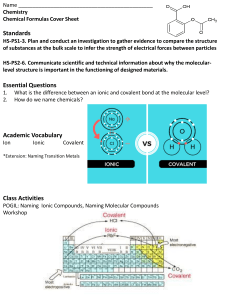
Naming ionic and covalent compounds Memorize these rules well before your session. Introduction to chemistry Issued by startmyclass. (SMC) Below are some of the rules that students need to keep in mind while naming ionic and covalent compounds. Remember! there are different set of rules for ionic and covalent compounds. We shall first look at the rules we should remember while naming Ionic compounds. 😃 Rules for ionic compounds. Mono-atomic cations have no change. (e.g Na+ can be simply called a sodium ion.) Mono-atomic anions have the suffix -ide at the end. (e.g F- can be called fluoride ion.) There are two important cases for ionic compounds. 1. Cations that can only assume a single oxidation state will follow the following naming template: cation + anion (with the suffix ide.) Although transition metals don’t follow these rules, we have some exceptions including Zinc, Al, and Ag. KI Potassium iodide. MgCl2 Magnesium chloride. Al2(CO3)3 Aluminium carbonate. NH4OH Ammonium hydroxide. Remember some polyatomic ions like OH and CO3 have special names and can used as such without adding the suffix -ide. Student can find the list of common polyatomic ions at the end of these notes. 2nd case: When cations can adopt more than one oxidation states, it wise to mention their oxidation states within the name. This is for all transition metals and larger group 3A and 4A metals. They will follow the following naming template : cation (oxidation state in roman numerals) + anion (with suffix- ide). FeBr2 Iron (II) bromide. Co(OH)3 Cobalt (III) hydroxide. PbO2 Lead (IV) oxide. Pb2O3 Lead (III) oxide. Rules for covalent compounds. Name the less electronegative element first, followed by the name of the more electronegative element with the suffix -ide. Many covalent compounds have common names like water, ammonia etc. Mention the number of each type of atom with the greek prefixes. 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa REMEMBER ! mono is not to be used for the 1st atom in the formula. CO Carbon monoxide. (no mono used for the 1st atom in the formula.) CO2 Carbon dioxide. PCl3 Phosporus trichloride. CCL4 Carbon tetrachloride. N2F4 Dinitrogen tetrafluoride. Whenever you are given a compound to name, determine whether its an ionic compound or a covalent compound. Use the rules accordingly. 🧑🎓 How to determine whether a compound is ionic or covalent in nature. Ionic compounds. 1. Will have large electronegativity difference. Usually a metal is paired with a non-metal. Covalent compounds. 1. Will have small electronegativity difference. A non-metal with another nonmetal or a metalloid.