Access PowerPoint Presentation
advertisement

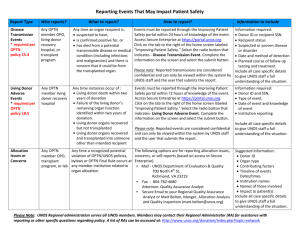
National Webinar to Review Non-Discussion Agenda Spring 2014 Public Comment If you are logged into the webinar, please enter the audio PIN Please put your phone on MUTE and do not place this call on HOLD Objectives of Call Non-Discussion Agenda Process Overview Present and Discuss Non-Discussion Agenda Proposals Regional Meetings Feedback to the Sponsoring Committees Background Regional Meeting Goals Discuss and comment on proposed policies and bylaws Collect feedback and provide to sponsoring committees Receive updates on OPTN/UNOS committee activities and projects Discuss regional business Spring 2014 Regional Meetings 17 proposals submitted for public comment 11 OPTN/UNOS committees with updates Committee projects that require regional discussion and feedback 1.5 additional hours to the standard 5 hour regional meeting Regional Meeting Agenda Divide the agenda into two sections Discussion Non-Discussion Discussion agenda format is the same as previous meetings Presentation at the regional meeting Discussion at the regional meeting Regional Vote Non-Discussion agenda format Items discussed today No discussion at the regional meeting Regional Vote Non-Discussion Agenda Proposals Proposal to Require the Reporting of Aborted Living Donor Organ Recovery Procedures (Living Donor Committee) Proposal to Allow Non-substantive Changes to the OPTN Policies and Bylaws (Membership and Professional Standards Committee) Proposed ABO Subtyping Consistency Policy Modifications (Operations and Safety Committee) Proposal to Require the Collection of Serum Lipase for Pancreas Donors (Pancreas Transplantation Committee) Proposal to Clarify Data Submission and Documentation Requirements (Policy Oversight Committee) Public Comment Proposals http://optn.transplant.hrsa.gov Proposal to Allow Non-Substantive Changes to the OPTN Policies and Bylaws Policy Oversight Committee Stuart Sweet, MD St Louis Children’s Hospital Ann-Marie Leary Ann-Marie Leary The Problem Sometimes we find obvious clerical (or nonsubstantive) errors in the OPTN Policies and Bylaws Currently, nothing in the Bylaws or Policies allows staff to make these changes The Executive Committee or Board of Directors must approve these simple changes Goal of the Proposal Allow staff to make non-substantive changes immediately, without Board approval Executive Committee would review these changes later How the Proposal will Achieve its Goal This proposal adds language to the OPTN Bylaws that will permit staff to make clerical changes: OPTN Bylaws Article X: Amendment of Charter and Bylaws OPTN Bylaws Article XI: Adoption of Policies What’s a “non-substantive” change? Capitalization or punctuation, as needed to maintain consistency with current policy Typographical, spelling, or grammatical errors Lettering and numbering of a rule or the subparts of a rule, according to style conventions in current policy Cross-references to rules or sections that are cited incorrectly because of subsequent repeal, amendment, or reorganization of the sections cited Supporting Evidence Many legislative and regulatory bodies have procedures that provide authority for making minor changes to their policies and legislation: § 30-149. Authority for minor changes to the Code of Virginia North Carolina General Statutes § 150B-21.20 (Codifier's Authority to Revise Form of Rules) Washington Revised Code § 1.08.015 (Codification and Revision of Laws – Scope of Revision) What Members will Need to Do This proposal will not require that members do anything or change their procedures If members print out copies of the Bylaws or Policies, they should periodically print out new, corrected versions Contacts for Feedback Chair Yolanda Becker, MD ybecker@surgery.bsd.uchicago.edu Vice Chair Susan Dunn, RN, BSN, MBA sdunn@donoralliance.org UNOS Staff Liaison Leigh Kades leigh.kades@unos.org Questions – Click hand button Proposal to Modify ABO Subtyping References for Consistency Operations and Safety Committee Theresa Daly, MS, RN, FNP New York-Presbyterian/Columbia The Problem Current OPTN policy contains different terms that have the same intended meaning Policy 2.6.B: “found to be non-A1 or non-A1B” Policy 13.7.B: “to a blood type A2 or A2B” Policy 14.4.A.i: “donor to be non-A1 (negative for A1) or non-A1B (negative for A1B)” Inconsistency may create confusion Goal of the Proposal Use consistent language for all subtype references How the Proposal will Achieve its Goal Pertinent references will read: Blood type A, non-A1 Blood type AB, non-A1B Supporting Evidence June 2011, OPTN published guidance based on work of ABO subtyping committee “It is important to know that the technically accurate term for A2 and A2B donors is ‘A1-negative’ or ‘A, non-A1’ because A2 is not directly tested for and many other rare subtypes exist (e.g. A3, Aint, etc.)”. Some OPOs reluctant to report “A2” subtypes due to this issue What Members will Need to Do Understand the meaning of the terms: Blood type A,non-A1 Blood type AB,non-A1B, Regional Representatives Region Name Email 1 Sukru Emre, MD, FACS sukru.emre@yale.edu 2 Deborah Maurer, RN, MBA maurerd@upmc.edu 3 Eric Gibney, MD eric.gibney@piedmont.org 4 dsamp@sleh.com 5 Diesa Samp, BSN, RN, CCTC Kristin Mekeel, MD, FACS kmekeel@ucsd.edu; kmekeel@rchsd.org 6 Mark Menotti, RN, MBA menotti@ohsu.edu 7 Colleen McCarthy, RN, BSN colleen.mccarthy@bcw.edu 8 Nancy Long, RN, BA, CCTC nlong@saint-lukes.org 9 Colleen O'Donnell-Flores, MHA Colleen.O'Donnell@nyumc.org 10 Ladora Dils, BSN, MHA, CPTC ddils@lifelineofohio.org 11 Laura Butler, NP-BC, MMHC laura.butler@vanderbilt.edu Questions – click hand button Proposal to Clarify Data Submission Reporting and Documentation Obligations Membership & Professional Standards Committee Carl Berg, MD Duke University Hospital Ann-Marie Leary Ann-Marie Leary The Problem Policy does not specify what has always been implied Data submitted through OPTN forms must be accurate Members must provide documentation to verify data accuracy Other policies explicitly state members must maintain or provide certain documentation Goal of the Proposal Clarify policy to alleviate confusion Reduce resources required to obtain documentation and review potential noncompliance issues Only applies to standardized forms required by Policy 18 What Members will Need to Do Should not have to change policies or procedures Will not be required to provide additional documentation during routine site surveys UNOS will not change how it monitors compliance May be asked to gather and provide relevant documentation upon request for MPSC review Regional Representatives Region Name Email 1 Heung Bae Kim, MD heung.kim@childrens.harvard.edu 2 David Reich, MD David.Reich@DrexelMed.edu 3 Charles Wright, MD Charles.wright@lifelinkfound.org 4 W. Kenneth Washburn, MD washburn@uthscsa.edu 5 Richard Perez, MD richard.perez@ucdmc.ucdavis.edu 6 Viken Douzdjian, MD vdouzdji@lhs.org 7 Julie Heimbach, MD heimbach.julie@mayo.edu 8 Christie Thomas, MB, FRCP, FASN, FAHA christie-thomas@uiowa.edu 9 Lloyd Ratner, MD lr2182@cumc.columbia.edu 10 Tim Taber, MD tetaber@iupui.edu 11 Robert Stratta, MD rstratta@wakehealth.edu Questions – click hand button Proposal to Require the Collection of Serum Lipase for Pancreas Donors Pancreas Transplantation Committee Jonathan Fridell, MD Indiana University Health Ann-Marie Leary Ann-Marie Leary The Problem Serum lipase is a direct indicator of pancreas quality Serum lipase is not reported for all potential pancreas donors Goal and Achieving the Goal Goal: Provide transplant professionals with critical information about the quality of the pancreas offered Change: Require collection of serum lipase for all pancreas donors Report lab’s upper limit of normal value for serum lipase test What Members will Need to Do OPOs will be required to report serum lipase values for all pancreas donors A new field gives OPOs ability to report the upper limit of normal value Regional Representatives Region Name Email 1 Heidi Yeh, MD hyeh@partners.org 2 Silke Niederhaus, MD sniederhaus@smail.umaryland.edu 3 Carlos R del Coro, MD delcoro14@gmail.com 4 Jacqueline Lappin, MD jlappin@sw.org 5 Fuad Shihab, MD fuad.shihab@hsc.utah.edu 6 Ramasamy Bakthavatsalam, MD baktha@uw.edu 7 Jon Odorico, MD jon@surgery.wisc.edu 8 Zoe Stewart, MD, PhD zoe-stewart@uiowa.edu 9 Pedro Sandoval, MD prs2109@columbia.edu 10 Muhammad Mujtaba, MD, FASN mmujtaba@iupui.edu 11 Douglas Hale, MD douglas.a.hale@vanderbilt.edu Questions – click hand button Proposal To Require the Reporting of Aborted Living Donor Recovery Procedures Living Donor Committee Christie Thomas, MD University of Iowa The Problem Aborted living donor organ recovery procedures may not be reported at time of event and therefore may be under reported Goals of the Proposal Improve safety of living donation Help quantify risk of living donation How the Proposal will Achieve its Goal Aborted procedures - new living donor adverse event category added to UNetSM Improving Patient Safety Portal Supporting Evidence Reliable count of aborted living donor organ recovery procedures does not exist Since 2003 - only 12 cases reported where a donation surgery was aborted after anesthesia was administered because of a threat to donor’s health What Members will Need to Do Recovery hospitals will report aborted living donor recovery procedures within 72 hours of the event as Living Donor Adverse Events through the UNetSM Improving Patient Safety Portal Regional Representatives Region Name Email 1 Sanjay Kulkarni, MD sanjay.kulkarni@yale.edu 2 Francis Weng, MD fweng@barnabashealth.org 3 George Therapondos, MD gtherapondos@ochsner.org 4 Matthias Kapturczak, MD, PhD mkapturczak@sakdc.com 5 Randolph Schaffer III, MD schaffer.randolph@scrippshealth.org 6 Christian Kuhr, MD christian.kuhr@vmmc.org 7 J. Michael Millis, MD Krista Lentine, MD, PhD Mmillis@surgery.bsd.uchicago.edu lentinek@slu.edu; krista.lentine@tenethealth.com 9 Carlos Marroquin, MD Carlos.Marroquin@vtmednet.org 10 Emilio Poggio, MD poggioe@ccf.org 11 Vinaya Rao, MD vrao3@uthsc.edu 8 Questions – click hand button Non-Discussion Agenda Process for moving a proposal to the Discussion Agenda 15% of member institutions within a region submit a request All requests must be received one week prior to the meeting date If the15% threshold is met: Proposal will be presented and discussed during Regional Meeting Regional Meeting No Presentation or Discussion Regional Vote Providing Feedback to the Committee Communicate directly with your Regional Representative Providing Feedback to the Committee Online Individual Public Comment Regional Meeting Information http://transplantpro.org Regional Administrator Contacts Region Regional Administrator Phone Number E-mail 1,4,9 Shannon Edwards 804-782-4759 Shannon.Edwards@unos.org 2,6,8 Betsy Gans 804-782-4814 Betsy.Gans@unos.org 3,11 Cliff McClenney 804-782-4742 Clifton.McClenney@unos.org 5,7,10 Chrystal Graybill 804-782-4631 Chrystal.Graybill@unos.org







![[#KULRICE-5011] Implement an improved way to delete](http://s3.studylib.net/store/data/008549563_1-dc78b1dd50020d5712604c75a2f497f4-300x300.png)


