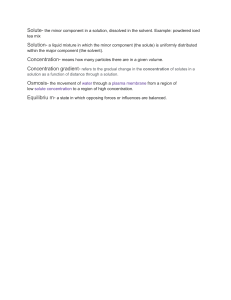
MODULE 3 THE CHEMISTRY OF THE ENVIRONMENT At the end of the module, you should be able to: TLO 7: Apply chemistry to the environment for calculations and design appropriate for water and air. TLO 8: Apply chemistry to the environment for calculations and design appropriate for soil Introduction Environmental chemistry is a branch of chemistry that deals with the study of effect of chemicals on environment. These include the formation of compounds, how chemicals get into the environment, the changes they undergo once introduced into the environment, the number of chemicals in the environment and how they enter the organisms and other things from the environment and the damage they cause. In other words, environmental chemistry deals with the effect of pollutants on the environment and the ways and means to reduce the contamination. This branch of chemistry is the study of pollutants behavior from the environmental point of view. The environmental chemistry is further classified into main areas; measurement of pollutants, and study of the behavior of the contaminants. Environmental chemistry is vital as chemicals introduced into the environment proves to be harmful not only to the environment but also to human health and economy. We have discussed below why environmental chemistry is essential and how it benefits human health, environment and the economy. Unit 1 The Chemistry of Water UNIT LEARNING OUTCOMES TLO 7: Apply chemistry to the environment for calculations and design appropriate for water and air. ENGAGE In its purest form, it's odorless, nearly colorless and tasteless. It's in your body, the food you eat and the beverages you drink. You use it to clean yourself, your clothes, your dishes, your car and everything else around you. You can travel on it or jump in it to cool off on hot summer days. Many of the products that you use every day contain it or were manufactured using it. All forms of life need it, and if they don't get enough of it, they die. Political disputes have centered around it. In some places, it's treasured and incredibly Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 1 difficult to get. In others, it's incredibly easy to get and then squandered. What substance is more necessary to our existence than any other? Water. At its most basic, water is a molecule with one oxygen atom and two hydrogen atoms, bonded together by shared electrons. It is a V-shaped polar molecule, which means that it's charged positively near the hydrogen atoms and negatively near the oxygen atom. Water molecules are naturally attracted and stick to each other because of this polarity, forming a hydrogen bond. This hydrogen bond is the reason behind many of water's special properties, such as the fact that it's denser in its liquid state than in its solid state (ice floats on water). We'll look closer at these special properties later. Water is the only substance that occurs naturally as a solid (ice), a liquid and a gas (water vapor). It covers about 70 percent of the Earth for a total of approximately 332.5 million cubic miles (1,386 million cubic kilometers) [source: U.S. Geological Survey]. If you're familiar with the lines "Water, water, everywhere, nor any drop to drink" from the poem "The Rime of the Ancient Mariner," you'll understand that most of this water -- 97 percent of it -- is undrinkable because it's saltwater. Only 3 percent of the world's water supply is freshwater, and 77 percent of that is frozen. Of the 23 percent that is not frozen, only a half a percent is available to supply every plant, animal and person on Earth with all the water they need to survive [source: National Geographic]. So water is pretty simple, right? Actually, there are a lot of things about it that scientists still don't fully understand. And the problem of making sure that enough clean, drinkable water is available to everyone and everything that needs it is anything but simple. In this article, we'll look at some of these problems. We'll also explore exactly what plants, animals and people do with water and learn more about what makes water so special. EXPLAIN WATER A chemist's view of the world is not as narrow as one might think! Yes, we start with the atom, and then go on to the rules governing the kinds of structural units that can be made from them. We are taught early on to predict the properties of bulk matter from these geometric arrangements. And then we come to H2O, and are shocked to find that many of these predictions are way off, and that water (and by implication, life itself) should not even exist on our planet! But we soon learn that this tiny combination of three nuclei and eight electrons possesses special properties that make it unique among the more than 15 million chemical species we presently know. When we stop to ponder the consequences of this, chemistry moves from being an arcane science to a voyage of wonder and pleasure as we learn to relate the microscopic world of the atom to the greater world in which we all live. Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 2 The molecule of water A molecule is an aggregation of atomic nuclei and electrons that is sufficiently stable to possess observable properties — and there are few molecules that are more stable and difficult to decompose than H2O. In water, each hydrogen nucleus is bound to the central oxygen atom by a pair of electrons that are shared between them; chemists call this shared electron pair a covalent chemical bond. In H2O, only two of the six outer-shell electrons of oxygen are used for this purpose, leaving four electrons which are organized into two non-bonding pairs. The four electron pairs surrounding the oxygen tend to arrange themselves as far from each other as possible in order to minimize repulsions between these clouds of negative charge. This would ordinarily result in a tetrahedral geometry in which the angle between electron pairs (and therefore the H-O-H bond angle) is 109.5°. However, because the two non-bonding pairs remain closer to the oxygen atom, these exert a stronger repulsion against the two covalent bonding pairs, effectively pushing the two hydrogen atoms closer together. The result is a distorted tetrahedral arrangement in which the H—O—H angle is 104.5°. Although the water molecule carries no net electric charge, its eight electrons are not distributed uniformly; there is slightly more negative charge (purple) at the oxygen end of the molecule, and a compensating positive charge (green) at the hydrogen end. The resulting polarity is largely responsible for water's unique properties. Because molecules are smaller than light waves, they cannot be observed directly, and must be "visualized" by alternative means. This computer-generated image comes from calculations that model the electron distribution in the H2O molecule. The outer envelope shows the effective "surface" of the molecule as defined by the extent of the cloud of negative electric charge created by the eight electrons. Hydrogen bonding The H2O molecule is electrically neutral, but the positive and negative charges are not distributed uniformly. This is illustrated by the gradation in color in the schematic diagram here. The electronic (negative) charge is concentrated at the oxygen end of the molecule, owing partly to the nonbonding electrons (solid blue circles), and to oxygen's high nuclear charge which exerts stronger attractions on the electrons. This charge displacement constitutes an electric dipole, represented by the arrow at the bottom; you can think of this dipole as the electrical "image" of a water molecule. Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 3 As we all learned in school, opposite charges attract, so the partially-positive hydrogen atom on one water molecule is electrostatically attracted to the partiallynegative oxygen on a neighboring molecule. This process is called (somewhat misleadingly) hydrogen bonding. Notice that the hydrogen bond (shown by the dashed green line) is somewhat longer than the covalent O—H bond. This means that it is considerably weaker; it is so weak, in fact,that a given hydrogen bond cannot survive for more than a tiny fraction of a second. Sticky, Wet Water Water has some unusual properties due to its hydrogen bonds. One property is cohesion, the tendency for water molecules to stick together. The cohesive forces between water molecules are responsible for the phenomenon known as surface tension. The molecules at the surface do not have other like molecules on all sides of them and consequently they cohere more strongly to those directly associated with them on the surface. For example, if you drop a tiny amount of water onto a very smooth surface, the water molecules will stick together and form a droplet, rather than spread out over the surface. The same thing happens when water slowly drips from a leaky faucet. The water doesn't fall from the faucet as individual water molecules but as droplets of water. Another important physical property of water, is adhesion. In terms of water, adhesion is the bonding of a water molecule to another substance, such as the sides of a leaf's veins. This process happens because hydrogen bonds are special in that they break and reform with great frequency. This constant rearranging of hydrogen bonds allows a percentage of all the molecules in a given sample to bond to another substance. This grip-like characteristic that water molecules form causes capillary action, the ability of a liquid to flow against gravity in a narrow space. An example of capillary action is when you place a straw into a glass of water. The water seems to climb up the straw before you even place your mouth on the straw. The water has created hydrogen bonds with the surface of the straw, causing the water to adhere to the sides of the straw. As the hydrogen bonds keep interchanging with the straw's surface, the water molecules interchange positions and some begin to ascend the straw. Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 4 Adhesion and capillary action are necessary to the survival of most organisms. It is the mechanism that is responsible for water transport in plants through roots and stems, and in animals through small blood vessels. Hydrogen bonds also explain why water’s boiling point (100°C) is higher than the boiling points of similar substances without hydrogen bonds. Because of water’s relatively high boiling point, most water exists in a liquid state on Earth. Liquid water is needed by all living organisms. Therefore, the availability of liquid water enables life to survive over much of the planet. Furthermore, water has a high specific heat because it takes a lot of energy to raise or lower the temperature of water. As a result, water plays a very important role in temperature regulation. Since cells are made up of water, this property helps to maintain homeostasis. The melting point of water is 0°C. Below this temperature, water is a solid (ice). Unlike most chemical substances, water in a solid state has a lower density than water in a liquid state. This is because water expands when it freezes. Again, hydrogen bonding is the reason. Hydrogen bonds cause water molecules to line up less efficiently in ice than in liquid water. As a result, water molecules are spaced farther apart in ice, giving ice a lower density than liquid water. A substance with lower density floats on a substance with higher density. This explains why ice floats on liquid water, whereas many other solids sink to the bottom of liquid water. In a large body of water, such as a lake or the ocean, the water with the greatest density always sinks to the bottom. Water is most dense at about 4°C. As a result, the water at the bottom of a lake or the ocean usually has temperature of about 4°C. In climates with cold winters, this layer of 4°C water insulates the bottom of a lake from freezing temperatures. Lake organisms such as fish can survive the winter by staying in this cold, but unfrozen, water at the bottom of the lake. SOLUTIONS Most of the materials that we encounter in everyday life are mixtures. Many mixtures are homogeneous; that is, their components are uniformly intermingled on a molecular level. Homogeneous mixtures are called solutions. Examples of solutions abound in the world around is. The air we breathe is a solution of several gases. Brass is solid solution of zinc and copper. The fluids that run through our bodies are solutions, carrying a great variety of essential nutrients, salts, and other materials. Solutions are extremely important. throughout the living world, solutions are necessary for maintenance and survival. In the human body, nutrients are transported in solution, while waste products are removed as solutions. In plants, all internal organs are constantly bathed in moisture; in photosynthesis, a wet surface is required on which gases can diffuse materials to the body. Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 5 A solution, since it is homogeneous, will have the same properties all throughout No matter where we take a sample from a solution, we will obtain materials with same composition as that of any sample from the solution. SOLUTION - is a homogeneous mixture of two or more substances * Homogeneous - only one phase; no part is separated by another by a detectable boundary. 2 components 1. solute - dissolved substance; present in smaller amount 2. solvent - dissolving medium; present in greater amount Aquation or Hydration - when water is used as a solvent SOLUBILITY - amount of solute that can be dissolved in a given amount of solvent at a specific temperature; usually expressed as grams of solute per 100 grams of solvent soluble - substance that dissolves in the solvent insoluble - substance that does not dissolve in the solvent solute solubility (grams solute/100 grams solvent) less than 0.1 0.10 - 1.0 1.0 - 10 greater than 10 qualitative solubility description insoluble slightly soluble soluble very soluble I. FACTORS AFFECTING SOLUBILITY A. SOLID in LIQUID 1. Shaking or Agitation 2. Powdering or Pulverizing 3. Temperature 4. Nature of Reactants - “like dissolves like” B. LIQUID in LIQUID 1. Miscible - dissolve in any amount in each other ex. alcohol + water; oil+ gasoline/CCl 4 2. Partially miscible - have limited solubility in each other ex. tincture of iodine + water 3. Immiscible - do not dissolve in each other and forms two separate layers upon mixing ex. oil + water C. GAS in LIQUID 1. Pressure - affects gases only * Henry’s Law - the weight of a gas dissolved by a given amount of solvent is directly proportional to the pressure exerted by the as wen in equilibrium with the solution. 2. Temperature Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 6 II. TYPES OF SOLUTIONS A. According to state or phase 1. Gaseous Solution Solute Solvent gas gas liquid gas solid gas 2. Liquid Solution Solute Solvent gas liquid liquid liquid solid liquid 3. Solid Solution Solute gas liquid solid Solvent solid solid solid Example air water vapor in air dust particle in air Example carbonated drinks alcohol in water sugar in water Example hydrogen in palladium metal amalgam alloy B. According to solubility 1. Unsaturated solution - a solution that can still take more of the solute in addition to what it already contains; less concentrated than a saturated solution 2. Saturated Solution - contains the maximum amount of solute that can be dissolved in the given amount of solvent; contains the maximum amount of dissolved solute necessary for the existence of an equilibrium between dissolved and undissolved solute 3. Supersaturated solution - contains more of the solute that it normally contains at an elevated temperature; more concentrated than a saturated solution C. According to the amount of dissolved solute 1. Concentrated solution - contains greater amount of solute 2. Dilute solution - contains lesser amount of solute; concentrated solution + water III. WHY SUBSATANCES DISSOLVE? SOLUTION FORMATION A. solute - solute attraction - attraction between solute particles B. solvent - solvent attraction - attraction between solvent particles * driving force for solution formation solute - solvent attraction - attraction between solute and solvent particles IV. METHODS OF EXPRESSING CONCENTRATION OF SOLUTIONS concentration - amount of solute present in a specified amount of solvent or solution A. PERCENTAGE METHODS 1. Percentage by weight or mass (%w/w) - most frequently used by chemists Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 7 ( ( ) ) 2. Percentage by volume (%v/v) - used when both solute and solvent are liquids 3. Percentage by weight-volume (%w/v) - usually encountered in hospitals and industrial settings - used when working with a solid solute and liquid solvent B. MOLE FRACTION (yi) C. MOLARITY (M) or FORMALITY (F) - most often used in chemical laboratories M = number of gram-molecular weight (mol) of solute per liter of solution F = number of gram-formula weight (mol) of solute per liter of solution D. NORMALITY (N) - number of gram-equivalent weights of solute per liter of solution - most often encountered in neutralization reactions f = factor (equivalent/mol) 1. acid: f - number of replaceable or ionizable H+ ex. HCl f=1 H2SO4 f=2 H3PO4 f=3 HC2H3O2 f=1 Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 8 2. base: f - number of replaceable or ionizale OHex. NaOH f=1 Ca(OH)2 f=2 Al(OH)3 f=3 3. salt: f - total + or - valence ex. NaCl f=1 CaBr2 f=2 Al2S3 f=6 E. MOLALITY (m) - used in experimental situations where changes in temperature are concerned - number of moles of solute per kilogram (kg) of solvent V. DILUTION CONCEPT A common problem encountered when working with solutions in the laboratory is that of diluting a solution of known concentration (usually called a stock solution) to a lower concentration. Dilution - a process in which more solvent is added to a solution in order to lower the concentration of the solution. The amount of solute present is now distributed in a larger amount of solvent. Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 9 Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 10 Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 11 Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 12 EXPLAIN Activity 1: 1. 2. 3. The Unique Properties of Water Self-Assessment No. 1 Enumerate and discuss the unique properties of water. Give examples for each. Water is the so-called universal solvent. Give everyday examples of these aqueous solutions. How would you able to increase the degree of saturation of a sugar solution? To be submitted in Google classroom on: ELABORATE & EVALUATE Activity 2: Solutions Self-Assessment No. 2 1. Compute the weight (g) of solute needed to make the solutions listed below: a. 1250 mL of 0.85 M AlCl3 b. 0.500 L of 9.25 N K2SO4 c. 350 g of 13.2 %w/w of Ca(OH)2 2. Calculate the %w/v of a solution made by dissolving 22.0 g CH 3OH (methanol) in C2H5OH (ethanol) to make 100 mL solution. 3. What is the %v/v of 10.0 mL of CH 3OH (methanol) dissolved in water to a volume of 40.0 mL? 4. A solution is prepared at 25oC by mixing 20 g of K2SO4 with 150 cc of water. The density of water at this temperature is 1 g/cc. What is the m of K2SO4? 5. Express the formality (F) the concentration of each of the following solutions: a. 28.4% NH3 by weight, having a density of 0.808 g/mL b. 69.5% HNO3 by weigt, aving a density of 1.42 g/mL 6. An aqueous solution of hydrofluoric acid is 12.5 molal with respect to HF and has a density of 1.070 g/mL at 25oC. Calculate a. mole fraction of HF b. %w/w of HF c. molarity d. molality 7. Calculate the volume of concentrated reagent required to prepare the diluted solutions indicated: a. 15 M NH3 to prepare 50 mL of 6.0 M NH3 b. 18 M H2SO4 to prepare 250 mL of 10.0 M H2SO4 8. Calculate the molarity of the solutions by mixing 250 mL of 0.75 M H 2SO4 with a. 150 mL of H2O b. 250 mL of 0.70 M H2SO4 To be submitted in Google classroom on: Prepared by: Engr. N. L. Escalante Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise of any part of this document, without the prior written permission of SLU, is strictly prohibited. 13