MSA Retreat Presentation CRAO FINAL
advertisement

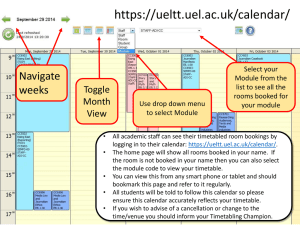
University of Michigan Medical School Calendar Review & Analysis Office (CRAO) Gina Vuocolo-Branch, Director November 1, 2011 Brief History of • June 2000, President Clinton issued an executive memorandum; Medicare will pay for routine care services provided during clinical trials – UMHS Leadership recognizes the need for a Clinical Research compliance office to ensure consistency and compliance to billing regulations • Clinical Research Billing Unit (CRBU) established; now known as CRAO • Hard stop in IRB approval process-Ancillary Committee status • Billing Calendar mandatory – Study activities cannot begin without CRAO approval Clinical Research Billing (CRB) Compliance Billing calendar completion and timely submission of subject enrollment to the Revenue Cycle Research Billing (RCRB) staff is essential to routing charges to the correct payor Billing Calendar Subject Enrollment Billing Compliance What Happens When You Don’t Get It Right? • • • • • • • • • • • • • Northwestern University John Hopkins Hospital Settlement TJU Univ of Louisville Beth Israel Deaconess Medical College of Georgia Univ of CA at San Diego Mayo University of Alabama Dr LaHaye Yale University Rush University $5.5 million settlement $2.6 million settlement $5.4 million cardiac device settlement $2.6 million settlement Mandated to review 1,151 open studies $920,000 settlement $6.1 million settlement $4.7 million settlement $6.5 million $3.4 million $1.1 million settlement 194,000 More than $ 1 million * Exclusion from government contracting, subcontracting or involvement in procurement programs can also be imposed CRB Issues Statistics Clinical Research Billing Issues Reporting 2011 140 125 116 120 109 100 Jul-11 78 80 Aug-11 Sep-11 60 38 40 27 20 15 2 1 0 1 1 0 3 5 1 0 1 0 CRB Issues Received Incorrectly charged Incorrectly charged to patient account to study account Other Avg Days to Close Categories Cost Discrepancy Special Request for account review Open CRB Issues Products and Services • Medicare Coverage Analysis (MCA) • Billing Calendar Review • Review the study protocol, informed consent, billing calendar, budgets, and contract for harmony including removing MSP Conditional Language and follow the UMHS Position Statement for Subject injury payment • Clinical Research Billing Issue resolution • Audit study exempt status • Various education sessions Interfacing Departments Internal Customers IRBMED Study Team Members Grants Office UMHS Revenue Cycle DRDA CCC CTO UMHS Compliance Investigators External Customer eThority Internal Customers Business Development MICHR CTO Office of Research IT Support (MSIS, ITS, MCIT) Subjects University Audit Revenue Cycle Executive Committee Staffing-Current Director Admin Asst 1 2 1 Analysts 1 1 3 2 Asst Analysts CRB Project Mgr CRB Support Asst Coding & Audit Analysts Staffing Trends FY 10 - 6 FTE’s FY 11 - 9 FTE’s Coders moved from UMHS Rev Cycle to reduce process review turnaround time FY 12 – 11FTE’s Implementation of MBECT Training, help desk, training manual, etc. MiChart CRB project Study Statistics July 2010 – June 2011 Current Tactical Initiatives • Final roll out of M Budget Enrollment Calendar Tool (MBECT) – Milestones & Analysis with Invoicing – New platform in 2012 • CRB Policy Implementation • Development of best practice policy & procedures • Identify root causes & trends of CRB issues and implement solutions • Re-design of content of website Efficiency Opportunities • Integrate MCRU billing calendar and budget in MBECT – Negates duplicative work for study team members • Continue collaboration with the IRB and DRDA in order to improve the approval process of studies Thank You Appendix • • • • Mission and Goals Organization Chart Key Performance Indicators Clinical Research Improvements Mission and Goal • Mission: The mission of the Calendar Review & Analysis Office (CRAO) is to complete a Medicare Coverage Analysis (MCA) and Billing Calendar Review for all Human Subject Clinical Trials that have billable items and services to assure consistency with all five core documents to support the appropriate adjudication of charges. • Vision: The CRAO’s vision is to support and collaborate with researchers and study teams to produce a uniform process for the Research Billing Calendar, Budget and Enrollment. We provide stellar customer service and foster partnerships with various other research support areas internal and external to UMHS. • Goal: The CRAO would like to ensure the Clinical Research Billing process is universally utilized throughout the research continuum and is compliant. •1 5 CRB and CRAO Timeline • June 2000 – President Clinton signs executive memorandum to authorizing “(Medicare) payment for routine patient care costs. . .and costs due to medical complications associated with participation in clinical trials” Sept. 2000 – National Coverage Decision (NCD) defined how Medicare will reimburse providers for services to patients on trial 2003 – Rush University Medical Center voluntarily disclosed billing errors related to NCD 2005 – UM Clinical Research Billing Team formed to review and recommend improvements to clinical research billing process November 2006 – Billing calendars required by investigators (no hard stop) January 2007 – CRBU organized under MICHR April 2008 – CRBU re-organized under Office of Research • Rebuilding staff and significant operational changes • Transitioning from ‘policing’ to ‘service’ model March 2009 – Implemented automatic workflow tracking system (Jira) September 2009 – Senior Manager hired and office named changed to CRAO Fall 2009 – Lean principles process March 2010 – Ancillary Committee status in eResearch (eRRM) October 2010 – M Budget Enrollment Calendar Tool (MBECT) roll out Organizational Structure UMMS Office of Research Calendar Review & Analysis Office CRAO Samuel Silver, M.D., Ph.D Medical Director CRAO Assistant Dean Research Associate Medical Director FGP Research Billing Compliance Teri Grieb, Ph.D Senior Director Administration Office of Research Gina Vuocolo-Branch Director Calendar Review & Analysis Office (CRAO) Paula Greene Administrative Assistnat Paulina Radenbaugh CRB System Coordinator Brigette Myers Coding & Auditing Analyst Collin Boyd CRB Support Specialist DeLynn Forman Coding & Auditing Analyst Lynda Hodges Associate Analyst Tina Felt Assistant Analyst Annie Browning Associate Analyst Wendi Schumacher Assistant Analyst Cindy Benedict Associate Analyst Key Performance Indicators • CRAO review turn-around-time • Number of new studies approved by each Analyst • Number of amendments approved by each Analyst • Number of CRB issues – Incorrectly billed as routine care (healthcare plan) – Incorrectly billed to Sponsor/7000 account Clinical Research Improvements • Instituted steeper clinical discounts for professional and hospital fees for clinical research • Implemented lean initiative to remove bottlenecks and increase efficiency of billing calendar workflow • Reduced turnaround time for the CRAO review process for new applications and amendments from 164 days to 15; average is now 13 days for new and 6 days for amendments • Launched clinical research pricing tool to assist study teams in preparing study budgets • Offered training workshops for faculty and study coordinators on the nuts and bolts of billing calendars • Improved access to clinical research support services by moving CRAO, IRBMED, MICHR and CCC Clinical Trials Office to NCRC • Partnered with Hospital to fund mechanism for payment for subject injury when not covered by sponsor (includes NIH) • Established CRAO as an Ancillary Committee within eResearch • Implement of M Budget Enrollment Calendar Tool; a robust clinical trials software application THANK YOU! www.med.umich.edu/u/medschool-crao/mbect.htm