2 (2)
0
0
20 (24)
Peripheral edema
13 (16)
0
0
0
13 (16)
Dizziness
12 (15)
0
0
0
12 (15)
Decreased appetite
11 (13)
0
0
0
11 (13)
Fatigue
8 (10)
0
0
0
8 (10)
*Changes in light/dark accommodation (no abnormalities on ophthalmologic exam)
N=82
Conclusions:
Frontline NSCLC therapy
Optimal therapy is rapidly evolving
Maintenance therapy is an option for highly
motivated patients
Clinical, histologic, and molecular biomarkers are
now important considerations for therapy
selection
Future advances in NSCLC outcomes will likely be
due to molecular selection
Support for clinical trials testing this paradigm is
essential