NO - About RSA Cat
advertisement

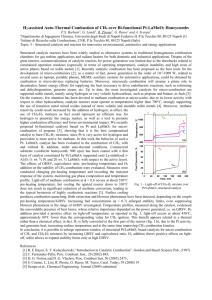
Fireside Combustion Catalyst The Problem SOx & NOx emissions caused by Industrial boilers & power stations: - Pollute the Environment - Toxic & Poisonous - Creates Smog - Boiler Inefficiencies due to poor coal quality Our solution Chinco Fireside Catalyst Our solution Chinco Fireside Catalyst NO capital outlay NO shutdown needed Effeciency & combustion will be increased Conforms to ALL local & international standards Most cost effective solution available Sustainable – 100% Organic-Metallic Compound Non-Toxic, Non-Piosonous & Non-Magnetic No damage to ecosystem if spilled Tried & Tested Chinco Fireside contains natural environmentally friendly additives which result in a highly effective combustion catalyst. Tried and tested in the South African industrial market over many years. The Product Chinco Fireside Treatment. Dosage 200 grams per ton coal. Organic-Metallic Compound. 100% Catalyst. Raw Manganese Ore Base Non – Toxic. Non – Poisonous. Non – Magnetic. No Damage to Land or Ecosystems when Spilled. How does Chinco Fireside Work ? The binding of Sulphur Dioxide (SO2) to the Transition Metal Complexes in the catalyst reduces the SO2 flue gases during the combustion process. The resultant lower SO2 levels reduces acid formation in the flue gas passageways thereby reducing fouling and slagging BENEFITS Chinco reduces particulate stack emissions Chinco enhances combustion, improving boiler efficiency by reducing the Carbon in Ash after combustion Reduced fuel costs Less soot blowing required due to improved combustion process. Saves wasted energy in producing less steam for soot blowing Improved combustion results in less Carbon in Ash thus less handling of ash and less ash disposal BENEFITS cont.. Boiler efficiency improved Ash fusion temperature is raised resulting in a more friable compound instead of hard clinker formation. This is more easily removed with soot blowers. Less insulating fireside deposits and thus less heat transfer problems. • NOx is a by-product of combustion – pollutant contributing to ozone, particulate matter and acid rain. • Thermal NOx is the largest contributor to the overall total NOx. • Under high temperature of combustion (1 425 °Celsius) • Thermal NOx is formed. N2 + O2 + Heat gives Nox Boiler NOx formation NOx Emissions without Catalyst 240 220 200 180 160 140 120 100 80 60 40 20 0 207 109 92 72 38 32 14 July 09 14 July 09 14 July 09 (Test 1) (Test 2) (Test 3) mg/Nm3 ppm NOx Emissions with Catalyst 100 90 80 70 60 50 40 30 20 10 0 21 7 18 24 6 8 16 July 09 16 July 09 16 July 09 (Test 1) (Test 2) (Test 3) mg/Nm3 ppm • Limits by Governments and other organisations on Sox (SO2 + SO3) emissions are being reduced as the harmful effects become more evident. • Studies have found that SO2 is a threat to both human health and the environment. SOx Emissions before Catalyst SOx Emissions after Catalyst