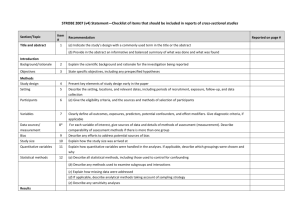
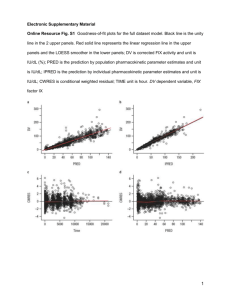
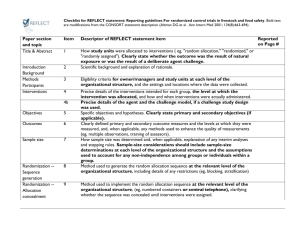
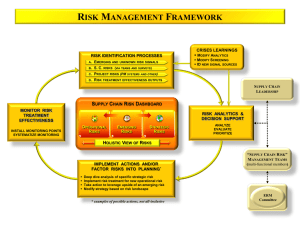
Best practices: Industry Example
advertisement

Best Practices in Modeling and Simulation Why do we need? Industry example Wonkyung Byon Pfizer Global Clinical Pharmacology Best Practices in M&S Great idea! …….well, whytododevelop we Hi Won, we need need any on an internal popguidance PK guidance…. Pop PKare modeling??? and you the chair! 2 Best Practices in M&S Pharmacometrics/MBDD Session 1: Is Model-based Drug Development Delivering on its Promise? Assessing the Business Impact Session 2a: Can Systems Pharmacology Provide a Way Forward in the Search for Novel Treatments in Alzheimer’s Disease? Session 2b: Model-Based Meta-Analysis in Oncology Session 2c: "Curve” balls in Pharmacometrics- Roundtable Session 3: Advances in Pediatric Physiologically Based Pharmacokinetic Modeling in Pediatric Drug Development Session 4a: Catastrophe and Chaos in Pharmacodynamics Session 4b: Population Modeling Using Complex PhysiologicallyBased Models 3 Best Practices in M&S Pharmacometric analyses • Six-fold increase in the number of New Drug Applications that include pharmacometric analyses over a recent 9-year span (from 2000 through 2008) [Lee, J.Y. et al. Clin. Pharmacokinet. 2011] • Enhanced knowledge management and decision-making through model-based drug development [FDA Critical Path 2004] 4 Best Practices in M&S 5 Best Practices in M&S Objectives? Assumptions? Communications? 6 Best Practices in M&S Best Practices/Guidance… Why? Pharmacometrics = Evolving and interdisciplinary science Pharma industry = GLP,GCP,GMP 7 Best Practices in M&S Good “Modeling” Practice Benefit Increased consistency High quality output Greater efficiency Training Risk “One size fits all.” Soon to be outdated 8 Best Practices in M&S FDA/EMA Guidance For Industry • Population Pharmacokinetics • Guideline on reporting the results of Pop PK analyses • Exposure-Response Relationships • End-of-Phase 2A Meetings • Pharmacokinetics and pharmacodynamics in the development of antibacterial medicinal products 9 Best Practices in M&S Recent Publications • Fundamentals of population pharmacokinetic modelling: modelling and software/validation methods [Clin Pharmacokinet. 2012;51(8):515-25/51(9):573-90] • Basic Concepts in Population Modeling, Simulation, and Model-Based Drug Development—Part 1/ Part 2: Introduction to Pharmacokinetic Modeling Methods [CPT: PSP. (2012) 1, e6 / 2: e38] • White Paper: Landscape on Technical and Conceptual Requirements and Competence Framework in Drug/Disease Modeling and Simulation [CPT: PSP. (2013) 2: e40] 10 11 (iii) An update to the current regulatory guidance on dose ranging/finding (see also BOS2 paper) will be debated. (iv) M&S methodology and reporting standards and good practice documents are required (see also BOS4 paper). This will be an industry-led initiative in the first instance, though regulators wish to be involved early in the discussion with the ultimate objective to develop guidance documents. 12 13 Best Practices in M&S Acknowledgement Phylinda Chan Carol Cronenberger Haiqing Dai Jennifer Dong Steve Riley Ana Ruiz Mike K. Smith Kevin Sweeney Mike Tortorici Sandra Allerheiligen Kevin Dykstra Megan Gibbs Rik De Greef Pravin Jadhav Mats Karlsson Lan Ni Daniele Ouellet Vikram Sinha 14 Best Practices in M&S Best Practices Process-related Technical PK/PD modeling Model-qualification Analysis plan/report/unblinding plan templates Strategic Go/No-Go decision-making 15 Coming Soon! 16 Clin Pharm Guidance Population Pharmacokinetic Analyses A to Z of Population PK Analysis ASSUMPTIONS Covariate selection Diagnostics Kowalski Prior knowledge SG HS R KI H NR KN A G E E I A 18 Assumptions/sensitivity analyses Analysis plan and report Leverage prior-knowledge Clinical relevance Communication 19 Challenges? 20 Opportunities! Internal “Wikipedia” page • Hub for collaboration • To facilitate discussions • To capture differing viewpoints • To assist in collaborative writing 21 “Why do you publish?” It provides opportunities to learn and improve “Living document” 22 Best Practices in M&S Good Modeling Practice Benefit Increased consistency High quality output Greater efficiency Training Risk “One size fits all.” Soon to be outdated 23 THANK YOU 24