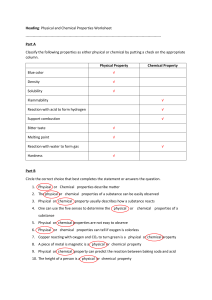
Reaction Rates Experiments: Temperature, Concentration, Catalysts
advertisement

Group 3 Experiment 1: Investigating the Effect of Temperature on the Rate of Chemical Reaction Using Food Coloring and Water Objective: To explore how temperature influences the rate of a chemical reaction using food coloring and water. Materials: Three beakers Hot water Food coloring (preferably a dark color Stirring rod or spoon for better visibility) Stopwatch or timer Cold water Dropper Tap water Procedure: 1. Start by dissolving the food coloring in tap water in the small beaker. 2. Put a 200 mL of cold water, tap, and hot water in the separately in the three beakers. 3. Add the same amount of food coloring (around 1 drop) into each beaker. 4. Once the food coloring is added, simultaneously start the stopwatch or timer two beakers three beakers. 5. Observe and record the time it takes for the food coloring to completely disperse in each beaker. Record your observations on the provided data table below. Data Table: Water Temperature Time for Food Coloring to Disperse (seconds) Cold Water 2 minutes and 07.77 secs Tap Water 1minute and 05.02 secs Hot Water 40.72 secs Discussion Questions: 1. Compare the time it took for the food coloring to disperse in each temperature of water. The food coloring dispersed the fastest in hot water (40.73 seconds), followed by tap water (1 minute and 5 seconds), and lastly, cold water (2 minutes and 7 seconds). 2. How does temperature affect the rate of the chemical reaction in this experiment? The food coloring dispersed fastest in hot water, followed by tap water, and slowest in cold water. This shows that higher temperature leads to a faster rate of the chemical reaction between the food coloring and water. 3. Can you relate this experiment to any real-world applications where temperature influences chemical reactions? For example, you are making a hot chocolate. When you mix cocoa powder into hot milk, it dissolves fast because the milk is hot. But if you mix cocoa powder into cold milk, it takes longer because cold milk makes it dissolve slower. So, just like in the experiment, temperature can change how fast things dissolve, like in making hot chocolate. Conclusion: We observed that higher temperatures result in faster disperse of the food coloring, while lower temperatures lead to slower disperse. This shows that the rate at which a substance disperses is influenced by the temperature. Experiment 2: Investigating the Effect of Concentration on the Rate of Chemical Reaction Using Vinegar and Baking Soda Objective: To explore how changing the concentration of baking soda affects the rate of the chemical reaction with vinegar. Materials: Vinegar (acetic acid) Baking soda (sodium bicarbonate) Measuring spoons Two small containers or beakers Stopwatch or timer Procedures: 1. Pour the same amount of vinegar into the two beakers. You can use about 50ml of vinegar for each beaker. 2. Using the two spoons, measure half a tablespoon of baking soda on the first spoon and measure a full tablespoon of baking soda on the other spoon. 3. After measuring the right amount of baking soda, pour the two spoons in the separate beakers at the same time. 4. Immediately start the stopwatch or timer when the baking soda comes contact into the vinegar 5. Observe and record the time it takes for the reaction to reach completion. Note any visible changes, such as fizzing or bubbling. 6. Record your observations on the provided data table below. Data Table: Baking Soda Concentration Half Tablespoon Full Tablespoon Time for Reaction to Complete (seconds) 28.56 secs 34.06 secs Discussion Questions: 1. How does changing the concentration of baking soda affect the rate of the chemical reaction with vinegar? Changing the amount of baking soda used changes how fast the reaction happens with vinegar. In our experiment, we saw that using more baking soda made the reaction a bit quicker than using less. This shows that adding more baking soda can make the reaction with vinegar speed up. 2. Can you explain the mechanism behind the reaction between vinegar and baking soda? When vinegar and baking soda mix together, they react to make bubbles and fizz. This happens because vinegar contains something called acetic acid, and baking soda has a substance called sodium bicarbonate. When they meet, they react and make carbon dioxide gas, which forms the bubbles we see. So, the bubbling and fizzing we observe are because of this reaction between the vinegar and baking soda. 3. How might this experiment be modified to investigate other factors that affect the rate of the reaction? To explore other factors affecting reaction rates, modify the experiment by adjusting variables such as temperature, stirring intensity, or using different concentrations of vinegar. Conclusion: We observed that using more baking soda makes the reaction with vinegar happen faster. This shows that the amount of baking soda affects how quickly the reaction occurs. Comparing the times for reactions with different amounts of baking soda helps us understand how concentration affects reaction speed. Worksheet: Investigating the Effect of Surface Area on the Rate of Chemical Reaction Using Vitamin C Tablets Objective: To explore how changing the surface area of vitamin C tablets affects the rate of a chemical reaction. Materials: Vitamin C tablets (ascorbic acid) Mortar and pestle (for grinding) Water Stopwatch or timer Two small containers or beakers Procedures: 1. Measure equal amounts of water into each container. You can use about 100ml of water for each. 2. Prepare one whole vitamin C tablet and grind the other vitamin C tablet into a fine powder using the mortar and pestle. 3. At the same time, pour the whole tablet and the powdered vitamin C in the separate container 4. Immediately start the stopwatch or timer when the tablets come into contact with water. Stir each container gently to facilitate the dissolution of the vitamin C. 5. Record the time it takes for the tablets to dissolve completely in each container. Record your observations on the provided data table below. Data Table: Vitamin C Tablet Surface Area Whole Tablet Pulverized Tablet Time for Reaction to Complete (seconds) 2 mins and 30.62 secs 1 min and 01.05 secs Discussion Questions: 1. Compare the time it took for took for the tablets to dissolve completely in each container. The whole tablet took longer to dissolve compared to the pulverized tablet. 2. Analyze the relationship between the surface area of the vitamin C tablet and the rate of the chemical reaction. Breaking the tablet into a fine powder makes more of its surface area touch the water. That's why the powdered tablet dissolved faster than the whole one. This is because the water molecules can reach more of the tablet's parts when it's in smaller pieces. So, the bigger the surface area of the tablet, the quicker it dissolves in the water. 3. How does changing the surface area of the vitamin C tablet affect the rate of the chemical reaction with water? Increasing the surface area by grinding the tablet enhances the rate of the chemical reaction. The pulverized tablet dissolved more rapidly, indicating that a larger surface area facilitates quicker interactions with water molecules. Conclusion: The experiment illustrates that altering the surface area of vitamin C tablets impacts the rate of the chemical reaction with water. The pulverized tablet, with increased surface area, dissolved more quickly than the whole tablet. This emphasizes the importance of surface area in influencing the speed of chemical reactions, as a larger surface area allows for more efficient interaction between the reactants. Worksheet: Investigating the Effect of Catalyst on the Rate of Chemical Reaction Using Vinegar and Baking Soda Objective: To explore how the presence of a catalyst affects the rate of a chemical reaction between vinegar and baking soda. Materials: Vinegar (acetic acid) Baking soda (sodium bicarbonate) Two empty bottles with narrow openings Balloons (two) Measuring cup Funnel (optional) Stopwatch or timer Procedures: Part 1: Preparation Pour 50 ml of vinegar into each empty bottle. Part 2: Setting Up the Experiment Stretch the opening of one balloon over the mouth of each bottle, ensuring a tight seal. Place a small amount of baking soda (about one teaspoon) inside one of the balloons. Ensure that both balloons are securely attached to the bottles, with no leaks or gaps. Part 3: Conducting the Experiment Carefully lift the balloon containing baking soda and shake it gently to allow the baking soda to fall into the vinegar. Observe and record any changes in the balloon's size and shape. This indicates the production of gas due to the chemical reaction. Start the stopwatch or timer as soon as the baking soda enters the vinegar. Monitor the balloons closely and record the time it takes for the balloon with baking soda to inflate fully. If the balloon inflates fully, stop the timer and note the time. If not, record the maximum inflation observed within a reasonable time frame. Part 4: Observations and Data Collection Record the time it takes for the balloon with baking soda to inflate fully or to reach its maximum inflation. Record your observations on the provided data table below. Data Table: Setup Time for Balloon to Inflate (seconds) With Catalyst (Baking Soda) 20.25 secs Without Catalyst 0 Discussion Questions: 1. What is a catalyst, and how does it affect the rate of a chemical reaction? A catalyst is a substance that speeds up a chemical reaction without being used up in the process. In this experiment, baking soda acts as a catalyst. It helps the reaction between vinegar and baking soda happen faster by providing a different way for the reaction to happen. 2. Can you explain the chemical reaction that occurs between vinegar and baking soda? When vinegar and baking soda mix together, they create bubbles and fizz. This happens because vinegar has something called acetic acid, and baking soda has sodium bicarbonate. When they come into contact, they react and produce carbon dioxide gas, water, and sodium acetate. So, it's like a fizzy reaction that you can see in the balloon. 3. How might this experiment be modified to investigate other factors that affect the rate of the reaction? We can try different things to see what makes the reaction faster or slower. For example, we can use more vinegar or baking soda to see if it changes how fast the balloon blows up. Or, we can try changing the temperature of the ingredients to see if that makes a difference. Conclusion: We observed that the presence of a catalyst (baking soda) affects the rate of the chemical reaction between vinegar and baking soda. We observed that when baking soda was present, the balloon inflated much faster compared to when it was absent. This demonstrates the role of a catalyst in speeding up chemical reactions. DOCUMENTATION Members: Rei Anne Narag Maricel Rodriguez Jamillah Carlos Mary Jane Tumanguil Alcher Baccay Johndale Parallag Renzel Velasco Adrian Pamittan Rodge Mora Christian Palogan* (absent)