MTD LIMITED PUBLICATION 90/102
Design and operational guidance on cathodic
protection of offshore structures, subsea
installations and pipelines
@
MTD Ltd 1990
ISBN 1870553 04 7
THE MARINE TECHNOLOGY DIRECTORATE LIMITED
19 Buckingharn Street, London WC2N 6Ef, UK Telephone 071-321 0674 Fax 071-930 4323
Foreword
The project leading to this guidance document was undertaken by UEG with
specialist authors under contract. It was funded jointly by the UK Department of
Energy and UEG, and was completed and published by MTD Ltd as a part of the
arrangements for MTD Ltd's absorption of UEG. At UEG, the Project Manager
for the work was Mr J d e Prey and at h4TD Ltd, Mr R W Barrett. The
Coordinating Editor for the project was Mr J N Wanklyn.
A Steering Group comprising potential users, specialists, section authors, the
Department of Energy, WEG and then MTD Ltd, provided the forum for
discussion and commented on the guidance document prior to publication. The
Steering Group comprised:
Mr 3 A Bray (Chairman)
Mr M D Allen
Mr B Balmer
Mr R W Barrett
Mr J A Clarkson*
Dr R A Connell
Dr R F Crundwell
Mr J de Prey'
Dr D Fairhurst
Dr B V Johnson
Mr M G Lunt
Mr D Shaw'
Mr R J Simpson
Mr D Wilson'
Marine Technology Support Unit
Spencer & Partners
BP Exploration
The Marine Technology Directorate Ltd
Brown & Root Vickers Ltd
Shell UK Exploration & Production
Pasminco Europe (Impalloy) Ltd
UEG
BP International Ltd
BP International Ltd
Department of Energy
John Brown Engineers & Constructors Ltd
The Steel Construction Institute
BWE Services Ltd
Section authors
Mr D Ames'
Dr V Ashworth
Mr B Bell'
Mr P Gammage
Dr R P M Procter
Professor K F Sander
Dr D Scantlebury
Mr L Skilton
Mr J L Tischuk
Mr J N Wanklyn
Mr G D Webb'
Mr N J M Wilkins
Mr B S Wyatt
Spencer & Partners
Global Corrosion Consultants Ltd
J P Kenny & Partners Ltd
Brown & Root Vickers Ltd
Corrosion & Protection Centre, WMIST
Consultant
Corrosion & Protection Centre, WMIST
John Brown Engineers & Constructors Ltd
Tischuk Enterprises
Consultant
J P Kenny & Partners Ltd
Consultant
Corrosion Control Services
+ No longer with this organisation
Every reasonable effort has been made to ensure that the guidance given in this
publication is based on the best knowledge available up to the time of finalising the
text. However, no responsibility of any kind for any injury, delay, loss or damage
can be accepted by MTD Ltd, the WK Department of Energy or others involved in
its publication. it is not intended for use as a mandatory or contractual document.
Cathodic protection of offshore structures
3
Preface
The principles of cathodic protection were clearly understood and concisely expressed by
Sir Humphrey Davy as long ago as 1824. At that time, Sir Humphrey was President of
the Royal Society, and his attention had been drawn by the Navy Board to the rapid
decay of the copper sheathing used as a cladding for the hulls of ships which were
constructed of wood and consequently highly susceptible to penetration by “teredos”
(wood borers). In this connection, Volta had discovered, in 1800, a method of generating
an electric current by means of a Voltaic Pile, and in 1832-1833 Faraday had put forward
the 1st and 2nd laws of electrolysis. Both Volta’s and Faraday’s discoveries formed the
basis for modern electrochemistry.
In his Bakerian lecture of 1806 Davy had advanced the hypothesis that “chemical
attractions may be exalted, modified or destroyed by changes in the electrical state of
bodies; that substances will only combine when they are in different electrical states; and
that by bringing a hody naturally positive artificially into a negative state its usual powers
of combination are altogether destroyed”. This statement summarises concisely the hasic
principles of cathodic protection.
In this paper to the Royal Society, which was read on 22 January 1824 Davy stated:
“Copper is a metaI only weakly positive in the electro-chemical scale; and
according to my ideas it would only act upon sea water when in the positive state;
and, consequently if it could be rendered slightly negative the corroding action of
sea water upon if would be nil”.
Davy then points out that this statement would apply irrespective of the purity of the
copper, and goes on to consider how it could be effected in practice. “I at first thought
of using a Voltaic cell (Volta’s original Voltaic pile consisted of alternate discs of copper
and zinc separated by pieces of cloth and immersed in diluted sulphuric acid) but
considered it hardly applicable in practice”. He then tried various combinations of
copper coupled to different metaIs and found that a piece of zinc as large as a pea or the
point of a small iron nail was adequate to preserve to 40 or 50 square inches (260 or 320
cm2) of copper, irrespective of its geometrical form.
In this paper to the Royal Society, Davy stated that the Lord Commissioners of the
Admiralty had given him permission to use ships of war to ascertain the practical value of
his results, and the first ship to be cathodically protected was the HMS Samarang in
which iron blocks were used successfully as anodes to protect the copper.
Thus Davy was responsible for establishing the principles of cathodic protection, and he
was the first to use sacrificial anodes to protect another metal. He also foresaw the use of
electrical power for protection, but his ideas at that time were in advance of technology.
Cathodic protection of offshore structures
13
Sacrificial anodes for protecting copper sheathing were used for only a relatively short
time, and although the precise reason is not known it has to be remembered that it was
about the time when the wooden hulls of warships were being replaced by wrought iron.
Another view is that in preventing the corrosion of copper it also weakened its antifouling properties! Cathodic protection then became dormant for about 100 years until
the early 1930s,when the oil companies in Texas used an impressed current system and
scrap iron anodes to protect underground pipelines. Today, it is quite usual for the time
interval between a discovery and its practical application to be only 10 to 20 years.
Control of the corrosion of North Sea offshore platforms by cathodic protection has
resulted in the rejection of many well established principles based on experience gained
in the protection of other structures. Thus it has been accepted that the most economical
method of using it was in conjunction with a protective coating, and that it was more
economical to use sacrificial anodes for small structures and impressed current for large.
To assess the position, the Cathodic Protection Study Group carried out by means of
questionnaire a survey of the experience gained by the operators using sacrificial anodes
(zinc or aluminium), impressed current or hybrid systems. All of them gave reasonable
protection with the sacrificial anode system, which was the most popular, proving to be
the most satisfactory. In the case of the impressed current system, the major problems
were mechanical and electrical rather than inadequate current distribution, in particular,
failure or operation resulting from disbonding of the cables. Only one operator used a
paint coating, and it appeared that protection of the bare structure did not result in
excessive consumption of anode material.
Over the years, and since the more widespread use of cathodic protection in the early
1930s for protecting underground pipes, there have been many developments in anode
design and construction. In the case of impressed current systems, the use of graphite as
a conducting anode material has declined with the development of composite anodes in
which platinum is used economically in the form of a thin coating on either titanium or
niobium.
As early as 1920, G Baum patented an anode (USPatent 1,477,009)consisting of
tantalum partly coated with a thin layer of platinum for the anodic oxidation of sulphate
to persulphate. In fact, many of the anodes used for impressed current cathodic
protection originate from those used in previously in electrolytic oxidation processes (e.g.
lead dioxide, magnetite, oxides of the platinum metals, etc).
In the case of sacrificial anodes, emphasis has been on the formulation of zinc,
aluminium and magnesium alloys which give the most negative potential and maximum
anode efficiency.
14
MTD Ltd Publication 901102
Although there have been a number of improvements in the technology of using cathodic
protection, it needs to be emphasised that this also applies to other methods of corrosion
control. An example is the Thames FIood Barrier, in which it had been envisaged that
the conventional methods of coatings and cathodic protection would be used for all
interior and exterior surfaces of steel in contact with Thames water. However, in the case
of the rising sector gate, the use of anodes was precluded because of the very small
distance of separation between the steel gate and the concrete sill which is about 225 mm
where the gate is supported by the gate arm, decreasing to about only 100 mm at the
centre.
For this reason, cathodic protection could not be used, and protection had to be confined
to a thick coal-tar epoxy coating which was formulated to resist the highly abrasive
conditions which occur by Thames water containing silt rushing through the gap when
the gate is raised into the defence position. The barrier has been in service for about 5
years, and it is understood that has been very little deterioration in the steelwork.
It is made clear from the title that this guide is intended primarily for offshore structures,
subsea installations and pipelines, and it is probably the most comprehensive single
publication dealing with these topics. However, it is apparent to me after reading the
page proofs that its scope is very wide indeed, and that it should be of value to all those
who are concerned with the protection of steeI structures in marine environments.
L.L. Shreir
Cathodic protection of offshore structures
15
Contents
LIST OF ILLUSTRATIONS
8
LIST O F TABLES
10
NOTATION
11
PREFACE
13
INTRODUCTION
17
1.1
1.2
Background
Scope of the guidelines and suggestions for its use
PRINCIPLES OF CORROSXON AND CATHODIC
PROTECTION OFFSHORE
2.1
2.2
2.3
2.4
2.5
2.6
2.7
2.8
2.9
2.10
Introduction
Aqueous corrosion
Polarisation diagrams
Corrosion of steel in sea water and seabed mud
The principles of cathodic protection
The application of cathodic protection
Protection criteria
Operating current densities
Possible adverse effects ofcathodic protection ofsteel
The need for uniform potential distribution
EFFECTS OF CATHODIC PROTECTION ON MECHANICAL
PROPERTIES OF STEELS
3.1
3.2
3.3
3.4
3.5
3.6
Introduction
Environmentallyassisted cracking
Effects of cathodic protection on environmental cracking
Susceptible materials
Effects of cathodic protection on corrosion
fatigue of low C-Mnsteels
Effects of cathodic protection on hydrogen embrittlement
of offshore materials
17
17
19
21
21
24
25
29
31
34
35
39
40
41
43
43
45
46
46
56
ORGANIC COATINGS AND CATHODIC PROTECTION
59
Introduction
Interaction of cathodic protection and coatings
Failure of coatings
Influence of rust and contamination at the interface
Shielding of anodes
Special coatings
Recent experience with coatingr
61
63
64
69
70
70
71
4.1
4.2
4.3
4.4
4.5
4.6
4.7
Cathodic protection of offshore structures
5
5.
CALCULATION AND MODELLING FOR THE DESIGN
OF CATHODIC PROTECTION SYSTEMS
5.1
5.2
5.3
5.4
5.5
5.6
5.7
6.
75
76
78
81
83
85
87
Lntroduction
"Traditional" design method
Formulation of the mathematical problem
The steel-sea water boundary
The mathematical model
Computational methods
Analogue models
CATHODIC PROTECTION SYSTEMS FOR STEEL
89
OFFSHORE STRUCTURES
6.1
6.2
6.3
6.4
6.5
6.6
7.
91
91
93
102
119
128
129
Cathodic protection of steel in concrete
North sea experience
Conclusions
131
131
136
144
147
152
CATHODIC PROTECI'ION SYSTEMS FOR SUBSEA
INSTALLATIONS AND PIPELINES
153
8.1
8.2
8.3
8.4
8.5
8.6
8.7
8.8
6
Introduction
Design objectives
General considerations
Carrying out the design
Engineering
Operating manual
CATHODIC PROTECTION SYSTEMS FOR CONCRETE
OFFSHORE STRUCTURES
7.1
7.2
7.3
7.4
7.5
7.6
8.
73
Introduction
The concrete environment
Corrosion of steel in concrete
Overview of systems for submarine pipelines
The place of cathodic protection in pipeline design
Design requirement for pipelines
Anode materials
System design calculations
Anode design and attachment
Pipeline interfaces
Monitoring of pipeline cathodic protection
155
157
159
165
169
176
181
183
MTD Ltd Publication 901102
9.
OPERATION, MONITORING AND SURVEYING OF
CATHODIC PROTECTION SYSTEMS
9.1
9.2
9.3
9.4
9.5
9.6
9.7
10.
STRUCTURAL ASPECTS OF CATHODIC PROTECTION
10.1
10.2
10.3
10.4
10.5
10.6
11.
Introduction
Location of anodes on fixed steel platforms
Weight aspects of the cathodic protection system
Wave action and cathodic protection
Attachment derails for anodes
Structural design of anodes
CURRENT LEGISLATION STANDARDS AND
GUIDANCE DOCUMENTS
11.1
11.2
11.3
12.
Introduction
Design review
Initial or commissioning survey
Fixed monitoring systems
Periodic surveys
Assessment of requirements for modification or retrofit
Conclusions
Legislation
Guidance documents
Key to the documents
REVIEW OF RECENT EXPERIENCE IN THE NORTH SEA
12.1
12.2
12.3
12.4
12.5
Introduction
Objectives
Procedure
Results
Discussion
189
191
192
192
193
202
207
208
209
211
211
216
217
219
220
223
225
227
238
239
24 1
243
243
244
244
REFERENCES
261
APPENDIX ESSENTIAL DESIGN INFORMATION
27 1
Cathodic protection of offshore structures
7
leIntroduction
1.1 BACKGROUND
The project was undertaken in response to an initiative by the Department of Energy
to build on the work of their Cathodic Protection Study Group (CPSG). That Group
was set up by the Department to review the practice of cathodic protection on
offshore oil and gas installations in UK waters, to consider the adequacy of existing
standards and rules, to identify requirements for further information, and to advise
on action required.
The CPSG sat from November 1979 until April 1982. It circulated an extensive
questionnaire to all major off3hore operators. Its conclusions reflected both the
practices adopted and the degree of corrosion actually being experienced at that time.
A recommendation of the CPSG called for the production of a "comprehensive
design and operation manual for cathodic protection systems for the North Sea".
These guidelines are aimed at meeting that recornmendation.
The guidelines were produced by a collaboration between specialist authors and
members of the Project Steering Group. They included several who had participated
in the original CPSG, others in possession of recent operational experience of CP in
the North Sea,and potential users.
The objective is to provide, in a single, widely available document, practical guidance
to designers and operational staff on the design, installation and operation of effective
cathodic protection systems offshore. The guidelines are intended to be of use to
engineers who are not CP specialists but who need familiarisation, also to be available
as a source book for specialists.
1.2 SCOPE OF THE GUIDELXNES AND SUGGESTIONS FOR THEIR USE
Three needs are answered by these guidelines.
First, they provide necessary background material for any engineer who encounters
cathodic protection of offshore structures either directly or indirectly. Section 2
provides the link between the principles of corrosion and with the practical aspects.
Section 9 provides guidance on the commissioning, operation, monitoring and
surveying of CP systems.
Second, guidance is given to members of design teams through every step of the
design process. All Sections refer to relevant documents, but Section I1 lists and
comments on the principal documents containing current legislation, standards and
guidance. Section 6 restates the fundamental design objective and discusses factors
affecting the choice of design criteria. Section 7 describes the properties of concrete,
leading to a discussion on the special features which characterise the electrochemical
corrosion of steel embedded in concrete. The experience of nine operators of
Cathodic protection of offshore structures
17
concrete structures is presented, particularly on how CP system performance has
compared with the original design. Section 8 reviews design requirements for the
cathodic protection of subsea installation and pipelines. Section 10 discusses the
effect of CP on the design of steel structures, Section 12 presents a review of existing
CP design, operation and monitoring practice on North Sea and other UK waters oil
and gas fixed steel platforms, utilising the results of a questionnaire updating the data
collected originally by the CPSG.
Third, various aspects are addressed in detail. Section 3 covers the effects of CP on
mechanical properties such as corrosion fatigue and hydrogen ernbrittlement. Section
4 covers organic coatings. Section 5 covers the calculation and modelling for the
design of CP systems.
18
MTD Ltd Publication W102
Section 2 Principles of corrosion and cathodic protection
offshore
Cathodic protection of offshore structures
19
2. Principles of corrosion and cathodic protection offshore
2.1
INTRODUCl'lON
This section provides an introduction to the principles of aqueous corrosion and
cathodic protection. The objective is to provide the link between the principles
and the practical application of cathodic protection to offshore structures.
Because corrosion is an electrochemical phenomenon, it is necessary to introduce
some elementary electrochemistry, the point being that it requires more than an
appreciation of eIectrica1 phenomena to understand and use cathodic protection
efficiently. Later sections of this publication deal with such diverse matters as the
concept of a protection potential, the inevitable variability of the protective current
density, the importance of coatings in many cases, and the nature and effect of the
calcareous deposit. This Section aims to show how each of these topics derive from
the principles of the subject.
2.2
AQUEOUS CORROSION
Aqueous corrosion is simply the chemical reaction of a metal with its aqueous
environment. The word "aqueous" may be translated fairly freely: the metal does
not have to be immersed in liquid water for aqueous corrosion to occur. For
example, open sea water, seabed mud, concrete, and even onshore soil or sand can
represent an aqueous environment from the point of view of corrosion.
It is helpful to consider a simple corrosion reaction to understand the nature of
corrosion more readily. The corrosion of steel in aerated sea water can be
represented by:
2Fe
steel
+
+ 2H,O
+ dissolved + water
0,
oxygen
-
2Fe++
+
40H'
ferrous
ions
+
hydroxyl
ions
In practice, the ferrous ion (Fe") is likely to oxidise further to ferric ion
( F e + + + )then to react with the hydroxyl ion (OH-) to produce insoluble ferric
hydroxide (Fe(OH),) which may loosely be called rust. However, Equation (2.1)
represents the primary chemical reaction of corrosion, and it is this reaction we
should consider further.
It is convenient to consider the metallic and non-metallic reactants in Equation
(2.1) separately:
2Fe
0,
-+
2Fe++
2H,O
+
+
4e4e--
40H'
Cathodic protection of offshore structures
21
To balance Equation (2.2) in terms of both mass and charge, it was necessary to
add four electrons to the right hand side. Likewise, to balance Equation (2.3), four
electrons were added to the left hand side. This reduces the cbemical reaction of
corrosion (Equation (2. I)) to two electrochemical reactions (reactions containing
both chemical entities and electrons). However, the sum of the two
electrochemical reactions yields the overall corrosion reaction, and the electrons
disappear.
Thus, it may inferred that aqueous corrosion is a chemical reaction which occurs by
an electrochemical mechanism. The inference is not confirmed by the foregoing
analysis, but all the available evidence points towards it. Perhaps the strongest
evidence is found in the efficacy of cathodic protection in controlling many
examples of aqueous corrosion.
T h e electron release reaction (Equation (2.2)) is referred to as an anodic reaction:
it is always responsible for the consumption of the metal. The electron
consumption reaction (Equation (2.3)) is referred to as a cathodic reaction, and it
is also responsible for consumption of constituents of the aqueous environment.
When corrosion occurs, there is no build-up of electrical charge. Electrons are
released and consumed at the same rate, The rate of the anodic reaction therefore
needs to be electrically equivalent to the rate of the cathodic reaction. Thus,
notwithstanding the fundamental importance of the anodic reaction in describing
metal loss and therefore corrosion, it becomes clear that the anodic reaction does
not occur without a viable cathodic reaction. It follows that the rate of the latter, if
it is sluggish, can control the rate of the former and thereby the rate of corrosion.
2.2.3 Model of aqueous corrosion
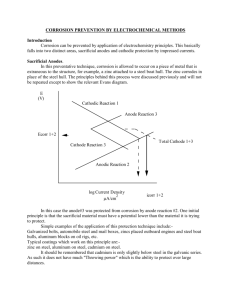
Figure 1 provides a schematic illustration of the corrosion reaction. Here the two
iron atoms dissolve from the surface at anodic sites to produce two ferrous ions in
solution, and each leaves two electrons in the metal. The electrons migrate through
the metal to another surface site (a cathodic site) where they react with incoming
oxygen and water to form hydroxyl ions. Finally, the negatively-charged hydroxyl
ions and the positively-charged ferrous ions migrate towards one another.
For significant corrosion to occur, this sequence of events has to occur many
trillions of times per second on 1 cm2 of surface. If the anodic reaction occurs
randomly over the metal surface, the corrosion attack is more or less uniform. If
the anodic reactions are restricted to a relatively small area on a large metal
surface, highly localised corrosion results. Inhomogeneities at the metal surface can
provoke this localisation of the anodic site. The presence of second phases in t h e
metal, small discontinuities in solid corrosion product, pinholes in metallic or
organic coatings, junctions of dissimilar metals, concentration or temperature
differences between different points of the surface, etc. are typical of such
inhomogeneities. All may lead to localised attack.
22
MTD Ltd Publication 90/102
O,* 2H,O
/
Corrosive
environment
Surface
t
Figure 1 Schematic representation of aqueous corrosion
Electr
!
potential
E,
Ecorr
/
\Cathodic
k tnetics
Ea
Figure 2
Polarisation diagram schematically representing the electrochemistry of
aqueous corrosion
Cathodic protection of off3hore structures
23
The schematic diagram (Figure 1) demonstrates that corrosion is a surface
phenomenon (i.e. it only occurs at the surface, and i t may therefore be
progressively affected by the changes which the corrosion reaction causes at that
surface). For example, if the corrosion product is soluble, so that it can freely
diffuse away from the surface, the corrosion rate may remain constant over long
periods. As soon as reactants become in short supply, the corrosion rate begins to
fall. If the corrosion product is sparingly soluble, or if it is unable to escape from
the surface for some reason, it is likely to accumulate as a solid product and stifle
the corrosion reaction. Consequently, a decreasing corrosion rate with time is
observed. Complete stifling is a highly desirable occurrence, but, as we have seen
above, imperfect protection of this type may lead to localised corrosion.
2.3 POLARISATION DIAGRAMS
For a clearer understanding of corrosion, and to be able to analyse a corrosion
situation, it is useful to formalise the corrosion reaction by graphical
representation. To achieve this, it is necessary to consider corrosion further.
Corrosion is a spontaneous process and, like aIL spontaneous phenomena, it occurs
because there is an energy advantage to be obtained. At equilibrium, all
electrochemical reactions exert an equilibrium electrode potential which depends
on the environmental conditions. Where corrosion occurs, the potential (voltage)
difference between the equilibrium potentials of the two electrochemical reactions
seeking to establish on the same surface provides the driving force for the reaction.
As a result, both electrode reactions are forced from their condition of equilibrium,
a net anodic reaction occurs in one case and a net cathodic reaction in the other.
The two potentials approach one another, because all or part of the voltage
difference is used in driving the individual reactions. For corrosion to be
spontaneous, not only does there have to be a difference between the two
equilibrium potentials, but the equilibrium potential of the putative cathodic
process has to be more positive, or less negative, than that for the anodic process.
These circumstances are represented schematically in Figure 2,which is a plot of
electrode potential, E, against the logarithm of the reaction rate. Because anodic
and cathodic reactions release and consume electrons, respectively, the anodic and
cathodic reaction rates are represented by an electrical current, I. For simplicity,
both reaction rates are plotted on the same axis, the direction of the current is
ignored, and only the magnitude is used to represent rate.
E, is the equilibrium potential for the cathodic reaction (e.g. for Equation (2.3)).
At E, there is no net cathodic reaction rate. The negative-going cume from E,
shows how, as the potential gets more negative, the cathodic reaction rate gets
faster. Conversely, E, is the equilibrium potential for the anodic reaction (e.g. for
Equation (2.2)), and again at E, there is no net anodic reaction. The positivegoing curve from E,, shows how, as the potential becomes more positive, the anodic
reaction gets faster.
24
MTD Ltd Publication 90/102
It was shown earlier that when corrosion occurs, the anodic reaction rate is exactly
equal to the cathodic reaction rate. It was not mentioned before, but it is true, that
in environments of good conductivity (e.g. sea water, seabed mud, etc.) the
corroding metal displays a single potential which lies between E, and E, In
Figure 2, this condition is met where the anodic and cathodic curves cross. The
potential at the crossover point is referred to as the corrosion potential, Ewm. It is
the single potential exerted by a corroding metal referred to above. The current,
ZCOtr,
is referred to as the corrosion current, and it is an electrical representation of
the corrosion rate. In practice, a corroding metal does not take up potential Ea or
E,, but spontaneously moves to E,,,,.
.
While the shape o f the individual E - log Z curves may vary, depending on
environmental conditions, the manner in which the diagrams, so-called polarisation
diagrams, are interpreted in terms of Em,, and I,,, remains the same.
2.4
CORROSION OF STEEL IN SEA WATER AND SEABED MUD
The corrosion of steel in sea water, or seabed mud can be adequately represented
by Equation (2.1), although the process normally proceeds to the precipitation of
ferric hydroxide.
On clean steel in sea water, the anodic process occurs with greater facility than the
cathodic. In consequence, the corrosion reaction can go no faster than the rate of
cathodic, oxygen reduction (Equation (2.3)). The latter usually proves to be
controiled by the rate of arrival of the oxygen at the metal surface, which, in turn,
is controlled by the linear water flow rate and the dissolved oxygen concentration
in the bulk sea water.
This may be represented on a polarisation diagram (Figure 3). At first, the
cathodic kinetics get faster as the potential becomes more negative from E,. This
has the effect of depleting the oxygen immediately adjacent to the metal surface,
thus rendering the reaction more difficult. Ultimately, a point is reached where the
surface concentration of oxygen has fallen to zero, and oxygen can then only be
reduced as and when it reaches the surface. Further lowering of the potential
cannot increase the cathodic reaction rate, because the kinetics are now governed
by potential-independent diffusion processes, A plateau, or limiting, current is
observed. Figure 3 shows that the corrosion rate is then equal to this limiting
current. The limiting current can be increased by increasing the oxygen flux, either
by raising the bulk oxygen concentration (the concentration gradient gets steeper)
or increasing the flow rate (the oxygen-depleted layer gets thinner). Both serve to
increase the corrosion rate as shown in Figure 3.
To a first approximation, it may be stated that the rate of corrosion of clean steel in
aerated sea water under turbulent flow conditions is directly proportional to the
bulk oxygen concentration and the linear velocity. Fick's First Law of Diffusion
and the Chilton-Colbourn Analogy can be used to calculate the precise effect of
oxygen concentration and Reynolds number (flow rate) on corrosion('). Using the
technique, Ashworth(') estimated the maximum corrosion rates of clean steel in
North Sea water at 7OC as shown in Table 1 (on page 27).
Cathodic protection of offshore structures
25
Electrc
!
potential
Note:
= Illn
Increasing f l o w o r oxygen concentration
Figure 3
Polarisation diagram representing control of corrosion rate by sluggish cathodic
kinetics (in this case controlled by the rate of arrival of oxygen at the surface)
and the effect of increasing oxygen availability
In practice, corrosion products and marine fouling build up on steel as it corrodes
in sea water. These generally produce lower corrosion rates.
Rowfands(2)suggests that the published experimental data indicate that, although
the corrosion rate of fully immersed steel is fairly rapid in the first few months of
exposure, it falls progressively with time, A value of 0.13 m d a may be taken as
reasonably representative in any part of the world. However, pits may grow at 3 to
10 times that rate.
Rowlands cites data collected by L a Q ~ e (which
~l
largely derive from small exposure
panels. Many marine structures, particularly those in shallow waters, are
simultaneously exposed to a number of discrete corrosive environments: the
marine atmosphere, the splash zone, the tidal zone, the fully submerged zone and
tbe mud zone. Rather different results to those discussed by Rowlands(*) are
obtained when large or interconnected panels are simultaneously exposed to a
26
MTD Ltd Publication 90/102
TABLE 1 ESTIMATED MAXIMUM CORROSION RATES OF CLEAN STEEL IN
NORTH SEA WATER AT 7OC
CORROSION RATE ( m d a )
0, concn
9PPm
1OPPm
0.107
0.120
0.134
0.107
0.123
0.138
0.154
0.096
0.111
0.128
0.144
0.160
0.6
0.104
0.121
0.138
0.156
0.174
1
0.120
0.140
0.160
0.179
0,199
2
0.160
0.187
0.213
0.240
0.266
4
0.240
0.280
0.320
0.360
0.400
6PPm
7PPm
0
0.080
0.094
0.3
0.091
0.4
flowrate
number of these zones. Commonly, the corrosion rate data are represented
schematically as in Figure 4, which derives largely from the work of Humble('),
although his experimental data are not quite so striking. The peak corrosion rates
are found immediately below the mean low tide zone and in the splash zone. The
typical mean corrosion rate in the splash zone, given quiet sea conditions, is
estimated(') as 0.25 to 0.75 m d a .
The difference between linked and isolated submerged test panels is most marked
below the mean low tide zone. The peak corrosion rate shown here in Figure 4 is
often attributed to galvanic action between steel in contact with the oxygen-rich
surface waters (the cathodic area) and the steel at somewhat greater depth exposed
to waters of lesser oxygen content (the anodic area). It is difficult to conceive that
the cbange in oxygen concentration with depth is sufficiently great to cause the
effect,and it may be that other factors come into play. Nevertheless, while the
explanations may remain in doubt, the general observation has been widely
substantiated.
Cathodic protection of offshore structures
27
Atmospheric zone
---------- ----------SDlash zone above
h;gh tide
....................
I
I n t e r t i d a l zone
--------___"I-------(
Continuousty
submerged zone
Mean low tide
P
-
Mud line
-----I--------------
Mud zone
Relative m e t a l thickness l o s s
Figure 4
Corrosion profile of steel piling alter 5 years exposure (alter Humble
t4')
It was noted above that macrofouling fauna and flora may help to reduce the
corrosion rate of steel. They may achieve this by further reducing the oxygen flux
to t h e surface. By contrast, they may have an adverse effect, because, if the fouling
is not continuous, pits may develop, and it is also possible for the fouIing species to
damage protective coatings. Finally, decay of the organic matter can lead to the
production of aggressive species (e.g. acids), and it may produce locally anaerobic
conditions which favour the metabolism of sulphate-reducing bacteria (SRB)
which have been shown to assist some corrosion processes.
Bacteria begin to colonise exposed metals almost as soon as they are placed in sea
water. Like the macrofouling, the slimes developed can inhibit oxygen transfer to
the surface, but equally the bacteria may produce aggressive metabolic products
such as acids and hydrogen sulpbide. In practice, it is not possible to make
corrosion experiments in live sea water without developing the bacterial colonies.
Thus, it may be taken that all the published marine corrosion data reflect their
presence.
In seabed mud, the mobility of dissolved oxygen is much reduced, furthermore it is
difficult for corrosion products to diffuse away. The corrosion rates should be
lower. However, anaerobic conditions may readily develop. Because these are
widely infected by SRB,the possibility of microbially-assisted corrosion is high.
On the basis of extensive experiments, Stott(6)gives as his view, that 0.5 mm/a may
be considered to be a realistic mean corrosion rate for mild steel when microbiallyassisted corrosion occurs. He regards 1.5 m d a as an upper limit, but he points out
28
MTD Ltd Publication 90/102
that the attack is highly localised and that penetration rates four of five times these
values may be locally anticipated. In extreme cases, corrosion rates in mud may be
comparable to, or exceed, those found in sea water. It should be borne in mind
that the sea bed around fixed structures may be supplied with nutrients from waste
materials, including oil-based drilling muds, which can promote the growth of SRB.
2.5
THE PRINCIPLES OF CATHODIC PROTECTION
By examining Equations (2.2)and (2.3), it is possible to infer what would happen if
an electrical intervention were made in a corrosion reaction. For example, if
electrons were removed from the metal surface through the bulk metal, it might be
expected that the potential of that surface would become more positive. Any
process releasing electrons at the surface would speed up, and any process
consuming electrons at the surface would slow down (Le. dissolution (Equation
(2.2)) would accelerate and oxygen reduction (Equation (2.3)would get slower).
This is not at all what is required!
Conversely, if electrons are supplied to the surface from the bulk metal, the
potential is expected to fall, the rate of dissolution to diminish and the rate of the
cathodic process to increase. The metal would therefore corrode less, and it would
then be said to be under cathodic protection. An increase in the cathodic reaction
rate is a consequence of cathodic protection.
Figure 5 shows a schematic polarisation diagram for the steel corroding with
oxygen reduction as the cathodic process. The corrosion potential is given by Ecorr,
and the corrosion rate, Icarr,is equaI in magnitude to the limiting current, Ilim,for
Note: At E , , p , c o r r o s mis reduced from I,,,, to I ,
by application of a current equal to C - A
At E, ,corrosion is reduced to zero by
e potential
applied cathodic current equal to the
limiting current
Anodic kinetics
C
Figure 5
Schematic diagram showing how corrosion can be reduced or stopped by
applying cathodic profecfion
Cathodic protection of ofhhore structures
29
oxygen reduction. Suppose the potential could be lowered from E,,, to E,,, the
anodic reaction rate would be lower at fp and the overall cathodic reaction rate
would remain equal to Ilim.However, the cathodic reaction now receives its
electrons from two sources (via the anodic process and from an external source of
current). The anodic reaction provides electrons equivalent to Ipand the
additional current required (C minus A) is provided from an external supply,
If the potential is lowered to E,, the dissolution stops completely and the cathodic
reaction rate remains Ilim.There being no anodic reaction at this potential, the
cathodic reaction now has to be sustained by the external electron supply alone,
and it therefore takes more supplied current (equal to (Airn - 0)) to reach E,, than to
reach E,,.
If the potential is moved below E,, Figure 5 shows that no further reduction in
dissolution is possible. Apparently, it shows too that there is no further increase in
the applied current demand. However, eventually, a second cathodic process,
which plays no part in the free corrosion reaction, becomes energetically viable at
these more negative potentials, This is the electrolysis of water to yield hydrogen
gas :
2H,O
+
2e-
-
20H-
+
H,
(2.4)
and the cathodic current increases very rapidly.
Thus we see that:
without cathodic protection the steel corrodes at a rate given by Ilim.
.
.
.
.
by lowering the potential to E,, it is possible to stop corrosion completely, but
this requires application of an external current approimately equal to Ilim.
by lowering the potential below E, the metal remains free from corrosion, but
this requires application of an external current approximately equal to I,im.
by lowering the potential to a value below E,,,, but above E,, the metal may be
partially protected with some economy in current supplied.
because the anodic E - log I curve is approximately logarithmic, a small initial
negative shift in potential (or polarisation) can give a large benefit in terms of
protection. A further shift of equal magnitude is 10 times less effective: the
benefits of further increments of polarisation become increasingly nugatory.
The whole foregoing discussion considers the fairly uniform corrosion of carbon
steels, but the application of cathodic protection to stainless steels should be briefly
mentioned. These materials have a much higher resistance to uniform corrosion,
but they are prone to severe attack within crevices and below deposits. For this
reason, they should be protected to the same potentials as carbon steels. Their
current density requirements may be expected to be similar to those discussed
earlier, though there is some evidence that the current density required by stainless
steels falls less with time than is the case with carbon steels.
30
MTD Ltd Publication W102
2.6
THE APPLICATION OF CATHODIC PROTECTION
There are two methods whereby eleceons can be supplied to polarise the surface:
using sacrificial anodes or an impressed current technique. The emf to drive the
current is electrochemical in origin in the case of sacrificial anodes and derives
from a dc generator in the impressed current technique.
2.6.1 Sacrificial anode method
If two metals are connected together while in contact with a suitable electrolyte
solution, a voltaic or galvanic cell is produced. Tbe open circuit driving voltage
derives from the natural potential difference which exists between the two metals.
If the circuit is closed, the potential difference drives an electrical current. The
more negative electrode behaves as an anode: it releases electrons to the circuit
and dissolves more rapidly, the more positive electrode behaves as a cathode and
dissolves less. The use of so-called sacrificial anodes in cathodic protection
capitalises on this phenomenon.
Assuming the structure to be protected is made of steel, sacrificial anodes made of
alloys of zinc, aluminium or magnesium are used to form the cell, because these
metals are, in appropriate circumstances, less noble (more negative) than steel.
The anode attachment to the structure is made through a steel core onto which the
anode material is cast. Thus the structure is in metallic contact with the anode
material and also in electrolytic contact with it once the structure is immersed.
This is represented in Figure 6, where it is seen that the four electrons released by
the dissolution of two zinc atoms are consumed in the cathodic reduction of oxygen
on the structure and hydroxyl ions are produced at the structure surface. Many
such electrochemical events, both simultaneous and sequential, lead to the full and
continuous protection of the structure. Figure 7 shows a number of aluminium
alloy anodes installed on a structure prior to launch.
Sacrificial anodes are discussed more fully in Section 6.
2.6.2 Impressed current method
In the impressed current method, the driving voltage is provided by a dc power
source rather than a spontaneous cell (Figure 8). As a result, there is no reason
why the anode metal should be more negative than the structure. Indeed, there is
an advantage in the material being more positive (i.e. more noble). Noble metal
anodes do not dissolve readily on anodic polarisation but sustain alternative anodic
reactions which involve decomposition of the environment, typically of water or
dissolved chloride ions in it:
2H,O
and
2C1'
-
0,
C1,
+
+
4H'
+
4e-
2e'
As a result, the anode materials remain virtually unconsumed, but hydroxyl ions
are still produced at the surface of the structure.
Cathodic protection of offshore structures
31
Protected structure
sea water
/in
2e
d 2 inc alloy
anode
,Steel
Figure 6
Figure 7
Representakion oi cathodic protection using
a zinc alloy sacrificial anode on a structure
in sea water
Sacrificial anodes installed in space
frame prior to launch
(courtesy lmpalloy Ltd)
I n s u l a t e d cathode cable
I
Protected
s t ruc t u re
Figure 8
32
core
'Power supply [ dc 1
* '
I.
I.
Insulated anode cable
-~ll~
Impressed current anode
Representation 01 impressed current cathodic protection using inert anode in
sea wafer
MTD Ltd Publication 901102
Shreir and Hayfield(') provide an excellent review of the types and properties of
impressed current anodes.
Figure 9 shows a platinised-titanium anode, with its armoured electrical
connection, installed on a structure prior to launch.
As Figure 8 shows, while the anodic reaction is often rather different, the
impressed current installation is similar to, and performs in every way like, a
sacrificial anode system. However, because the driving voltage is substantially
larger than in a sacrificial anode installation, the current provided hy an individual
anode is very much greater. This fact features substantially in the merits and
demerits of the two systems,
2.6.3 Choosing a cathodic protection system
The choice between installing a sacrificial anode system or an impressed current
system depends on the importance of the merits and demerits of the two
approaches .
Sacrificial anodes have the advantages that they work independentIy of a source of
electrical power, they cannot be incorrectly attached to the structure, their output
depends on their composition and shape so there is no current control function to
exercise, it is difficult to over-polarise the structure, and, with careful anode
distribution, it is easy to obtain uniform cathodic polarisation.
The advantages of the impressed current technique are that it is possible to have a
large adjustable driving voltage, so that relatively few anodes need to be installed
even to protect large, uncoated structures in high resistivity environments.
Figure 9
Platinised titanium impressed current
anode installed on structure member
prior to launch
(courtesy lmpalloy Ltd)
Cathodic protection of offshore structures
33
The disadvantages of sacrificial anodes include the high labour cost of installing a
large anode burden, and the very high cost of installing additional anodes in sihc to
make good a deficient installation. The disadvantages of tbe impressed current
system include the need for a reliable dc power supply, the fact that protection
cannot begin until power is available, the danger of over protection (given badly
placed anodes or poor voltage control), the difficulty of obtaining uniform cathodic
polarisation on complex shapes, and the possibility that the power supply may be
incorrectly attached, producing anodic polarisation of the structure.
A sumey of British offshore practice and experience@)reveals that, in general,
sacrificial anode systems have performed better in the North Sea than impressed
current systems. The main adverse comments on sacrificial anodes referred to
evidence of under design and deficient anode attachment. Impressed current
systems were regarded as mechanically and electrically unreliable because of
problems with anode performance and the integrity of cables. All these are
deficiencies of the design rather than intrinsic problems of either system.
2.7
PROTECTION CRITERIA
For reasons given in Section 2.8, the cathodic current supplied to a structure, or
even the current density, is a poor indicator of protection. It is practice to use a
potential criterion. This may be justified by accumulated experience and finds
support in Figure 5 where, providing the potential is reduced to E,, corrosion
ceases.
The accepted criterion for full protection of steel in aerated sea water is a polarised
potential of -0.80 V measured with respect to a silver/silver chloriddseawater
reference electrode (see Section 6.3.4). That value finds support with DIIV(’~’’) and
NACE(11”2).However, under anaerobic conditions (e.g. some seabed muds),
because of the ossibility ofmicrobially-assisted corrosion, a value of -0.90 V is
In the case of high strength steels (yield strength3700 MPa), it
is important to avoid over-polarisation (see Section 2.9), and for this reason the
potential should fall within a potential window between -0.80 V and -0.95 V. In
quoting the protection potential, the value is given with respect to a silver/silver
chloriddseawater reference electrode.
It is not possible to directly measure the electrode potential of a metal in an
aqueous solution, This is because such potentials are a measure of the electrical
potential difference between the metal and the aqueous environment in which it
stands. To measure this difference, the metal needs to be connected to one side of
a voltmeter and the aqueous environment to the other. To achieve the latter
connection, a metal conductor has to be inserted into the aqueous phase. This
conductor introduces its own electrode potential, which inevitably becomes
included in the measured value. The problem is resolved by using a conductor of
reproducible and defined electrode potential (i.e. a so-called reference electrode).
The emf of the cell made between the reference electrode and the metal in the
given environment is therefore measured when an attempt is made to measure the
electrode potential. It now becomes clear that to have any meaning the electrode
potential has always to be quoted with respect to a defined reference electrodes.
34
MTD Ltd Publication 90/102
-0.40
Intense corrosion
--___----------------------------------------------Freely corroding
-0.60
blistering o-f paints and increasing danger of
structure ernbri ttlement
0,
-2 -1.10
->
.-m - 1 . 2 0
c
e
al
c
-1 30
J
Figure 10
The corrosion cathodic protection and over-protection regimes expressed as
function of electrode potential
There is a range of reference electrodes available, but the silver/silver chloride
reference electrode is most commonly used in sea water. A second, less precise,
electrode used in sea water is a clean zinc block. Figure 10 gives an indication of
the values of the same electrode potential measured against both of these reference
electrodes. Detailed descriptions of reference electrodes are given in Section 9.4.1.
As an alternative protection criterion, NACE(1'.'2)suggest a negative potential shift
of 300 mV when the current is switched on. It is difficult to see where this holds
any advantage over the straightforward potential criterion referred to above. It
certainly takes no account of the possibility of microbially-assisted corrosion or
over-polarisation of high strength steels.
2.8
OPERATING CURRENT DENSITIES
It was shown that the corrosion rate of clean steel in sea water is controlled by the
flux of oxygen to the surface (i.e. the rate approximates to the limiting current given
(Figure 3). It was also shown (Figure 5) that the external current required for full
protection approximates to the limiting current. Table 1 records the maximum
corrosion rate under specific conditions, and it follows that if that rate is converted
to an equivalent current density, the value represents the maximum cathodic
current density required to protect the steel. Using Faraday's Laws of electrolysis,
the data of Table 1 have been converted to give the current densities in Table 2.
Cathodic protection of offshore structures
35
TABLE 2 ESTIMATED MAXIMUM CURRENT DENSITY REQUIRED TO
PROTECT CLEAN STEEL IN NORTH SEA WATER A T 7OC
CATHODIC PROTECTION CURRENT DENSITY (mNm2)
O2 concn
6PPm
7PPm
8PPm
9PPm
1OPPm
91
120
114
flowrate
0
68
0.3
78
91
105
118
113
0.4
82
85
109
123
136
0.6
a9
103
118
133
148
1
102
119
136
153
170
2
136
159
182
205
227
4
205
239
273
307
341
~
1
I%e increase of current density with oxygen concentration and flow rate reflects the
effect of these factors on the limiting current (Figure 3). It is clear that the current
density required is not a matter of choice, but that it is a function of the
circumstances and environment of exposure. Clearly, it is unsatisfactory as an
indicator of adequate protection. This is why potential proves so much more
satisfactory.
2.8.1 Mest of coatings
While organic coatings (paints) are not entirely impermeable to the oxygen and
water, they do restrict corrosion when applied to the surface of the metal. The
bulk of the corrosion on a painted surface occurs not beneath the intact coating,
hut at the base of small pinholes in it.
If cathodic protection is applied to a painted surface, the coating acts as a
substantial resistive barrier to current flow and what protective current does pass
enters at the pinholes. From the foregoing, it is clear that the paint does most of
the protection and the cathodic protection system reinforces it at the weak spots.
Coatings and cathodic protection are truly complementary.
On most structures, it is not regarded as economic to install cathodic protection
without applying a good quality coating. Thus marine pipelines usually have such a
coating. In the North Sea, it was unusual to coat production platforms. This
36
MTD Ltd Publication 90/102
reflects, in part, the difficulty of programming a complex coating exercise into a
tight onshore fabrication schedule and the uncertainties over the long-term
performance of coatings on structures designed to operate for more than 25 years.
The latter point highlights the fact that, although the application of a coating
llsaves" current, the cathodic protection system needs to be sufficiently flexible to
accommodate an increasing current demand with time as the coating degrades or is
damaged. DXIV(~)
offer some arbitrary guidance in this area (see Section 11).
Thus, for a 30-year life and a coated structure in an environment demanding
120 mA/m2 for bare steel, the initial, mean and final current densities are
recommended to be 2.4, 30 and 72 mA/m2, respectively. While the predictions are
probably pessimistic, the data underline the flexibility required in the design for
very large coated marine structures. They also demonstrate the substantial current
savings a coating permits.
2.8.2 Effwt of calcareous deposits
When cathodic protection is applied, the cathodic reaction is stimulated. Thus,
according to Equation (2.3), an excess of hydroxyl ion develops at the steel surface.
Where excessive polarisation occurs and hydrogen gas is produced, the production
of hydroxyl ions is increased still further (Equation 2.4).
Sea water contains bicarbonate ions which form a pH-dependent equilibrium with
carbonate ions:
An increase in hydroxyl ion concentration (a rise in pH) displaces the equilibrium
in favour of the carbonate ion (COi-1. Thus, when cathodic protection is applied,
the carbonate ion concentration in the near-surface solution increases.
The sea water also contains calcium ions which react to form an insoluble product,
caIcium carbonate, with carbonate ions:
Increase in the concentration of either ion favours precipitation of calcium
carbonate. It follows that cathodic protection in sea water can lead to deposition of
calcium carbonate at the protected surface.
-
The sea water also contains magnesium ions which form an insoluble hydroxide:
Mg*+
f
20H-
Mg(OH),
Thus magnesium hydroxide may also form at a cathodically protected surface.
Cathodic protection of offshore structures
37
In practice, both products can, and do, form to produce what is called a calcareous
deposit. Calcium carbonate deposits the more readily (i.e. with a smaller interfacia1
pH rise) and therefore forms at low current densities. Magnesium hydroxide
deposits less readily (i.e. not until the interfacial pH has risen to about 9.3 from the
natural value of 7.5 to 8.3). This can only be achieved at higher current densities.
The calcareous deposits interfere with oxygen mass transfer to the surface. They
are therefore protective and, rather like a paint film, they reduce the current
required for cathodic protection. In general, deposits rich in calcium carbonate are
more protective than those rich in magnesium. Thus scales formed at lower
current densities tend to be more protective. This is because the magnesium
hydroxide introduces mechanical weakness into the scale and at the current
densities required to produce it, hydrogen is generated which disrupts the scale as
it escapes from the surface(”).
However, over the current density range where the scales formed are
predominantly calcium carbonate, the higher the current density the thinner and
more compact is that scale and the better the protection it offers. In consequence,
the scales become more protective as the current density is increased until an
optimum level is reached when further current density increase leads to loss of
protection.
When cathodic protection is applied in sea water, the current densities noted in
Table 2 are a temporary requirement. As time passes, the calcareous films form
and the current demand diminishes, perhaps to less than 20% of the initial value,
Of course, any disruption of the film by mechanical damage (including storm
damage) or excessive hydrogen generation leads to a temporary increase in current
demand. Fortunately, the scales reform with remarkable speed and excellent
coherence with the residue of the former scale once normal conditions are reestablished,
The importance of calcareous film formation in the context of modelling is
discussed in Section 5 .
2.8.3 Effect of fouling
Although marine fouling can lead to unacceptable weight loadings and additional
drag, it is believed (Section 2.4) that it can reduce the corrosion rate by decreasing
the oxygen flux. But, because the fouling may introduce pitting attack or permit
microbially-assisted corrosion, it is not always welcomed.
The benefits of fouling in the application of cathodic protection are also not clear
cut. Certainly, if the fouling serves only as an oxygen barrier, it is effective in
reducing the current demand, although it may well serve to impede current flow to
areas suffering some corrosion. However, particularly if microbially-assisted
corrosion is encouraged by the fouling, the need for a more negative protection
potential may outweigh any current density advantage gained. There is no
unequivocal evidence that fouling is beneficial to cathodic protection, and some
circumstantial evidence that it is deleterious.
38
MTD Ltd Publication 90/102
2.9
POSSIBLE ADVERSE EFFECTS OF CATHODIC PROTECTION OF STEEL
It was shown in Section 2.2 (Equation 2.3) that the cathodic reaction during
cathodic protection in aerated sea water generates alkali. Excessive polarisation
generates more alkali, also hydrogen gas (Equation 2.4)), and it can be extremely
damaging as well as wasteful.
The formation of bydroxyl ion is not in itself serious, indeed it favours the
formation of calcareous scales and generates a more benign environment for steel.
Nevertheless, it is important that any coating applied to the surface should be able
to tolerate the alkali produced. Oil-based and oleo-resinous paints are softened by
chemical reaction with alkali and may blister. They are therefore unsuitable for use
with cathodic protection. Bituminous paints, epoxy resins, chlorinated rubbers and
particularly coal tar epoxy paints are resistant to alkali, and all are very suitable.
Even here, excessive polarisation can destroy the coating adhesion, and disbonding
results (see Section 4).
The generation of hydrogen is an indication of over polarisation and a consequent
waste of current. Moreover, tbe hydrogen can disrupt otherwise protective
calcareous scales, causing an increase in current demand. It is also widely believed
that hydrogen generation can detach coatings from the surface. Tbe evidence is
slight, and it is more probable that chemical changes which occur at the
coatin&/metalinterface (as a result of cation migration through the coating)
promote delamination. Whatever the mechanism, delamination of tbe coating by
over protection puts the cathodic protection system under greater pressure.
Of more importance is the fact that sea water has an adverse effect on the fatigue
life of structural and pipeline steels used offshore. It is found that cathodic
protection inhibits fatigue crack initiation and the propagation of very short cracks.
To that extent, cathodic protection is beneficial. Unfortunately, catbodic
protection, and more particularly over protection, accelerates the propagation of
longer cracks (of mm dimensions). These effects are discussed in Section 3.
Steels within the strength level range 300-500 MPa having a ferritidpearlitic
microstructure are widely used offshore. Under static loading conditions, and in
the absence of significant hydrogen sulphide contamination, they are resistant to
hydrogen embrittlement at modest levels of cathodic protection. However, higher
strength steels with a quenched and tempered microstructure are susceptible. The
austenitic stainless steels do not suffer hydrogen embrittlement even when subject
to over protection. By contrast, duplex stainless steels with unfavourable
metallurgical structures are vulnerable to cracking even when exposed to modest
cathodic protection. The implications of cathodic protection, and overprotection
for both C - Mn steels and stainless steels are dicussed in Section 3.
Figure 10 shows the relationship between potential and the corrosion, cathodic
protection, and over protection rkgimes. It also shows the relationship between
these potentials as recorded on two different reference electrode scales.
Cathodic protection of offshore structures
39
2.10 THE NEED FOR UNIFORM POTENTIAL DISTRIBUTION
When designing a cathodic protection system, it is important to ensure that the
whole structure is adequately protected, and that nowhere is it substantially over
protected, This is to avoid the economic cost of wasting current in the overprotected areas as much as avoiding the possible adverse effects referred to in
Section 2.9.
Using sacrificial anodes, which are low voltage sources of current, the problem of
achieving uniform distribution of current is not too difficult to solve. As long as it
is possible to distribute the anodes freely in rough correspondence to the exposed
surface area, a uniformly polarised structure is likely to be achieved. Particular
difficulties may arise when the local steel area is high and restrictions are placed on
the siting of the anodes (e.g. at nodes).
Using the impressed current system, the problem is more severe. Impressed
current anodes are relatively high voltage, almost point sources of high current
density. Unless they can be designed and installed to stand well back from the
structure, or dielectric ‘shields’ are installed to deflect the current, it is remarkably
difficult to avoid over protection in one place in the interest of achieving adequate
protection elsewhere.
That a degree of over protection is inevitable is recognised by at least one authority
which recommends(9)a 25 to 50% up-rating of the total installed current
availability when the impressed current technique is to be used rather than
sacrificial anodes. It therefore becomes a matter of very considerable concern to
create an impressed current design which achieves, as far as possible, the optimum
potential distribution given the inherent difficulty of the technique used on
complex structures. It is rendered all the more difficult by the non-ohmic
character of the E - log Zbehaviour of a cathode and, more particularly by its time
dependent response in s e a water as the calcareous scale develops. Section 5
considers this problem in some depth.
40
MTD Ltd Publication 90/102
Section 3 Effects of cathodic protection on
mechanical properties of steels
Cathodic protection of offshore structures
41
3. Effectsof cathodic protection on mechanical
properties of steels
3.1
INTRODUCTION
Two of the basic design requirements for offshore structures, subsea installations
and pipelines are that:
They must possess adequate static strength.
They must possess adequate fatigue endurance.
Thus the static strength of fmed offshore platforms must be sufficient, with an
adequate safety factor, to carry the normal weight of the suucture and to withstand
the loads imposed by 100-year extreme height waves. Similarly, the static strength
of high-pressure gas transmission lines must be sufficient, with an adequate safety
factor, to withstand the hoop stresses imposed on the line by the gas pressure.
However, in many instances (particularly in the case of offshore steel jacket
structures), it is the second design requirement of adequate fatigue endurance
which is of primary importance to the integrity and life of the structure^('^).
Specifically, the fatigue properties of the structures are required to withstand the
relatively small cyclic loads imposed by waves of normal heights impacting on the
shuctures every 10 to 15 seconds for up to a 30-year design life (i.e. up to about
lo9 load cycles).
The group of phenomena known as environmentally-assisted cracking includes,
among others, corrosion fatigue(") and hydrogen embrittlement('@. In principle,
these two phenomena can impair both the static strength and the fatigue endurance
of offshore structures. Furthermore, the application of different levels of cathodic
protection can markedly affect susceptibility to, and the severity of, both corrosion
fatigue and hydrogen embrittlement. It is therefore essential that designers,
installers and operators of cathodic protection phenomena should give careful
consideration to the effects of these phenomena on the mechanical properties of
the structures, components and materials which are being protected.
3.2
ENVIRONMENTAUY-ASSISTEDCRACKING
3.2.1 Corrosion fatigue
Fatigue is a process of time-delayed crack initiation and relatively slow crack
propagation which may occur in metallic structures and components which are
subjected to cyclic loading. The presence of fatigue cracks in offshore structures
can greatly reduce their static strength and load-bearing capability. In principle,
fatigue cracks can initiate in smooth, defect- and flaw-free components, and they
can propagate in completely inert environments. In practice, however, fatigue
cracks tend to initiate at various types of flaw, defect and stress-raiser. For
example, fatigue cracks in offshore steel jacket structures almost invariably initiate
at the toes of the tubular joint welds, where the "hot-spot" and residual stress levels
Cathodic protection of offshore structures
43
are highest, and where there may be welding defects. Furthermore, both initiation
and propagation of fatigue cracks are facilitated and accelerated, relative to the
behaviour in inert environments and air, by the presence of a corrosive
environment such as sea water. The process is then referred to as corrosion fatigue.
In the case of ferritic-pearlitic C-Mnsteels (such as BS43M) structural steels and
API-SLX linepipe steels) exposed to sea water environments, accelerated initiation
of corrosion fatigue cracks is primarily an anodic process, the cracks tending to
initiate sooner, and preferentially, at localised corrosion sites around MnS and
other inclusions (although still in the general vicinity of tubular joint weld toes).
However, in these steels accelerated propagation of corrosion fatigue cracks in sea
water, even under free corrosion conditions, is primarily a cathodic process, the
increased crack growth rates being the result of hydrogen generated at or near the
tips of cracks.
Two basic approaches are used to determine the fatigue and corrosion fatigue
behaviour of metals:
1. The use of smooth (i.e. initially uncracked) specimens to generate curves
relating the number of cycles to failure, N,to the cyclic stress range, S (so-called
S-N curves).
2. The use of pre-cracked specimens in conjunction with fracture mechanics
concepts to generate curves of the cycle-based crack growth rate, da/dN, against the
cyclic stress intensity range, AK
The current practice is to use the S-Napproach for general design and assessment
of offshore structures, while the fracture mechanics approacb is used for analysis, at
the design stage, of critical components in a structure and for appraisal of existing,
known defects. However, there is no doubt that the fracture mechanics approach is
becoming increasingly widely used and important, particularly since its effective use
can result in both reduced costs and increased safety.
3.2.2 Hydrogen embrittlement
Metals exposed to a source of hydrogen and then either simultaneously or
subsequently exposed to essentially static tensile stresses may experience hydrogen
embrittlement, a process while again involves time-delayed crack initiation and
relatively slow crack growth. The presence of hydrogen cracks in offshore
structures, subsea installations and pipelines can reduce their static strength and
load-bearing capability to the extent that failure occurs. Such cracks can also act as
initiation sites for fatigue and corrosion fatigue cracks. There are a number of
sources of hydrogen in metals which can result in embrittlement, but the two which
are of particular importance in the present context are hydrogen generated by the
corrosion reaction at free corrosion potentials and hydrogen evolved at more
negative potentials, as a result of the application of cathodic protection.
44
MTD Ltd Publication 90/102
Three basic approaches are used to determine the hydrogen embrittlement
behaviour of metals:
1. The use of smooth specimens and static loads to generate time-to-failure
and threshold stress data.
2. The use of smooth specimens, combined with monotonic tensile loading, as in
slow strain rate stress corrosion testing.
3. The use of pre-cracked specimens in conjunction with fracture mechanics
concepts to determine threshold stress intensity, &
,, values and to generate
curves of the time-based crack growth rate, dddt, against stress intensity, K.
3.3
EFFECTS OF CATHODIC PROTECTION ON ENVIRONMENTAL CRACKING
In general, the effects of cathodic protection on corrosion fatigue and hydrogen
embrittlement are from one or a combination of the following factors:
1. Cathodic protection systems are desigued to either partially or completely
suppress the anodic, metal dissolution reaction on the protected structure or
component:
M ~ M *++ne
(3.1)
2. Application of cathodic protection tends to promote the hydrogen evolution
reaction:
H+
+ e-.'/a,
It therefore favours entry of hydrogen into the material of construction of the
protected structure (of also into any associated peripherals and secondary
components).
3. Cathodic protection in sea water results in the formation and deposition of
calcareous deposits on the protected structure, When these deposits form in
small cracks they tend to increase crack closure effects, and they may even
block the crack completely.
In general terms, suppression of the anodic dissolution reaction and formation of
calcareous deposits (items 1 and 3 above) tend to have beneficial effects on
corrosion fatigue and hydrogen embrittlement, by slowing down or even eliminating
crack initiation andor crack propagation. O n the other hand, increased hydrogen
evohtion and entry (item 2) tend to have adverse effects on corrosion fatigue and
hydrogen embrittlement, and to promote or accelerate crack initiation andor crack
propagation. The existence of these opposing trends accounts for the complex
nature of the effects of cathodic protection on environmentally-assisted cracking.
Cathodic protection of offshore structures
45
3.4
SUSCEPTIBLE MATERIALS
By far the most widely used metallic materials of construction for offshore
structures and pipelines are low C-Mnsteels. Typical examples are the BS4360
grade 50 series of structural steels and the API-5LX series of linepipe steels. These
steels have a basically ferritic-pearlitic microstructure, and typically have they
strength levels within the range 300 to 500 MPa. It is important to emphasise that,
under free corrosion conditions, steels of this type are not susceptible to stress
corrosion cracking in sea water. Furthermore, under static loading conditions and
in the absence of significant hydrogen suIphide contamination, they are resistant to
hydrogen embrittlement in sea water, both at the free corrosion potential and
under moderate levels of cathodic protection. They only show susceptibility to
hydrogen embrittlement when simultaneously subjected to cathodic protection at
potentials below about -0.9 V (sce) and to continuous dynamic straining (as in slow
strain rate stress corrosion testing), or when exposed to high levels of hydrogen
sulphide (as in sour gas and oil environments).
However, steels of this type are susceptible to corrosion fatigue crack initiation and
propagation in sea water. Furthermore, this susceptibility can be markedly affected
by the application of cathodic protection. Therefore, because these are the most
widely used materials, and because fatigue performance is often of primary
importance to the integrity and life of offshore structures, the bulk of this Section is
concerned with the effect of cathodic protection on corrosion fatigue crack
initiation and propagation in low C-Mn,ferritic-pearlitic steels. However, other
materials of construction which are potentially susceptible to hydrogen
embrittlement are being increasingly used offshore. For example, duplex stainless
steel is used in pipelines, and quenched and tempered steel in tension legs.
Furthermore, most structures have secondary components or peripherals such as
high strength bolts and fasteners attached to them. These may be fabricated from a
wide range of ferrous and non-ferrous materials. This Section therefore also briefly
covers the effect of cathodic protection on the susceptibility of these materials to
hydrogen embrittlement.
3.5
EFFECT OF CATHODIC PROTECTION ON CORROSION FATIGUE OF LOW
c-MnSTEELS
3.5.1 S-N Approach
A schematic
S-Ncurve is shown in Figure 11. The ordinate is the cyclic stress
range, S, in MPa and the abscissa is the number of cycles to failure, N. Even
though plotted on the ordinate, S is the independent variable, and Figure 11
records the number of cycles to failure as a function of the cyclic stress range.
Logarithmic scales are usually and most conveniently used for both axes. Curves of
the type shown in Figure 11 are generated experimentally by using an appropriate
specimen and loading mechanism to subject a representative sample of a particular
steel to constant-amplitude cyclic loading in a representative environment and at an
appropriate frequency.
46
MTD Ltd Publication 901102
Cycles to failure, N
Figure 11 Schematic S-N
curve
In practice, and particularly offshore, structures and components are likely to be
exposed to a complex sequence of load and stress cycles with varying amplitudes
and frequencies, rather than to constant amplitude and frequency loading. The
prediction of fatigue lives using experimental data such as those shown in Figure 11
therefore needs to be based on an appropriate model of fatigue damage.
Specifically, it is assumed that irreversible fatigue damage occurs with each stress
cycle, and that such damage accumulates linearly up to a fixed level, at which point
failure occurs. In the basic S-Nfatigue design approach, each load cycle
experienced by a potential crack site in a structure or component gives rise to a
stress cycle with a stress range, S. The "i" th stress cycle, with stress range, Si,
therefore gives rise to an increment of fatigue damage 1Ni,where Niis the number
of stress cycles to failure under constant amplitude cyclic loading at a stress range
of &. Fatigue failure under variable amplitude cyclic Ioading then occurs when the
linear, cumulative sum of all the individual increments of fatigue damage reaches a
fixed level, i.e. when:
where ni is the number of load cycles which gives rise to stress cycles with a stress
range Si. Equation (3.3) is known as Miner's Rule or Miner's Summation("). In
this equation, the value of A,the fatigue damage summation failure limit, is usualIy
taken as 1, although this value may be reduced (i.e. an increased safety factor may
be introduced) for components that, for example, are critical to structural integrity
or particularly difficult to inspect.
Cathodic protection of offshore structures
47
In summary, therefore, the prediction of fatigue lives using S-Ncurves requires(14):
1. Reliable, long-term statistical information on the expected load history. For
offshore structures, this information would include data on the characteristics
(significant wave height, mean zero crossing period, mean direction, etc.) of
individual sea states, and on the proportion of time for which each sea state
exists.
2. Accurate stress analysis, combined with accurate estimates of local stress
concentration factors, which allows the expected variable-amplitude wave
loading history to be converted to an expected cumulative variable-amplitude
local (or "hot-spot") stress history at various locations on the structure or
component. For each location, this information may be represented by curves
of Njagainst Si.
3. Appropriate and reliable S-Ndesign curves (i.e. curves derived from the
experimental S-Ndata for representative samples of material tested using an
appropriate specimen, component or substructure under appropriate
mechanical and environmental conditions).
Figure 12 illustrates the effects of sea water environments and the application of
cathodic protection on the S-Ndesign curves for low C-Mn structural steels such as
the BS4360 grade 50 series or equivalent specifications with yield strengths not
greater than 400 MPa("). Curve A in this illustration represents the basic,
recommended S-Ndesign curve for tubular steel joints with full penetration welds
exposed to air, or to sea water with adequate corrosion protection. This curve
represents the mean of the experimental fatigue data shifted to the left by two
standard deviations. For N < lo7 cycles it is described by:
log N = 12.16 - 3 log S
(3.4)
For N > lo7 cycles, a change in slope from -3 to -5 is recommended, to allow for
the less damaging effect of low stress cycles. For unprotected joints in sea water, it
is recommended that curve B in Figure 12 is used. This curve represents curve A
shifted to the left (i.e. the fatigue life is reduced) by a factor of 2. Also the change
in slope at N > lo7 cycles is not applied.
In summary, the curves presented in Figure 12 indicate that the corrosion fatigue
life of C-Mn, ferritic-pearlitic steels in sea water is about half the fatigue life in air,
but that the a lication of cathodic protection restores the corrosion fatigue life to
the air value@ The important practical implications of this conclusion for
offshore tubular steel jacket structures are:
48
MTD Ltd Publication 90/102
1000
V
L
c
VI
U
.-
I
Unprotected joints
in sea water
Design l i f e , N ( cycles 1
Figure 12 Basic S-N design curves lor profected and unprotected tubular steel joints
1. Fatigue is of primary importance to the integrity and life of such structures.
2. Fatigue crack initiation and propagation occur preferentially at tubular joint
weld toes.
3. The fatigue life assessment of these areas is based on the assumption that they
are adequately protected from corrosion, and that the corrosion fatigue life is
therefore at least equal to that in air.
4. It is therefore essential that the tubular joint weld toes should not be under
protected, even though these areas may be the most difficult to protect
cathodically. However, over protection of these areas can also have a
detrimental effect on fatigue life.
3.5.2 Crack growth rate approach
A schematic fatigue crack growth rate curve is shown in Figure 13. The ordinate is
the fatigue crack growth rate, da/dN, in dcycle and the abscissa is the cyclic stress
intensity range, AK, in MN/m”. Logarithmic scales are usually and most
conveniently used for both axes, and the resulting sigmoidal curve exhibits three
regions. At intermediate AK values, region B, there is a linear relationship
between log dddN and log AK (i.e. the Paris Law(’’)):
dddN = C
Cathodic protection of offshore structures
49
T hreshoid
a K +h
Log cyclic stress intensity range,
Figure 13
aK MN/m~'21
Schemafic fatigue crack growfh
rate curve
where C and m are constants, is obeyed. At low AK values, region A, the crack
growth rate is lower than predicted by the Paris Law, and it hecomes very low as
the cyclic stress intensity threshold, A&, is approached. At high AK values, the
crack growth rate is greater than predicted by the Paris Law, and it becomes very
high as the fracture toughness of the material, hC,
is approached. Curves of the
type shown in Figure 13 are generated experimentally by using an appropriate
precracked specimen and fatigue machine to subject a representative sample of a
particular steel to cyclic loading at an appropriate frequency in a representative
environment.
The use of fracture mechanics to predict the fatigue lives of structures from crack
growth rate data is complex, and a full description is beyond the scope of this
Section. Briefly, however, the anaIysis is based on the fundamental assumption that
all real structures originally contain flaws and defects or develop them in service.
The analysis then seeks to estimate fatigue life by calculating the time required for
50
MTD Ltd Publication 90/102
the growth of fatigue or corrosion fatigue cracks from these flaws and defects, until
In principle, the number of cycles required for a given increase
failure
in crack length may be calculated by integrating the Paris equation between the
appropriate AK limits. In summary, therefore, the prediction of fatigue lives using
fracture mechanics requires:
Reliable inspection techniques to enable flaws and defects to be located and
sized, both before a structure or component is put into service and throughout
its normal service life.
Accurate stress intensity factor solutions (i.e. relationships between stress, crack
length and stress intensity) for all the relevant joints to enable the expected load
history, conventional stress analyses, and crack location and size data to be
combined to give the expected cyclic stress intensity range history for all cracks
present in service.
Appropriate and reliable fracture toughness data and curves of fatigue or
corrosion fatigue crack growth rate against cyclic stress intensity range.
The effects of seawater environments and the application of cathodic protection on
the fatigue crack growth rate of BS4360 grade 50D steel are shown in Figures 14
and 15, respectively("). The Paris constants for the curves shown in Figure 14 are:
air
C = 1.0 x lo-", m = 3.0
sea water C = 6.0 x lo-", m = 3.0
In summary, the cunres presented in Figure 14 suggest that the upper bound
corrosion fatigue crack growth rate of C-Mnferritidpearlitic steels in sea water is
up to six times the mean fatigue crack growth rate in air. Figure 15 indicates that,
even when cathodic protection within the range -0.7 to -0.9 V (AdAgCl) is
applied, the crack growth rate in sea water is still about three times that in air.
Over protection to -1.1 V (AdAgC1) results in an increase in the crack growth rate
to about six times the air value. The important practical implication of these
conclusions is again that the application of normal levels of cathodic protection has
a heneficial effect on corrosion fatigue hehaviour, but that over protection is
detrimental.
However, it is important to note the discrepancy which exists between the S-N
approach on which the design of tubular joints is currently based, Figure 12,and
the crack growth rate data, Figure 15. Specifically, the former indicates that the
application of normal levels of cathodic protection restores the unprotected
corrosion fatigue life in sea water to the life in air. On the other hand, at normal
levels of cathodic protection, the corrosion fatigue crack growth rate is still about
three times greater than the fatigue crack growth rate in air. The reason for this
discrepancy lies in the fact that the fatigue lives of the specimens used to generate
the S-Ndata on which Figure 12 is based are dominated by crack initiation and by
the growth of short cracks. These processes are delayed by the application of
Cathodic protection of offshore structures
51
Figure 14
Effect of sea waler on the faligue
growth rate of BS 4360 grade 50D
steel
Figure 15
52
1
i0
Cyclic s t r e s s intensity range,
00
~ l K ~ M N l r 1n ~ ’ ~
€tfect of potential on the corrosion latigue crack growth rate of BS 4360 grade
50D steel in sea water
MTD Ltd Publication 90/102
cathodic protection, in particular by the suppression of the anodic, dissolution
reaction and by the deposition of calcareous deposits. However, the data in Figure
15 relate to the propagation of long, through-thickness cracks, at a rate which
depends on the amount of hydrogen evolved at the crack tips. Although this is a
minimum over the potential range -0.7 to -0.9 V (sce) the amounts of hydrogen
generated are still significant and much greater than the amounts of hydrogen
which are present at the tips of fatigue cracks growing in air.
3.5.3 Review of recent experimental data
The data base on which the basic fatigue design curve for tubular joints in air is
based is continually increasing as new experimental data become available. Thus a
recent analysist2') of the experimental data suggests that, for 32-mm joints,Equation
(3.4) should be modified to:
This change in the value of the constant from 12.16 to 12.33 re resents a 46%
increase in the design life in air. However, Sharp and ThorpPL)also suggest that
the fatigue life of unprotected tubular joints in sea water may in fact be less than
half the value in air and, in sea water temperatures of 2OoC, may be as low as o n e
third of the value in air.
Because of the cost involved in testing large welded tubular joints, the S-Ndesign
curves shown in Figure 12 were originally based to a considerable extent on fatigue
tests carried out on relatively small, planar welded specimens. For example, the
typical experimental data@) shown in Figure 16 relate to cruciform welded
specimens fabricated from 38-mm thickness BS4360 grade SOD steel plate tested at
a frequency of 0.167 Hz and an R ratio (the ratio of the maximum and minimum
stresses) of -1,in air and in synthetic sea water at 5°C at the free corrosion
potential and at -0.85 V (AgAgC1). It was on the basis of a large volume of similar
data that it was originally concluded that the fatigue life in sea water is restored to
the air value by the application of normal ievels of cathodic protection. However,
this conclusion needs to be qualified and regarded with some caution, for the
following reasons:
The great majority of the tests on which this conclusion is based were carried
out in synthetic sea water and under potential control. In general, relatively
little effort was made to determine whether or not a calcareous deposit formed
during the tests, and, if so, whether it was similar in terms of composition,
structure, etc. to the calcareous deposits which form on cathodically protected
steel structures in the North Sea. Similarly, little effort was made to determine
whether the cathodic current densities used during the corrosion fatigue tests
were ofa similar magnitude to those applied in practice. Of particular
importance is the fact that, in service, the current density falls markedly with
time as the calcareous deposit forms. The absence of a calcareous deposit or
the application of unrealisticaIly high current densities during the corrosion
fatigue tests could have a significant effect on the results obtained.
Cathodic protection of offshore structures
53
An increasing amount of data from tests on large welded tubular joints are now
available. In general, these show that there is good agreement between the air
fatigue lives of planar and tubular specimens. However, it is a matter of
concern that the data appear to indicate that, contrary to the design curves
shown in Figure 12, the corrosion fatigue life of large welded tubular joints in
sea water is not fully restored to the air value by the application of normal levels
of cathodic protection (z1.2394Dz). Typical data are shown in Figure 17. In this
illustration, the solid line is the Department of Energy design curve for air and
protected joints (18) from Figure 11, and the broken line is the mean of their
data from which the design curve derives. Data points relate to actual tests on a
fuli size welded tubular joint (lS). The reason for the different effects of
cathodic protection on the corrosion fatigue life of welded tubular joints and
planar welded specimens is apparent from Figure 18(%). This illustration shows
curves of the crack length as a function of specimen life for the two types of
specimen. Clearly, the life of planar specimens is dominated by crack initiation
and growth of short cracks, while the life of tubular specimens is dominated by
the growth of much longer cracks. For the reaons discussed above, the former
processes are much more effectively inhibited by the application of normal
levels of cathodic protection than the latter.
There are obvious and potentially serious practical implications of data such as
those in Figure 17 for the fatigue life of welded tubular joints which are already in
sewice. Specifically, such data tend to suggest that the service life of existing joints
may be somewhat shorter than the original design life, or at least that smaller safety
factors are incorporated in their design than originally intended.
400
300,
250,
0
@\ O *
&A
200,
.:\\
O\
150,
0
@\
I
m
a
w-loo,
r
al
C
rn
0
Air results
Sea water I f r e e corrosion )
o Sea water ( cathodic protection I
I
m
L
vl
0
Mean line
for air
data
QI
m
L
c
Ln
.5 50
U
d
U
8
10‘
Figure 16
54
I
1
1
1
8
1
I
8
I05
I
1
I
1
)
I
8
:o6
I
.
1
1
.
1
.
1
3’
S-N data for planar welded joints of BS 4360 grade 50D steel in air and in sea
water, without and wifh cathodic protection
MTD Ltd Publication 901102
Figure 17
Comparison of experimental S-N dafa
for tubuhr welded joints wifh f h e
design curve for protected joints
1o6
15
50
Percentage of l i f e
Figure 18 Curves of crack depth against percentage of fatigue lite tor planar and tubular
welded specimens
Cathodic protection of offshore structures
55
3.6
EFFECTS OF CATHODIC PROTECTION ON HYDROGEN EMBRITI'LEMENT
OF OFFSHORE MATERIALS
It is important to emphasise once more the fact that low C-Mn,ferritic-pearlitic
steels such as the Bs4360 grade 50 series of structural steels and the API-5LX series
of linepipe steels are not susceptible to stress corrosion cracking in seawater
environments or marine atmospheres. Neither are they susceptible to hydrogen
embrittlement in sea water unless either they are simultaneously subjected to
continuous dynamic straining (as in slow strain rate testing) and potentials more
negative than about -0.9 V (sce), or unless the environment contains significant
levels of hydrogen sulphide (e.g. sour gas and oil products, discussion of which is
outside the scope of this Section). In practice, therefore, the question of the effect
of cathodic protection on hydrogen embrittlement of offshore materials is mainly
relevant to high strength materials (i.e. strength levels above 700 MPa), such as
quenched and tempered low alloy steels, heavily cold-worked, high C-Mnsteels
and certain non-ferrous alloys,
A number of UK and foreign design rules, recommended practices, etc. (see
Section 11) quote specific potentials below which steels of particular strength levels
may experience hydrogen embrittlement. Typically, it is suggested that potentials
more negative than -0.9 V (Ag/AgCl) may be harmful to steels with strength levels
above 700 MPa, and that steels with strength levels above 800 MPa should not be
subjected to potentials below -0.8 V (Ag/AgCl). However, it has to be emphasised
that although these guidelines are of considerable use, they are empirical, and they
are hased only on somewhat limited service experience. As such they should be
treated with appropriate caution. This caution is required because hydrogen
embrittlement is a particularly complex phenomenon. The susceptibility of a
specific component to cracking depends, inter alia, on the strength level of the
particular material of construction, tbe total applied and residual stress level, and
the level of cathodic protection. It is therefore not possible to specify a simple
rule-of-thumb regarding "safe" and "unsafe" levels of cathodic protection which
applies to all materials in all environments under all service conditions. The only
broad generalisation which can be made is that the risk of cracking increases as the
strength level of the particular material of construction increases, as the total tensile
stress level increases, and as the service potential of the component is made more
negative(16). It is therefore essential that designers and operators of offshore
cathodic protection systems bear in mind the fact that these installations poIarise
not only the primary structure which is being protected, but also all the secondary
structures, peripherals and components which are electrically and electrolytically
connected to the primary structure. When these secondary items contain, or
consist of, highly stressed, high-strength materials, there is a significant risk of
embrittlement, cracking and failure which must be taken into account and
alleviated if necessary.
Finally, mention has to be made ofthe possibility of hydrogen embrittlement and
cracking of duplex stainless steel pipelines and components as a result of cathodic
protection. While the strength level of duplex stainless steels is not as high as that
56
MTD Ltd Publication 90/102
of the other materials discussed in this section, it is higher than that of conventional
linepipe steels such as the MI-SLX series. The susceptibility of cathodically
protected duplex stainless steels to environmental cracking is still an active research
topic. However, it currently appears that hydrogen embrittlement as a result of
normal levels of cathodic protection is only likely to be a practical problem If the
austenite content of the steels falls to unacceptably low levels. This is most likely
to occur at welds, as a result of poor welding practice. Provided there is a good
phase balance (i.e. a minimum of about 40% austenite in the microstructure),
duplex stainless steel parent plate is not susceptible to cracking in normal seawater
service at normal cathodic protection potentials and current densities or at realistic
levels of over protection. Welded material is equally resistant to cracking, provided
the content and distribution of austenite in the heat affected zone and weld metal is
satisfactory.
Cathodic protection of offshore structures
57
Section 4 Organic coating and cathodic protection
Cathodic protection of offshore structures
59
4. Organic coatings and cathodic protection
4.1
INTRODUCTION
4.1.1 The purpose of organic coatings
The design of a typical marine cathodic protection system begins with a knowledge
of the current density demanded by the particular structure to be protected. This
value varies, depending on the location of the structure. Typically, in the Northern
North Sea a mean value of 90 m N m Zis quoted(9). In terms of cathodic protection,
a major requirement for an organic coating is to reduce the area of exposed
steelwork, and thus to reduce the overall electrical demand on the cathodic
protection system. It is therefore essential that any organic coating chosen should
be compatible with a submerged marine situation in general and with cathodic
protection in particular. In dealing with cathodic protection and coatings, the
environment is normally fully immersed. It is convenient to survey the various
coating situations which are found under fully immersed conditions in the absence
of cathodic protection and to assess the likely change in the situation when
cathodic protection is applied. However, in general terms, the effect of cathodic
protection on immersed coated steel is to alter the rates of various processes which
are likely, but not to alter the overall mechanism or,indeed, to make matters
worse.
4.1.2 Various coating situations on steel substrates
Figure 19 is the ideal case where the coating is intact and well adherent to the steel
surface. This occurs when an appropriate coating is applied to a well blasted steel
surface. Such a coating is permeable to both oxygen and water, and its success as
an anti-corrosive measure relies on its high ionic resistance to slow down the
possible corrosion process to an extremely small value, and on an adequate wet
adhesion force to maintain the coating in intimate contact with the metal. Such a
coating is expected to protect well under an immersed condition, and the presence
or absence of cathodic protection does not substantially affect the performance in
this situation. The coating merely has to be formulated to be compatible with a
marine condition. A list of recommended systems is given later.
Figure 20 is a similar situation to Figure 19, except that there has been damage to
the coating, leading to metal being exposed to sea water at the point of damage. In
the absence of cathodic protection, this situation leads to corrosion of the steel at
tbe point of damage and disbonding of the coating from around the hole. The
mechanism of disbonding is thought to be the result of cathodically-generated
alkali penetrating under the coating. The disbonded region is usually very thin,
and with a transparent coating it can be seen as a slight change in the optical
reflection in this area. The effect of cathodic protection in this situation is to
polarise the exposed steel in a negative direction, which increases the rate of the
cathodic reaction on the steel and reduces the rate of the anodic corrosion
reaction. The overall effect is to generate more alkali in the vicinity of the hole
and to increase the tendency of the coating to disbond away from the hole. This
situation forms the basis of all the standard cathodic disbonding tests and much of
the data available. This aspect will be referred to later.
Cathodic protection of offshore structures
61
Sea
Coating
Coating
Subst rate
Substrate
I:. .
;:-
. ':,:,
.. . .. .. . . . , ... .
. . -
Figure 19
Intact and well adherent coating
'
.
. -
,
;'
.
:-: . . . . .
:_: ,I
. . . . . ..., . .'- .
, -'
.
'.. .
,.
.. - -
.- , . , ' ; .
Figure 20
Well adherent coating with damage
extending to the substrate
Figure 21 shows a situation similar to that of Figure 20, except that an area of
unbonded or poorly bonded coating is present at the interface, close to the hole.
This could result from a variety of causes, including organic contamination on the
interface, inappropriate viscosity during coating application, and presence of heat
transfer at the interface. In the absence of cathodic protection, the electrochemical
corrosion at the hole could generate both cathodic disbonding as already described
and a cathodic area under the coating at this poorly adherent area. Generation of
alkali under the coating enlarges this blister, and strong alkali solutions could be
present therein.
Figure 22 summarises the situation where the coating has been applied to
inadequately prepared steel where some rust and residual salt have been left
behind on the surface of the steel. As stated previously, for most coating systems,
the rates of oxygen and water permeation through them is relatively rapid and
therefore do not determine the corrosion rate. The residuai salts left behind under
the coating as a result of inadequate cleaning before coating application are
dissolved in the water which permeates the coating to form a concentrated salt
solution at the interface. There is now the situation of the sea water on one side of
the coating and a concentrated solution on the other, This sets up an osmotic force
which causes the water to migrate from the dilute to the concentrated solution.
Thus water is driven into the interfacial region. The same situation could occur
with the use of a water-sensitive anti-corrosion pigment in the primer coat (e.g. zinc
phosphate or zinc chromate), and the use of such pigments as primers in multicoat situations is not recommended. The ideal priming coat is considered to be a
thin ''wash coat" of the complete system, only diluted down with solvent. Some
barrier coatings which are formulated to be applied at very high levels of thickness
claim to be genuine barriers and to rely on their ability to reduce the permeation of
oxygen and water to provide an anti-corrosion function. Such coatings are less
susceptible to the effects described above.
62
MTD Ltd Publication 901102
Coating
Coating
Substrate
Substrate
Figure 21
Damaged coating together with
region of poor adhesion
Figure 22
Coating with region of poor adhesion
resulting from application on to rusty
surface
The presence of water, oxygen, electrolyte and iron at the paintlmetal interface sets
up a corrosion cell under the coating, with blistering of the coating away from the
metal. Within the same blister, anodic and cathodic areas co-exist. The cathodic
areas are predominantly at the edge of the blister and contribute to further paint
detachment.
EIectro-endosmotic water absorption is a different phenomenon which is frequently
referred to. The voltage gradient generated by either a corrosion cell or the
cathodic protection can cause further water absorption into the coating by a
process known as electro-endosmosis. A detailed explanation of this process is
beyond the remit of this Section and the interested reader is referred to the
literature(28). However, cathodic protection designers are aware of the tendency of
the environment around an impressed anode ground-bed to dry out, bewuse of the
electrical field gradient around the anode. In the same manner, the environment
around the cathode tends to absorb more water, and if the cathode carries an
organic coating, water will be driven into this coating.
4.2
INTERACTION OF CATHODIC PROTECTION AND COATINGS
4.2.1 Cathodic protection and underfilm corrosion cells
Where the situation under the film shown in Figure 22 is isolated electrically from
the solution by the high resistance of the film and the film is structurally intact, the
potential developed by the cathodic protection system is not effective under the
film and the cathodic protection does not affect the corrosion rate of the
The only remedies are to avoid this situation in the first pIace by ensuring the
absence of residual soluble material before coating, to make the coatings so thick
that oxygen and water permeation become very slow and therefore rate
determining, or to make the coatings mechanically weak in tension so that at the
beginning of the blistering process, the coatings fracture. This exposes the steel to
the solution and thus to the potential of the cathodic protection system, and
corrosion ceases.
Cathodic protection of offshore structures
63
4.2.2 Paint and coating systems and their compatibility with cathodic protection
The most common paint systems (e.g. oil modified systems and alkyds) are
composed of organic binders which contain ester bonds as part of their main
chemical structure. Ester bonds are susceptible to hydrolysis and bond breakage,
particularly in an alkaline environment. Because alkali is the main cathodic
reaction product, such paint systems are not recommended for use under
submerged conditions either with or without cathodic protection. Coatings which
are suitable for use in submerged conditions should be chosen to minimise the
number of ester links in their main polymer structure, Thus, coatings which are
recommended for use in submerged conditions with cathodic protection include:
pure epoxy, coal tar epoxy, pitch epoxy, vinyls, vinyl tar, chlorinated rubber, coal
tar, bitumen and polyurethanes formulated with predominantly ether linkages.
4.3
FAILURE OF COATINGS
4.3.1 Cathodic disbonding
The situation shown in Figure 20, in combination with cathodic protection, leads to
a generation of alkali at the SteeYsolution interface. This may lead to loosening of
the bonds between the coating and the steel adjacent to the defect. This process is
known as cathodic disbonding. This deleterious interaction of cathodic protection
with organic coatings has led to extensive research, and Section 4.3.2is a summary
of the current proposed mechanisms. For further reading, References 30 and 31
are considered relevant.
4.3.2 The mechanism of cathodic disbonding
Figure 23 is a diagram of a coating which has a defect through to the solution and
which also has an area of disbonding at the paidmetal interface. Under normal
cathodic protection potentials, the main cathodic reaction is the reduction of
dissolved oxygen to produce the hydroxyl ion. Charge balance with the sodium ion
produces a solution of sodium hydroxide at the steelholution interface. The
coating adjacent to the gap is susceptible to disbonding, and detachment may occur.
Alkali movement up the steelhating interface is thought to be responsible for the
actual disbonding reaction. The precise atomic features of the disbonding
processes are still under some discussion.
Some workers favour alkali reaction with the air-formed oxide film at the
steeycoating interface. This produces break-up of the oxide film and consequent
disruption ofthe interface. Other theories suggest a reaction between the alkali
and the layers of the coating immediately adjacent to the coating/metal interface.
The rate of progress of cathodic disbonding depends upon availability of metal
cations to neutralise the hydroxyl ions. These ions can either diffuse through the
coating or up the gap at the coatindmetal interface. The relative importance of
each process depends on a variety of factors, including coating thickness, coating
permeability, and the distance from the defect to the disbonded front.
64
MTD Ltd Publication 90/102
Sea
Coating
Substrate
Figure 23
As Figure 20, following cathodic
disbonding
Figure 24
As Figure 21, following cathodic
disbonding
Sometimes the cathodic disbonding is also manifest as a blister underneath the
coating (see Figure 24). In this instance, the rate of growth of the blister is
governed by cation transport across the film. Blister growth can be reduced if the
ion in question is slow moving. For example, disbonding in solutions containing
sodium ions is much faster than in solutions containing calcium ions. Where blister
solutions are anatysed, strongly alkaline solutions are found for coated steel in sea
water.
4.3.3 Standard tests for cathodic dhbonding
The requirement for a standard test to assess cathodic disbonding arose in the use
of externally coated pipelines for use in underground service where the pipeline
receives cathodic protection. The purpose of such tests is to simulate the
consequential effect of cathodic protection on the coating adhesion adjacent to the
damaged area of coating by assessing loosening of the coating.
Some cathodically-protected gas lines have failed by stress corrosion cracking
associated with coating disbondment and the presence of dilute
carbonatehicarbonate solutions under the disbonding coating(32).This has
provoked further interest in cathodic disbonding.
The test procedures are straightforward, in that a coated panel is damaged to
produce a defect penetrating through to the steel, then immersed in an electrolyte.
Cathodic protection is applied, and the extent of coating disbondment is assessed at
the end of the test.
The ASTM 0 8 1985 standard(33),is the latest revision of a test method first
published in 1968. The first critical variable which is employed is the potential of
the specimen. Originally, a magnesium sacrificial anode was used at a potential of
Cathodic protection of offshore structures
65
-1.45 to -1.55 V (Cu/CuSO,). Later, an impressed system was also specified, but the
specimen is polarised to the same potential range. The second critical variable is
the nature of the electrolyte. A tap water solution containing 1% by wt each of
sodium chloride, sodium sulphate and sodium carbonate is used.
The standard admits that this test may not be representative of practice and that,
“ability to resist disbondment is a desired quautity on a comparative basis, but
disbondment per se in this test is not necessarily an adverse indication. The virtue
of this test is that all dielectric type coatings now in common use will disbond to
some degree thus providing a means of comparing one coating with another. Bond
strength is more important for proper functioning of some coatings than others and
the same measured disbondment for two different coating systems may not
represent equivalent loss of corrosion protection”.
In other words, just because a coating disbonds in this test does not mean that the
particular coating is no good, and simply because two coatings perform in a similar
manner in the test does not mean that they are equally capable or incapable of
providing corrosion protection.
The British Gas test, PSKW6:PART 1(34),uses a solution of 3% sodium chloride
with the specimen polarised to a potential of -1.5 V (SCE) using an impressed
system only, Two temperatures are also recommended: 2OoC and 50°C. This test
forms the basis of British Standard BS3900 : Part F11~985(~’),
specifically for land
based coated structures. A major difference between this Standard and the British
Gas test is that the Standard is carried out isothermally at 23 i 2°C and not
additionally at 50°C.
The accompanying British Standard BS3900 : Part F10:1985(36),is based on COIPM
(Comitd International Permanent pour Protection des Matdriaux en Milieu Marin)
test and is intended for use in marine environments, However, this standard states
that,
“For coatings for application to subsea pipelines it may be necessary to consider
this Part or Part F11 or and alternative procedure;”
This standard uses either natural sea water or synthetic sea water to BS
1391:1952(”). It also specifies a specimen potential of -1000 mV (SCE). This
potential is considerably less severe than those previously specified.
The following sections discuss the effects of various parameters on the rates of
cathodic disbonding. It should be realised that the information presented is
derived from laboratory-based experiments, using methods based on the standard
tests described above. Different coating systems were used at different thicknesses,
from different manufacturers. Therefore the conclusions are of a qualitative rather
than a quantitative nature.
66
MTD Ltd Publication 90/102
4.3.4 Time effects
Laboratory tests of relatively short duration (less than 6 months) conclude that the
disbondin rate, measured as the rate of change of the disbonded area, is
constant
Reference 39 suggests that after an extended immersion period of 1
year, the dishonding rate is reduced to a constant value. However the potential
employed was -1.0 V (CdCuSO,).
4.3.5 Potential effects
Evidence in the literature (30*38*40)
suggests that the more negative the potential, the
more rapid the rate of disbondment. This work is within the potential range -600
to -1500mV (SCE). Even without the application of cathodic protection, there is
still a h i t e amount of disbondment around a coating defect. There is no evidence
to suggest that the rate of disbonding changes at a particular potential. The
linearity over the whole range implies that the same mechanism of disbonding
occurs. At the relatively noble potential, -1000 mV (SCE), the only mechanism
can be alkaii generation derived from oxygen reduction. This mechanism is
thought to apply across the whole range, and the other suggested mechanism of
cathodic disbonding (hydrogen generation at lower potentials) is no longer thought
likely. This conclusion is also supported by a recent CEOCOR publication
(Western European Committee on Corrosion and Protection of Conduits)(4I).
4.3.6 Effects of temperature
Two temperature effects are to be considered. The first is the situation where there
is a thermal gradient across the coating from the steel substrate to the environment.
The second is where the system is at an elevated, but constant, temperature. The
former is a more common phenomenon, the latter is more easily studied in the
laboratory. A thermal gradient across a coating, with the steel at a lower
temperature than the wet environment, is well known to increase the tendency for
the coating to form water-filled blisters. This tendency is thought to be the result
of water permeation through the film. It is not possible to give specific advice on
this area, because little fundamental work has been carried out, except to say that
the tendency depends on many variables, inter alia : the coating and its specific
formulation, the polymer used in the coating, the presence and chemical nature of
plasticisers and retained solvent, the coating thickness and the temperature
gradient. The interaction between a thermal gradient and cathodic protection has
been reported(42),but coatings were not considered as part of this study. However,
it is highly likely that there is a substantial interaction between heat transfer,
cathodic protection and coatings.
Isothermally, the effects are more clear. In general, the rates of cathodic
disbonding increase with increasing temperature. The only definitive study(30)
calculated the activation energies involved for a particular system as 9500 cdmol.
This value was ascribed to the disbonding process being diffusion controlled. A
study reported in a recent publication(41),describes a two-stage test procedure. The
first stage involved an intact coated specimen immersed at 7OoC and -1.5 V (SCE),
Cathodic protection of offshore structures
67
and the second stage involved the making of an artificial holiday in the coating and
continuing the test. The results demonstrated that such a test was extremely
damaging to the specimens, with extensive blistering and disbonding. The main
controlling factor was the strength of the electrolyte used.
A German study referred to by Trotman(40),looked at temperatures between 5°C
and 35°C. An increase was observed in the breakdown by blistering by factors of
between 2 and 3, as the temperature increased from 15°C to 35OC. Recent work by
H i g g i n ~ ( ~using
~ * ~ )epoxy
,
powder and a potential of -1.5 V (SCE) showed a
substantial increase in cathodic disbondment over the range 20°C to 85OC. For
example, for a non-pretreated, but grit blasted, steel there was a five-fold increase
in disbonding over this range.
4.3.7 Effect of film thickness
There is a discrepancy in the published literature on the effect of coating thickness
on the rates of cathodic disbonding. Some workers demonstrate that over a
complete range of thicknesses, the rates of disbonding are reduced with increasing
thickness, whereas other workers have shown that this applies only up to a certain
critical thickness after which the disbonding rate becomes inde endent of
thickness. Subscribing to the first view is evidence by TrotrnanY4), looking at film
thicknesses between 100 and 400 gm. The coating systems he reported were vinyl
tar, tar epoxy, chlorinated rubber, and aluminium bitumen. Further confirmation
of this view arises from a recent paper by Coulson and Temple(45). They used an
electrolyte of sodium chloride, a potential of 1.5 V (SCE), and fusion-bonded epoxy
with thicknesses ranging between 0.1 and 0.7 mm. The alternative view has been
put forward by Jin et CZZ.(~). Chlorinated rubber at a potential of -1.05 V (SCE)
was employed, and this work suggests that between 10 and 50 m, there is a strong
thickness dependence. Above 50 urn, there is no thickness dependence.
,
an alkyd and a potential of 0.8 V (SCE) in a 0.5 mol
Leidheiser et C L ( ~ )using
sodium chloride NaCl solution also showed that, at thicknesses greater than 50 ccm,
the disbonding rate is independent of thickness.
4.3.8 Metal surface profile
Because the disbonding process is a surface phenomenon, any increase in the
apparent area of contact by increasing the roughness of the substrate should reduce
the rate of disbonding. Two studies have demonstrated this effect. C o u l ~ o n ( ~ ~ )
showed that a change in profile from 0.053 to 0.09 mm produced a linear reduction
of 50% in the rate of disbonding. Other work(38)showed a similar behaviour.
4.3.9 Nature of the coating
Slight changes in coating formulation can cause large effects on the cathodic
disbondment performance. The study by The British Ship Research Association(40)
stated that,
"primers of the same generic type produced by different manufacturers were
overcoated with the same anti-corrosive paint and tested under cathodic
protection."
68
MTD Ltd Publication 90/102
It concluded,
"the spread of results obtained within a generic type is too great to be able to allow
any general conclusions to be drawn on t h e behaviour of a specific material."
The CEOCOR report(4') concluded that,
"If different types of coating are compared, a lower rate of disbondment for one
type of coating in a CD test does not necessarily mean a better performance in
practice ."
This seems to suggest that a performance criterion for a coating assessment based
on the cathodic disbonding test is somewhat dubious. Set against this is work by
D i ~ k i e ( ~who
~ ) , showed a substantial difference in anti-corrosion performance
between two similar coating systems which differed slightly in their chemical
structure so as to make the superior coating more resistant to cathodicallyproduced alkali.
Recent information from an operator gave a general assessment of the tendency of
certain generic coating types to cathodicaIly disbond. This was based on the British
Gas Test(34).It was stated that, in general, fusion-bonded epoxies are better than tar
epoxies, which are in turn better than tar enamels. However, it was agreed that
minor modifications to coating formulations could have a substantial influence on
the performance of the coatings in a cathodic disbonding test. Such a test should
not be the onIy performance indicator, but it should be one of many tests which
need to be carried out to help assess the quality of the coating.
4.4
INFLUENCE OF RUST AND CONTAMINATION AT THE INTERFACE
Brief references in the literature suggest that where the coating is applied to a steel
surface with inferior surface preparation, the expected performance may not be
adequate. Morgan(47),describes the case where water has accumulated at the
painvmetai interface. He states,
Y n such cases corrosion can occur and cathodic protection from the outside will be
of n o effect in preventing it",
Tr~tman(~'),
refers to work where salt contamination at the interface,
"can reduce the useful life of a coating system with cathodic protection by at least
one half '.
S ~ h w e n k ( ~also
l ) , concluded that,
"cathodic protection is not effective in closed blisters".
Cathodic protection of offshore structures
69
In work using an artificial blister(29),with micro-electrodes inserted within it, both
the potential in the blister and the corrosion rate in the blister were measured as a
function of applied cathodic protection. It was confirmed that the presence of
cathodic protection from without had a negligible effect on the corrosion under the
coating.
To prevent such problems from occurring in practice three approaches can be
adopted:
An adequate standard of cleanliness can be specified during the coating stage
of the steel. Typically the Swedish Standard(46)is available to assist the
specifying engineer or designer.
Where there is a likelihood of contamination at the coatin&lmetalinterface, the
coating can he so formulated by the manufacturer that any corrosion product at
the interface causes coating fracture, and the coating no longer screens the metal
from the applied potential of the cathodic protection.
The coatings could be so thick that they act as an oxygen and water barrier, and
this would stifle the underfilm corrosion process. Each coating system would
have a minimum thickness level where this effect would occur. This value is not
normally known with any degree of accuracy for most systems, but a value of
5 0 0 Irm is normally quoted.
4.5
SHIELDING OF ANODES
In the immediate vicinity of the anode in the cathodic protection system,
particularly in the case of impressed anodes, it is important to provide a coating for
the steel which can resist the negative potentials close to the anode. The
requirements for such a system have been determined empirically, but they follow
the principles discussed here.
First, the steel should be at a high standard of cleanliness with an adequate profile
produced by abrasive blasting, typically SIS 05-59-00 1967 S a h . A coating has to
be used which can be applied at coating thicknesses of 1 to 2 mm, typically a
trowelling coal tar epoxy system. The coating should be applied evenly without any
defects. Where a pair of bracelet anodes are installed around a pipe, the gap
formed where the two anodes meet should be filled with a layer of hot applied coal
tar or bitumen.
4.6
SPECIAL COATINGS
4.6.1 Field joint coatings
British Gas experience on this and other aspects of coating for steel pipes and
fittings has recently been summarised in a comprehensive review paper by
Norman(49). He considers three factory-applied systems for pipeline coatings:
epoxy powder, extruded polyethylene, and hot applied glass fibre-reinforced coal
tar enamels.
70
MTD Ltd Publication 90/102
For field jointing of pipes coated with powder epoxy, three possibilities are
provided. Ideally, the joint is coated with epoxy powder. This requires a
sophisticated procedure where the weld area is abrasively blasted, the steel is
preheated by induction, and the powder is sprayed immediately onto the blasted
area. A British Gas Standard(49)covers this process. The second alternative
involves a hot airless spray urethane or epoxy system. Hand-applied versions are
also possible. The third alternative is the use of cold-applied laminate tapes,
together with fillers, putties and mastics. Abrasive blasting is still a requirement
prior to coating.
For polyethylene coated pipes, fieId joints are coated with cold-applied laminated
tapes, heat-shrinkable materials or grease-based tapes, again after abrasive blasting
of the welded area.
For pipes coated With coal tar enamel, liquid epoxy or urethane, the recommended
procedure, following blasting, involves spray or brush application of an epoxy or
urethane system or cold-applied laminate tape.
4.6.2 Pipeline wraps
A wrap is a plastic tape consisting of either polyethylene or polyvinyl chloride
which is coated with an adhesive, then applied to the pipeline. The wrap may be
applied cold, in which case the adhesive is grease based, or it may be applied hot
and the adhesive is pitch or bitumen. According to Norman(49),wraps are used for
the repair of polyethylene coated gas lines, and for repair and joint coating for coal
tar enamel coated lines. Cold-applied tapes are more common than hot-applied
tapes.
4.7
RECENT EXPERLENCE WITH COATINGS
As is highlighted in Section 6, when carrying out a cathodic protection design study
on a coated steel structure, a critical variable is the area of uncoated steel.
Furthermore, it is also necessary to assess whether the coating is breaking down
and thus how the area of exposed steel changes during the design life of the
structure. Such information is enshrined in Recommendations and Codes of
Practice, a typical example being that produced by Det norske Veritas(’). Values
are expressed as an initial, mean and final percentage of coating breakdown.
Recent information has been obtained from an operating company on coating
performance of two offshore platforms coated with a two-coat tar-epoxy system.
These performance data were produced by inspection of the underwater section of
the structures, and they are based on anode wastage and general observation of
coating condition. The structures were at early and mid-life, and they were
designed for a 25-year life. It was stated that the initial coating breakdown was
estimated at 2% damage to cover construction damage. The rate ofincrease o l
exposed steelwork with time, estimated after diver inspection, was determined to be
1%per annum. Thus for a 25-year life, the area exposed at the end is 27%. This
Cathodic protection of offshore structures
71
value is somewhat less than that quoted by Det norske Veritas, where an exposed
area at the end of life is 30% for a 20-year life and 60% for a 30-year life. Thus for
structures which are designed to the Det norske Veritas rules(’) and coated as well
or better than the system quoted above, the cathodic protection system is unlikely
to be under designed.
Information from the same source was also sought on the design approach for
coated marine pipelines. In this situation, the design values which are normally
used are 3% damage initially, to cover a worst case situation of poorly applied field
joints, and thereafter n o change in coating breakdown with time. This value
applies to a thick tar enamel coating plus a concrete weight coating. For fusionbonded epoxy coating, the value for coating breakdown at the end of a 25-year life
is 10%. If correct, the implication of these values is that many pipelines designed
to Det norske Veritas recommendation^(^) are grossly over designed in terms of the
amount of anode materiel supplied.
72
MTD Ltd Publication 90/102
Section 5 Calculation and modelling for the design of
cathodic protection systems
Cathodic protection of offshore structures
73
5. Calculation and modelling for the design of cathodic
protection systems
5.1
INTRODUCTION
It was shown in Section 2 that the rate of corrosion of steel in sea water can be
controlled by adjustment of the potential difference between the stee1 and the
conducting electrolyte (sea water). It is typically desired that the steel should be
held, with respect to the electrolyte, at a potentiaI some 300 mV more negative than
would be the case with corrosion freely taking place. This change of potential is
effected in a cathodic protection system by causing an appropriate electric current
to flow into the steel from the electrolyte. To bring about current flow, one or
more anodes need to be suitably placed in the electroIyte, and the electric circuit
completed by connection back to the steel structure. In the case of sacrificial
anodes, the emf driving the current is electrochemical in origin. In the case of an
impressed current system, a suitable generator is provided.
The basic problem of design of the system is to determine the number, size and
placement of anodes which give an adequately uniform potential shift at every
point on the steel structure. Because of tbe dangers of over protection, it is as
important to Iimit the maximum potential shift as it is to keep above the minimum
required for protection.
For any particular protected structure, the potential shift at any one position on the
structure is not simple to predict. Not only does it depend on geometry of both the
structure and the anode distribution, but it is affected by the condition of the steel
surface and, through conduction in the electrolyte, by the behavjour of every other
point on the structure. Finite conductivity results in potential drops in the body of
the electrolyte comparable with the changes desired at the steel. In order to begin
design, it is thus necessary to have information OD.the electrical behaviour of the
steel-electrolyte interface, in the form of a relationship between potential shift and
current densityy.This relationship forms the polarisation curve (Section 2) for the
electrode-electrolyte combination. In the case of protected steel in sea water, tbe
situation is complicated by the formation of a calcareous scale on the steel surface,
resuIting from the local change in pH brought about by the current flow. This scale
has a protective effect, and it results in a reduction in the current density required
for a given potential shift.
The ideal design method is doubtless one which provides complete specifications in
one step. This is seldom attainable, and design is more likely to be an iterative
procedure starting from the simplest method available. The present situation is no
exception, and the remainder of this Section considers ways in which calculation
can provide an analysis of a trial design, with a view to testing its adequacy and
improving the design where it fails to meet requirements.
Cathodic protection of offshore structures
75
In the analysis of a trial design two aspects may be distinguished. One of these
concerns calculation of t h e current flow in the resistive electrolyte between t h e
anode (or anodes) and the steel structure, the other concerns the relation between
potential shift and current density at the steel-sea water interface. The flow of
current in a uniform medium is well understood, and various numerical techniques
leading to accurate analysis are available. This problem is considered in Section
5.3.
The steel-sea water interface is far less well understood, and a number of uncertain
and varying effects are involved. This aspect will be considered in Section 5.4.
Prior to these sections the "traditional" design method is presented and discussed
in Section 5.2.
The application of mathematical analysis to the problems of current flow in the
presence of polarisation at the boundaries has attracted attention for many years,
both in the corrosion context and that of electroplating. There is a good
bibliography in Reference 50. The important change to the present day is the
increase of computing power which can make these methods practicable to apply in
rea1-life situations.
5.2
"TRADITIONAL" DESIGN METHOD
5.2.1 Current demand
This starts from the premise that in order to give the required potential shift at the
steel surface, a prescribed current density into the steel is required. It is recognised
that the assumed value of current density varies with conditions such as seawater
temperature, oxygen content and hydrodynamic flow, and hence that different
values are appropriate to different locations. Further, the protective effect of
calcareous scale progressively reduces the current demand. Thus, whereas with a
fresh steel surface a potential shift to -800 mV (with respect to Ag/AgCl) may
require a current density of 0.15 N m 2 ,the same shift might finally be achieved
with perhaps 0.1 N m 2 following deposition of the calcareous scale. An estimate of
the total area of steel to be protected then enables the total current demand to be
predicted. An anode system has to be designed which will deliver this magnitude
of current.
Further considerations depend on whether the system uses sacrificial anodes or an
impressed current.
5.2.2 Sacrificial anodes
Consider the simple situation portrayed in Figure 25, illustrating a steel surface, S,
protected by an anode, S,. Some lines of current flow in the electrolyte are
indicated.
76
MTD Ltd Publication 90/102
S e a water
Sea water
Figure 25
Current flow through a sacrificial anode
The circuit is completed through the anode supports. In an ideal situation, the
surface S, will be at a uniform potential of -800mV (AdAgCl). The potential of
the surface S, is specific to the anode material: for an aluminium-zinc-mercury
anode, a value of -1050 mV (AdAgCI) is typical. Thus the current which flows is
determined by the driving emf (in this case 250 mv) and the resistance between
anode and cathode. With the assumption that S, and S, are each equipotential
surfaces, this resistance is that between two conducting electrodes of the same
shapes immersed in the seawater electrolyte. Even with this assumption (incorrect,
because in practice S, is not at a uniform potential), this resistance is not simple to
determine. Theoretical treatments have taken it to be the same as the resistance
between S, and a large electrode at a great distance (resistance to “infinity”), but
even this is only simple to determine for shapes such as the sphere. In practice a
variety of semi-empirical formulae are used, examples of which are given in Table
3.
TABLE 3 FORMULAE USED FOR CALCULATION OF ANODE RESISTANCE
Modified Dwight,
for long, slender
anodes
(p/ZnL)[ln ( 4 U r ) -11
Modified Petersen
0.21P/A’-’~
McCoy
0.315 p / a
where L = length
r = radius
where A = area
In this table, p = resistivity, in
Q
Cathodic protection of offshore structures
m, length in metres, resistance in ohms.
77
Discussions concerning these formulae can be Found in Reference 51.
As an example, application of the Dwight formula to an anode 1.5 m long and
0.5 m diameter in sea water of resistivity 0.25 Om gives a value of 0.058 n.
Assuming an available emf of 0.25 V,the current output is 4.3 A, sufficient to
protect some 30 m2 of steel surface. As the anode is consumed, the resistance
alters, leading to a reduction in output.
5.2.3 I m p r d current anodes
In the case of an impressed current anode, the current delivered can in principle be
varied at will. However, it is found that anodes are affected at high current
densities, and attention has to be paid to the possibility of high electric field
strengths in the vicinity of an anode. Over protection also has to be avoided.
5.2.4 Distribution of anodes
In general, the total current necessitates a number of sacrificial anodes. These are
distributed over the structure in a way judged to promote uniformity of current
density. The same is true of impressed current anodes, although the output of
individual anodes may be higher (and therefore fewer are required).
5.25 Discussion
It is clear that the theoretical basis of design outlined above is deficient in certain
respects. It is basic to the calculation that the current is spread uniformly over the
steel surface. No account is taken of the shape of the steel surface, which may
greatly influence the local current density. For example, re-entrant corners or
arrays of pipes can give rise to local shielding and consequent under protection.
The Iack of uniformity of current distribution also means that the steel surface
should not be assumed to be at uniform potential, another assumption inherent in
the calculation. The value of anode resistance arrived at by the use of formulae
such as those in Table 3 is also liable to be incorrect. In principle, each anode
shape requires its own formula, and in any case the resistance depends on the
proximity of the steel surface. In the case of impressed current anodes, a Further
difficulty arises in that the voltage drops caused by the high current densities within
the anode material may make it incorrect to assume that the anode surface is at
uniform potential. (This assumption is usually satisfactory for sacrificial anodes.)
It is relevant therefore to enquire if other methods are available which will lead at
least to a more accurate estimate of current and potential distribution over the steel
to be protected. Such methods are now discussed.
5.3
FORMULATION OF THE MATHEMATICAL PROBLEM
Referring to the simple configuration of Figure 25, the problem is to determine the
current flow in the prescribed geometry subject to:
(a) the surface, S,, being at a constant potential:
78
MTD Ltd Publication 90/102
(b) the potential at any point on the surface S, being related, through a
polarisation curve, to the current density at that point.
Before further discussion, the matter of a suitable definition of potential has to be
discussed. Electrochemical potential is defined as potential of the metal with
respect to the electrolyte, as measured by some suitable half-cell such as Ag/AgCl
(i.e. it takes the electrolyte as the reference level). This is not satisfactory for
analysis of a situation in which the electrolyte is not at a uniform potential.
Consider Figure 26. In this illustration, the measurement of potentiaI at three
points is illustrated, at C' , very close to the steet; at A' ,very close to the anode;
and at a point P in the electrolyte. In Figure 26(a), the link between metallic
anode and metallic cathode has not been established, so that the electrolyte is at a
uniform potential. The reading of the half-cell at P is Ecorr,the electrochemical
potential of the steel in sea water under freely corroding conditions. Making the
link between anode and cathode causes current to flow in the electroIyte from
anode to cathode (Figure 26(b)). Thus the cell at P records a potential Ep more
negative than Etolc,say Ec - Ep- The cell at C' also reads more negative, E,,,, + 9,
q being the overpotential; the cell at A' will continue to read EA provided the
anode does not exhibit an overpotential.
{ a ) No link between anode and cathode
Figure 26
( b ) Link made between anode and cathode
Measuremenf of e l e d r o d e potential
The variable chosen for the analysis is that denoted by V * above. Allowing P to
move to the point C'
Cathodic protection of offshore structures
79
AIowing P to move to A’ ,
or
VA
= E,- EA
(>O)
Within the electroIyte, assumed homogeneous and linear, the potential V has to
vary in such a way that no current accumulates at any point, only leaving or
entering the system at the boundaries S, , S, This condition is expressed
mathematically by saying that V satisfies Laplace’s differentia! equation, which in
Cartesian co-ordinates has the form
.
=O
The current density is given by the equation
Of particular interest is the current density at the steel surface, J,, which may be
symbolically expressed in the form
the direction n representing the normal to S, out of the electrolyte into the
cathode. The equation has to be solved subject to the conditions:
over SA V = E,
over S,
V = -
9
- EA = V,
(53
(5.7)
In tbese equations, Jc is given by Equation (5.5) and q(J3 indicates the
overpotentid corresponding to that current density.
A third condition results from the connection between anode and cathode
total current entering the cathode = total current leaving the anode.
(5.8)
It is known that with such conditions at the boundaries, a unique solution for V
results.
Laplace’s equation is of widespread application, and numerical methods of solution
are available. It may be noticed that the classical conditions, to which many
numerical programs refer, are either constant potential on both boundaries, or
specified current density. The fact that Equation (5.7) does not fall into these
categories may make certain numerical routines of the “Laplace solver” class
inapplicable. Before proceeding further, the nature of Equation (5.7) needs to b e
considered.
80
MTD Ltd Publication 90,402
5.4
THE STEEL-SEAWATER BOUNDARY
Equation (5.7) applies at each element of surface area of the cathode and can thus
be simulated by a 1-dimensional cell as indicated in Figure 27.
_ -
Simulation of one-dimensional cell
In such a cell, the resistance of the sea water is negligible compared to the effective
resistance of the cathode process and the circuit resistance. A plot of potential
electrode reading, E, against current density, J, over the range of interest takes the
form shown in Figure 28, in which the numericat values are to be taken as
indicative only. Such a curve may be taken as relevant to clean steel. The precise
values depend on factors such as the content of oxygen in the sea water,
hydrodynamic flow past the steel (which has the effect of altering the oxygen
content) and temperature: a greater oxygen content requires a greater current
density for the same potential shift. These effects can be simulated in an
appropriate experimental configuration. In any numerical technique, the use of a
non-linear relation of this type greatly increases the time for computation, and for
many purposes a linear approximation is used. A possible line is indicated in
Figure 28.
If such a cell as indicated in Figure 27 is allowed to run, as time progresses it is
found that the current drops. This is because of the formation of calcareous scale,
as mentioned earlier: the passage of current and consequent reaction raises the pH
of the sea water immediately in front of the steel, causing the preci itation of
calcium carbonate and magnesium hydroxide. The general effect(5z53)is to give a
sequence of polarisation curves as indicated schematically in Figure 29. The effect
is difficult to quantify. The nature of the scale is dependent on the current density
as well as on the time elapsed.
Cathodic protection of offshore structures
81
-1100
V ImV I
-1000
- 900
-800
- 700
-600
- 500
0.1
0
0.2
Current density,) ( A h 2 )
E ImV, Ag l A g C 1 I = E , , , , + ~ = €c,,rr-V
Figure 28
Electrode potential plotted againsf currenf density, in sea water
Time
Figure 29
82
Polarisation curves
MTD Ltd Publication 90/102
For optimum protective effect, the current density has to be neither too high nor
too low: unduly high and unduly low current densities both produce less protective
scales. Neither is it clear whether or not a final state is approached, a matter
complicated in any event by removal of scale during storms. All-in-all, it seems
that at the best only a crude allowance For scale formation can be sensibly built into
calculations.
5.5
THE MATHEMATICAL MODEL
The combination of a Laplace-solving computational routine with appropriate
approximations to the boundary conditions constitutes a model which can be used
to investigate system behaviour and thus act as an aid to design. The following
sections consider the limitations and possible uses of such a model.
5.5.1 The model
Modem computing techniques make feasible the obtaining of solutions of Laplace's
equation in situations of considerable complexity. It is in the boundary conditions
that the greatest uncertainty lies. Tt has to be remembered that:
The accuracy of the mathematical model is only as good as the accuracy of the
data used.
.
The accuracy of the model can only be assessed by comparison of predictions
with data measured in the field. Reliable, detailed data on full size systems is
sparse.
A further point may be made:
I
Exact predications may not be necessary, provided the margins of protection
can everywhere be guaranteed adequate.
"Improved" calculations can be considered in three steps:
.
The "traditional" method can be improved by eliminating uncertainties
connected with estimation of resistance. This could be done by taking anode
and cathode surfaces as equipotentials and evaluating current density
everywhere. Alternatively, the cathode condition might be assumed to be
constant current.
The linear approximation to the polarisation curve at the catbode may be
incorporated, in the form
av
(an),
=KV
Cathodic protection of offshore structures
(5.9)
83
in which K is a constant having dimensions of m-'. For sea water with
resistivity 0.25 Qm,a value of K = 0.1 corresponds to 0.12 A/mz at 300 mV.
By repeating with a lower value of K some indication of the effect of
development with time can be obtained.
The use of an experimentally determined polarisation curve can be considered
at the expense of greatly increased computation times.
In the process of making any choice between these alternatives, it is essential to
bear in mind the various uncertainties which may be built into the basic input data,
and to avoid endeavouring to obtain more accurate results than the data permit.
5.5.2 Limited analysis
The above section looked briefly at tbe possibility of analysis of a complete system.
The traditional design method of Section 5.2 suffers principally in that no account
is taken of the effect of the geometry of the structure, with the result that certain
critical a r e s may be under protected. The close proximity of two surfaces, as in
nodes or arrays of pipes, may result in screening from the anode. In such
situations, there is no obvious guide for tbe designer. For example, in the node of
Figure 30 how close needs an anode to be placed in order to protect the weld
areas? A mucb simpler example is illustrated in Figure 31, a single anode on one
side of a tubular member. It is not obvious how well the side of the member
opposite to the anode is protected. Being truly three-dimensional, numerical
methods are needed to solve this problem. Such examples can be multiplied, and
much useful information can be obtained with the aid of a suitable program.
Figure 30
Typical node on a tubular member
84
h4TD Ltd Publication 901102
The above paragraphs essentially form an almost philosophic introduction.
Practice is far more complicated. Tt is not difficult to imagine the application of
these methods to a "simple" structural configuration like that of Figure 25. It is
clearly far more demanding to apply them to a complicated structure containing,
for example, many nodes of the type shown in Figure 30. It may well he necessary
to tackle an overall design, of perhaps an oil rig, in stages. The whole structure
may be investigated on a coarse scale, subsequently applying more detailed analysis
to individual elements, such as nodes. A lot depends on the type of computer
program used and it has to be emphasised that such a program is a specialised
development (34 to 56)
55.3 Analysis of operational systems
The above hvo sections considered design problems, although emphasis was placed
on the need for adequate basic data derived from experiment. A somewhat
different application is the possibility of using the model as an aid to the survey of
an operational system. The parameter most likely to be measured is potential, at as
many points as can reasonably be attained. Although current density is of
significance, it is more difficult to measure. The results of a potential survey can be
used as input data to the model, and they are sufficient to make a complete
solution possible. From this solution, current densities can be derived, and anode
outputs estimated. Such a process can also be used to facilitate the positioning of
an extra anode if improved protection is required in some particular area.
5.6
COMPUTATIONAL METHODS
It is not the purpose of this review of mathematical modelling to go into details of
any particular programs. However, it may be of use to briefly explain some of the
terms used in discussing such programs. Before doing so, it should perhaps be
stated that there is nothing new in these methods: work on the application of
numerical techniques to cathodic protection systems is at least 20 years old. What
has changed is the speed at which computation can be effected, making analysis of
useful structures feasible. The use of computer graphics also makes both the input
of data and analysis of output data simpler and more understandable.
There are three methods of tackling the numerical solution of Laplace's Equation
(5.3):
finite differences
finite elements
boundary elements.
With each of these, the differential equation is used to derive a set of h e a r
algebraic equations in which the unknowns are potentials at a large number of
points, and the knowns relate to values on the boundaries. The structural shapes
are reflected in the coefficients and form of the equations. Techniques are
available to solve such sets of equations with very large numbers of unknowns.
Cathodic protection of offshore structures
85
Figure 31
Figure 32
Single node on a tubular member
Resistance network to replace
conducting electrolyte
5.6.1 Finite differences
In this method(s7),potentials are taken at an array of points regularly spaced over
the region of interest, perhaps on a cubic lattice, of sufficiently small size to allow
accurate simulation of boundary shapes. The differentials of potential are then
repIaced by differences, e.g.&/ax becomes (V(x + h) - V(x))/h. Hence the term
"finite differences". This process is equivalent to replacing the conducting
electrolyte by an array of resistors, as indicated in Figure 32. This is the basis of
the "Resistance Network" analogue approach. The number of points is of the
order of ( L/h)3 where L is a typical dimension of the region in which the field is
varying. This can clearly be very large.
5.6.2 Finite elements
In this methodQ8),a particular algebraic form approximating the potential over a
limited region is assumed. The method is most clearly explained in two
dimensions, although extension to three is straightforward. In two-dimensions, the
space is divided into triangles, and over each triangle the potential is assumed to
vary linearly. The three values at the three vertices are the unknowns. An overall
expression for field energy is set up and minimised with respect to the unknown
potentials, thus leading to a set of linear algebraic equations as before. This
method has fewer unknowns, because of the advantage which can be gained by
using larger triangles in regions where the field is clearly changing slowly. Setting
up a suitable mesh of triangles is carried out by computer program.
86
MTD Ltd Publication 90/102
5.6.3 Boundary elements
This method(’g) makes use of the fact that values of potential over the boundary of
a current-carrying region determine the potential everywhere, and hence also the
electric field gradient at the boundary. The bounding surfaces are divided into
suitable elements of area, and for each an unknown potential and normal field
gradient is assigned. Thus, the unknowns depend on elements of area rather than
elements of volume, and they are much reduced compared to the earlier methods.
This advantage is to some extent offset by the fact that the form of the equations
makes them less convenient to solve. The method is particularly advantageous if
several runs are needed, each with a different set of conditions on the same
geometrical boundaries. This may well be a useful feature for calculations in the
field of cathodic protection.
5.7
ANALOGUE MODELS
In many spheres of engineering, uncertainties can often be resolved with the aid of
reduced scale models. In the present instance, this method is not of straightforward
application. The reason for this may be seen as follows. Suppose that a potential
field satisfying the equations of Section 5.3 has been found for a full-size structure,
and let the potential found over the steel surface be described by a function V,. At
any point on that surface, this potential is related to current density J, by Equation
(5.7), V, = - q(J3 and J, is also related to the field by Equation (5.5). Consider
now a model reduced in scale by a factor A (> 1) and keeping the same point-forpoint potentials over anode and steel as in the full-size system. The new value of
(dV/dn), is increased over the old by a factor A, with a corresponding increase in
current density. Thus, in the new model, Equation (5.7) is no longer satisfied,
showing that an experiment with a reduced scale system does not give correct
potentials. However, it is observed from Equation (5.5) that if the reduction in
scale is accompanied by an increase in resistivity by the same factor, J, and V,
remain in the correct relationship. Such an increase in resistivity can be brought
about by dilution of the electrolyte. However, such dilution then leaves uncertainty
in so far as it may affect electrochemical polarisation and the formation of
calcareous scale.
Thus, reduced-scale modelling does not provide a simple method of assessment.
Experiments reporting investigations using dimension and conductivity scaling are
reported in Reference 60.
Cathodic protection of offshore structures
87
Section 6
Cathodic protection systems for steel offshore
structures
Cathodic protection of offshore structures
89
6. Cathodic protection systems for steel offshore structures
6.1
INTRODUCTION
The purpose of this Section is to provide guidance for the design and installation of
successful cathodic protection systems. Because of the complexity of some modern
offshore steel structures, and of the environmental factors which can materially
affect their in-service operation, it is not intended to cover every combination of
structure type and operational environment. Rather, this Section attempts to
identify the principal factors which should be addressed when preparing the design
of a cathodic protection system and to make recommendations as to how sucb a
system can be developed and put into operation.
The reader is cautioned that the design of cathodic protection systems should be
handled by suitably experienced and competent corrosion specialists. Optimum
designs are most likely to be achieved when the designer is an integral member of
the project team responsible for the overall design of the structure to be protected.
This tends to minimise the problem of communicating valuable, frequently vital,
information between the cathodic protection designer and engineering disciplines
whose work is affected by t h e cathodic protection system. The importance of
establishing and maintaining good communications in the design phase can be
readily appreciated from the wealth of information necessary for completing a
detailed cathodic protection design (Table 4).
6.2
DESIGN OBJECTIVES
The principal objectives of the cathodic protection system design can be
summarised as follows:
.
to provide and efficiently distribute sufficient current to achieve the selected
criteria for protection
to provide complete protection to the structure for its full design life, or as
otherwise specifically agreed with the appropriate certifyrng authority
to consider making provision for any foreseeable additions to the structure or
changes in its operating parameters, or in the environmental conditions.
to locate all component parts of the system to minimise the possibility of their
disturbance or damage, without compromising the effectiveness of the protection.
to ensure that the system does not induce deleterious interactive effects on
associated structures or pipelines or any existing pipelines and structures. Where
interactive effects cannot be avoided, the designer has to ensure that appropriate
measures are taken to obviate deleterious consequences.
to ensure that the system does not jeopardise the integrity of the structure for its
full design life.
Cathodic protection of offshore structures
91
TABLE 4 INFORMATION TO BE CONSIDERED IN PERFORMING CATHODIC
PROTECTION DESIGN WORK
A
STRUCTURE DESIGN DATA
B
O F F S H O m SITE LOCATION DATA
Design life required of CP system
and the structure.
Water depth, oxygen content,
velocity, turbulence, temperature,
resistivity, tidal effects and
suspended solids.
Fully detailed and dimensioned
construction drawings.
Chemical composition of the water
General arrangement drawings showing
its relationship to the sea bed, lowest
astronomical tide level (LAT), mean
water level and maximum water level
for all operational conditions.
Numher, size and location of raiser pipes
conductors, I-tubes, caissons and other
appurtenances.
Extent of use and type of protective
coatings
Availability of electrical power
(for impressed current CP)
Presence in the water or sea bed
of pollutants, depolarising bacteria
or marine borers.
Geological nature of the sea bed
and its susceptibility to scour
Adjacent facilities including pipefines and details of their cathodic
protection system
Susceptibility to stratification of
the water and the resultant effect
or its resistivity temperature and
oxygen content.
Proposed construction schedule
Structure fabrication methods and
fabrication site (if known)
Any weight limitation constraints
of the installed CP system.
Performance history of previous or
or existing CP systems in the same
environment.
Safety requirements.
Protective current density requirements
to achieve the applicable protection
criteria, ohtained from site surveys
or reliable documentary sources.
Constraintdlimitations on the installation and in-service maintenance/
monitoring of the CP system,
Susceptibility to adherent marine
fouling, including type, rate of
growth, and variation with water depth.
Use of metallic materials of construction more noble than carbon steel
which would affect the CP system design.
Use (if any) of high strength steels
or other metallics used in the structure
which may be suhject to a reduction in
mechanical properties when under
cathodic protection.
92
MTD Ltd Publication !Who2
6.3
GENERAL CONSIDERATIONS
6.3.1 Codes and Standards
Prior to the start of design work, the Code or Standard to which the design must
comply has to be established (See Section 11). This information is sometimes
defined in the design contractor’s conditions of contract, and usually alIudes to
codes or standards issued by one of the following:
appropriate certifying authority (e-g. Lloyd’s, DnV, Al3S etc.)
.
owner or operator of the structure
appropriate government agency
designer’s own corporate standards,
The most commonly used code for structures located on the North West European
Continental Shelf is DnV Recommended Practice RPB401 “Cathodic Protection
Design”(’), adherence to which is usually mandatory for structures located in the
Nonvegian Sector of the North Sea.
For other offshore locations, compliance with a specific Code is frequently not
imposed, and the selection of a Code or Standard is left for the cathodic protection
designer to propose. However, in selecting a Code or Standard, the minimum
requirements have to be mutually agreed between the owner and operator of the
structure and the applicable certihing authority.
It should be appreciated that DnV RP8401,NACE Standard RP-01-76(1983)(”)
and other Codes and Standards are recommended practices, and they do not
provide exhaustive information on the design of cathodic protection systems.
Therefore, wherever possible, the designer should adopt design parameters and
practices which can be substantiated by a comprehensive body of proven and
reliable data derived from long-term experimental or operational experience
directly applicable to the structure under consideration and in relation to its
operating environment. When such data are not available or are incomplete, the
designer should aIways err on the conservative side in his proposals, paying due
consideration to the multitude of operating and environmental factors which affect
the performance of cathodic protection systems and the level of protection to be
achieved in service.
Where the design code or standard has not been specified, the designer should seek
clarification from the owner of the structure.
6.3.2 Information required for the design
Information required to prepare a comprehensive cathodic protection system
design varies to some extent, depending upon the type, complexity and operational
parameters of the structure.
Cathodic protection of offshore structures
93
As a general rule, the information required to design the cathodic protection
system can be divided into the following categories:
.
.
technical and operating data for the structure
offshore site location data and existing corrosion data pertinent to the offshore
site location.
Table 4 indicates the information considered to be needed to allow the cathodic
protection designer to perform a comprehensive detailed design. For structures
designed to be operated at more than one location during their operational life, the
information listed in Column B is required for eacb offshore location.
6.33 Current density requirements
Detailed knowledge of the offshore site environmental conditions needs to available
in order to determine the most important cathodic protection design parameters,
which are the cathode current densities required to achieve and maintain adequate
cathode polarisation.
The environmental factors which influence cathode current density requirements
are included in Column B of Table 4. However, it is the complex interaction of
these factors which will determine the level of current density required (see also
Section 2). Although electrolyte resistivity is an important factor in determining
the aggressivity of the environment in almost all situations (thus influencing the
current density requirements), it is the presence and abundance of depolarising
agents, principally oxygen, which have the most profound effect. Lowering the
temperature of a given water composition increases its resistivj ty, but also increases
oxygen solubility.
If turbulence and increased water flow is then introduced, the availability of oxygen
at the cathode surface is further increased, so that higher current densities are
required to achieve protection, If water velocity and turbulence are sufficiently
high (particularly if the water contains suspended solids), the calcareous deposit
normally formed on the cathodic surface at protective potentials may be prevented
from forming, or, if formed, may be mechanically stripped off in service, thus
leading to high levels of current density being required to achieve and maintain
protection.
NACE Standard RP-01-76(I2)summarises environmental conditions at several
offshore geographical locations, and it provides a very approximate guide to the
current density required to maintain protected potentials on bare steel once
polarisation is established. More specific current density recommendations are
given in Table 5 , reproduced from DnV Recommended Practice RPB401‘’).
Guidance is provided on initial, average (mean) and final current density
requirements for various offshore locations. The initial and final current densities
are higher than those recommended by DnV in earlier publications in accordance
with current best practice, but some authorities consider the DnV values unduly
cautious.
94
MTD Ltd Publication 90/102
TABLE 5 GUIDANCE O N MINIMUM DESIGN CURRENT DENSITIES FOR
CATHODIC PROTJZCTION OF BAlRE STEEL
I
AREA
Current density (mA/m2)
Initial
North Sea (northern sector, 57' to 52' N)
North Sea (southern sector, up to 57O N)
Arabian Gulf
India
Australia
Brazil
West Africa
Gulf of Mexico
Indonesia
Pipelines (burial specified)
Saline mud (ambient temperature)
180
150
130
130
130
130
130
110
110
50
25
Mean Final
90
90
70
70
70
70
70
60
60
40
20
120
100
90
90
90
90
90
80
80
40
15
The experience from areas north of 62% in the North Sea is limited. Initial design
current densities in t h e range 250 to 300 mNm2 may be necessary in order to
obtain adequate protection in these areas.
This recognises the findings of several studies in recent years that a higher initial
current density assures a more rapid and complete polarisation of the structure and
the more complete formation of calcareous film, resulting in a reduction in the
mean current density required to maintain adequate polarisation and film
formation. The increased final current density ensures that the structure always
remains polarised or is rapidly re-polarised after periods of heavy weather and
turbulence. As a further precaution against the depolarising effects of storms and
turbulence, an increase in the current density applied to the uppermost parts of the
submerged structure is recommended (e.g. DnV RPB401 recommends that the
current densities in Table 5 should b e increased by 10% for the first 20 m of the
structure below mean water level).
The minimum current density values recommended in DnV RPB401 are based on
extensive experimental and offshore operational data, and as such they are
considered to provide a sound basis for design purposes. The use of lower values
should only be considered with caution. If adopted, they have to be substantiated
by a comprehensive body of supporting data based on long-term monitoring and
operation experience. For example, some operators with comprehensive
monitoring systems or survey practices tend to use lower values of mean current
density. These are based upon their own comprehensive body of supporting data
for their structures operating in specific geographical locations.
6.3.4 Protective potentials
The most widely accepted criterion to determine that satisfactory cathodic
protection has been achieved is the measurement of the structurdelectrolyte
interface potential with respect to a stable reference electrode.
Cathodic protection of offshore structures
95
For offshore applications, the two most important reference electrodes in
commercial use are silverhilver chloride (AglAgCl) and anode zinc (Zn)
conforming to US Mil Spec. 18001J(6') or ASTM B418 Type 2@') high purity zinc.
The silver/silver chloride reference electrode potential is affected by the chloride
concentration of the electrolyte and thus by the electrolyte resistivity. Figure 61 (on
page 199) gives the corrected potential €or various resistivities of water at 2OoC.
However, for a given salinity, resistivity increases with decreasing temperature and
Figure 66 (on page 276) may be used to determine resistivity €or a variety of
salinities and temperatures.
For steel structures located in clean aerated sea water and in anaerobic conditions
such as saline mud, protection is generally accepted to be achieved at temperatures
up to 2S0C when the structure potentials are in the ranges shown in Table 6.
TABLE 6 POTENTIAL LIMITS FOR CATHODIC PROTECTION OF STEEL
1
Protective potential
-
Environment
Positive limit
Negative limit
Aerated sea water
-8OOmV wrt AglAglAgCl -1050mV wrt Ag/AgCl
O.OmV wrt Zinc
+250mV wt Zinc
Anaerobic
conditions
-900mV wrt Ag/AgCl
+ 150mV wrt Zinc
- 1050mV wrt Ag/AgCl
O.OmV wrt Zinc
It is not sufficient to achieve protected potentials at some locations on a structure
but not others, nor just during the calmest periods of weather but not during winter
storms. Complete and efficient cathodic protection demands that all immersed and
buried steel surfaces should be maintained at the desired structurdelectrolyte
potential for all environmental and operational design case conditions and
throughout the full life of the structure.
Steels and non-ferrous alloys possessing a high yield strength may be subject to
hydrogen embrittlement if subjected to potentials tending towards the negative
potential limits given in Table 6. Caution should therefore be exercised when
deciding the maximum negative potential to which such materials are subjected.
More detailed information on the effect of cathodic protection on mechanical
properties of metals is given in Section 3.
Should a reliahle body of data indicate that for a given component or material an
unacceptable reduction in mechanical properties of critical cathode circuit
components would occur if polarised to potentials more negative than around
-900 mV Ag/AgCl, the design of a conventional sacrificial or impressed current
system which meets this stringent requirement becomes extremely difficult. If
anode locations can be remotely sited from such susceptible components, it should
he possible to design a cathodic protection system to fulfil this polarised potential
96
MTD Ltd Publication 901102
criterion. However, if this is not possible, it is necessary to resort to alternative
less conventional measures. For example, at least one offshore operator to date'(63)
has successfully employed thermal sprayed aluminium coatings for critical high
strength steel components on a major North Sea structure, as an alternative to a
conventional sacrificial anode system. The benefit in using this galvanic coating is
that it provides an essentially uniform protected potential of -800 to -900mV
AgAgC1 over the entire coated surface area.
6.3.5 Choice of system type
There are three types of cathodic protection system, each of which, when correctly
designed, installed and operated, can effectively protect a fixed offshore steeI
structure for its design life. These are :
Sacrificial : comprising anodes cast from reactive metals (normally zinc or
aluminium alloys), which are more electro-negative than the structures requiring
protection, and which require no external source of power.
Impressed currenf :comprising anodes manufactured from materials which are
essentially inert and powered b y an external source of direct current.
H y b r 2 : comprising a mixture of sacrificial anodes and externally powered
impressed current anodes.
The principal technical advantages and disadvantages of sacrificial, impressed
current and hybrid systems are summarised in Table 7.
The use of the term "impressed current system" can be misleading, because for
most offshore applications an impressed current system is used in combination with
a small number of sacrificial anodes, forming a hybrid system. Sacrificial anodes in
hybrid systems are provided on structures to ensure that adequate polarisation of
the critical nodes is maintained at all times, even if the power supply to the
impressed current anodes fails or is switched off temporarily to permit manual
underwater inspection or cleaning of the structure by divers. Some early impressed
current systems were provided with inadequate sacrificial anode back-up to
perform this critical task, and significant corrosion damage has been reported in
times of unplanned and planned impressed current shutdowns.
The same considerations apply equally to jacket structures with one important
addition, namely that a power source to drive the impressed current system is
generally not available until the topside power generation equipment is installed
and commissioned. On large deepwater jackets in the North Sea, this may be a
year or more after installation of the jacket, protection for t h e interim period being
provided by high current, short-life sacrificial anodes.
It is strongly suggested that designers contemplating an impressed current system
for North Sea applications should provide full sacrificial back-up. The sacrificial
anodes should provide full protection for a minimum of 2 years, plus an allowance
for periods of possible impressed current system shut-down during subsea surveys
and maintenance throughout the design life.
Cathodic protection of offshore structures
97
The obvious technical attractions of sacrificial systems, as illustrated in Table 7,
make this the type most often chosen for offshore structures. Also,for many
offshore structures, sacrificial systems are the most economical option to the Owner
when taking into account both capital expenditure and the running costs over the
design life of the structure. However, generalisations on economic advantages and
disadvantages can be misleading, because they differ widely for each type and size
of structure and according to the design constraints imposed by the environmental
conditions prevailing at different offshore locations. For this reason, economics are
not included in Table 7.
In view of the inherent simplicity and reliability of a properly designed sacrifical
system, it is very easy to dismiss impressed current or hybrid systems at the outset
of design work and thereby to overlook the less obvious (but nonetheless real)
benefits such systems have to offer, principally weight savings and operational
flexibility. Two notable projects where the advantages of hybrid systems were
constructively exploited are the Murchison and Hutton platforms in the UK Sector
of the North Sea.
Faced with severe weight constraints, the designers of Murchison and Hutton
carried out detailed assessments of alternative sacrificial and impressed current
designs. These showed that although sacrifical anodes could not be dispensed with
entirely, substantial weight savings could be made by using impressed current
systems as the primary means of protection on both platforms.
This was despite their vastly different structural configurations, Murchison(w) being
a deep water conventional jacket, and H ~ t t o n @the
~ ) world’s first tension leg
platform.
The adoption of hybrid systems for weight-saving reasons on Murchison and
Hutton is significant, for this is perceived to be their primary advantage over
sacrificial systems. In the case ofHutton, the installed weight of the primary
impressed current system, plus supplementary sacrificial anodes located close to the
main node joints,was approximately 60 tonne. An equivalent totally sacrificial
system would have weighed around 250 tonne.
In most cases, impressed current systems are more likely to be commercially
competitive for buoyant structures such as tension leg platforms and semisubmersibles than for conventional jacket structures.
The relatively simple geometry and large, flat surfaces of buoyant structures are
ideally suited for protection to be provided by a small number of bigh current, low
voltage, flush-mounted anodes. Cables to reference electrodes and anodes can be
easily and economically routed through ballast tanks and man access ways in the
pontoons and columns of the hull.
98
MTD Ltd Publication 901102
TABLE 7 PRINCIPAL ADVANTAGES AND DISADVANTAGES OF
SACRIFICIAL AND IMPMSSED CURRENT SYSTEMS
ADVANTAGES
iACRIFIClAL ANODE
SYSTEM
IhiPRESSED C U R R m
iimple, reliable and
ree from in-service
)perator surveillance
Flexibiliry under
widel y-varying
operating conditions
Flexibility under
idtly-vaI$ng
operating conditions
iystem installation
s simple
Weight advantage for
large capacity, longlife systems
Weight advantage for
large. capacity, long -
Arge weight penalty
o r large capacity,
ong-life systems
Relative complexity
of system demands
high level of detail
design expertise
Relative complexity
of system demands
high level of detail
design expertise
aesponse to varying
iperating condirions
s limited
Systems installation
is complex, and a
power source is
required
System installation
is complex, and a
power source is
required
Bydrodynamic loading
:an be high
Perceived diver risk
from electric shock
Perceived diver risk
from electric shock
(see para 6.4.4.1.)
In-service operator
surveillance required
i n service operator
surveillance required
Permanent potential
monitoring system
cssen tial
Permanent potential
monitoring sysrem
essential
SYSTEM
HYBRID
SYSTEM
life systems
krmanenr potential
nonicoring system
Jot essential
DISADVANTAGES
Vulnerable to loss
of power
Not recommended for
North Sea wirhout
full sacrificial
back-up (i.e. as
part of a Hybrid
System)
Impressed current systems may also be cost competitive for conventional jacket
structures of simple geometry, located in relatively benign offshore waters, and on
which reference electrode and anode cables can be safely instalIed in substantial
conduits routed along the outside of the structural tubulars.
Impressed current systems are less likely to be cost competitive on large jackets of
complex geometry located in hostile environmental conditions. Complex node
geometries are unlikely to allow large capacity anodes to protect all surfaces
adequately on account of shielding effects. Moreover, the hostile environment may
demand the difficult and expensive routing of anode and reference electrode cables
inside the structural tubulars in order to ensure their mechanical safety. This
necessitates a large number of stress-raising penetrations in the tubulars below
water adjacent to the anodes and reference electrodes.
Cathodic protection of offshore structures
99
In summary, the choice of system type should be based on a tborough technical
and commercial assessment of each individual structure, taking into account all
operational and environmental considerations.
6.3.6 Cathodic protection and coatings
The subject of cathodic protection and coatings is covered in detail in Section 4.
However, some important aspects of the interaction between protective coatings
and cathodic protection are discussed here in relation to the cathodic protection
design process.
The usual objective of applying high performance coatings is to improve current
distribution and to reduce the current density required to achieve protection, thus
reducing the weight and cost of the cathodic protection system.
For some buoyant structures such as tension Ieg platforms or semi-submersibles,
the saving i n weight may alone justify the use of a high performance coating system,
even if no saving is made on the combined cost of coatings and cathodic protection.
For other structures (even conventional jacket structures), detailed costings may
reveal an overall economic benefit in completely or partially coating the structure
in order to itduce the size of the cathodic protection system.
However, the cathodic protection designer needs to be suKciently confident that
any such theoretical cost and weight savings are valid, and that they can be
substantiated by long-term testing or operating experience. A coating system which
performs less efficiently than the design study predicted may well increase cathode
current density requirements to values greater than the cathodic protection system
can deliver. Furthermore, the increased current demand aimost certainly reduces
anode life to less than that of the original design. It is therefore imperative that
great care should be exercised in the choice of supplementary coating systems,
including their requirements for surface preparation and application, and the
handling of coated steel surfaces prior to placing in service.
Furthermore, caution should be exercised when specifying any coating which is
required to endure frequent or even occasional abrasive cleaning (e.g. to remove
fouling), particularly if the cathodic protection system is to be designed on the basis
of an assumed long-term efficiency of such coatings.
The fundamental difference between applying cathodic protection to a coated
rather than a bare structure is that, as the coating efficiency declines through
natural degradation, wear, and mechanical damage, the current required to
maintain polarisation increases. This is the exact opposite of the characteristic of
current demand against time for most bare structures, where the initial current
density required to achieve polarisation is high but reduces considerably, because
of the formation of calcareous films on the catbodic surface (see Section 2.8.2).
These films act as a barrier to oxygen transport, thus effectively performing as a
barrier coating on the cathode surfaces. However, some researchers(&)found that
in seawater temperatures below 2OoC the formation of calcareous films is inhibited,
and that they do not form at all at 3 O C and lower.
100
MTD Ltd Publication 90/102
The dilemma for the cathodic protection designer is the difficulty in assessing the
in-senice loss of coating properties and the depee of mechanical damage the
coating suffers prior to putting it into service and during its service life. Guidelines
on the likely loss of coating efficiency (or breakdown) are shown in Table 8,
reproduced from DnV RPB401. Additional information on coating breakdown is
given in the Appendix, Table 26.
DnV recommend that the breakdown values in Table 8 should be increased if the
coating is "particularly exposed to wear and mechanical damage", but there is no
elaboration on this. Overall current densities for a coated structure are obtained
by multiplying the bare surface current density values in Table 5 by the percentage
of coating hreakdown.
There is little reliable published data on coating hreakdown values for offshore
structures, and while the guidelines given in Table 8 are considered by some to be
over consewative, they remain the most authoritative currently available.
TABLE 8
GUIDE ON COATING 3REAKDOWN CRITERIA
FOR CATHODIC PROTECTION DESIGN
I
Coating breakdown
Lifetim;.years
20
30
40
I 1 1 I
Inirl
2
2
2
Me;
15
25
40
Fin:","'
30
60
90
Another type of coating which may cause complications with the design of cathodic
protection systems is the pre-fabrication primer. Certain steel mills apply a prefabrication primer to the plate or tube during production, the primer providing
protection during shipment, storage and fahrication, and making the steel much
cleaner to handle. However, if pre-fabrication primers are used, it is nof
recommended to reduce the design current densities to less than would be used, for
bare steel.
6.3.7 Anti-fouling coatings
In waters subject to marine fouling, it may be necessary for the owner or operator
of offshore structures to undertake regular cleaning of the immersed steelwork in
order to remove excessive fouling deposits which may otherwise cause excessive
wave loading, leading to structural failure.
In an attempt to eliminate the necessity of cleaning (or at least reduce its
frequency), some offshore owners and operators have studied the application of
inherently anti-fouling materials to the submerged and splash zones of their
structures. A material favoured €or this application is 90:lO cupro-nickel (Cunifer
lo), which is considered environmentally safe, unlike most conventional antifouling paints.
Cathodic protection of offshore structures
101
For the cathodic protection designer, the main concern with the use of noble
metallic materials such as Cunifer 10 is the highly active galvanic couple they can
set up with carbon steel should electrical continuity be established between them.
This results in large amounts of current being drained from the cathodic protection
system whicb it may not have been designed to deliver, thereby reducing the
protective potentials of the structure to possibly unacceptable levels, a k o reducing
the useful life of the cathodic protection system. Additionally, the surface area of
the noble material receiving current tends to lose its anti-fouling properties.
Extreme care should be taken in the testing and assessment of noble metallic antifouling materials to ensure they do not have a potentially detrimental effect on the
performance of the cathodic protection system, and to confirm that any such risk
can be quantified and the necessary precautions taken in the design of the cathodic
protection system.
A further point to b e considered for structures subject to heavy marine fouling is
the mechanical damage which cIeaning operations may cause to coatings in the
submerged and splash zones. Conventional coatings possess little or no inherent
anti-fouling properties, and they tend to suffer similar rates of fouling as bare steel.
6.4
CARRYING OUT THE DESIGN
6.4.1 Design premise
Prior to performing any detailed design work, it is recommended that a Design
Premise be prepared. The purpose of this document is to describe in outline the
designer’s proposals on which the detailed cathodic protection design work will be
based. This should include the following information as a minimum:
Scope, covering:
items of the structure to be protected
.
.
items or locations of the structure where cathodic protection may not be viable
for technical, practical or economic reasons and what alternative protection
measure are proposed
.
items or locations where cathodic protection may be supplemented by coatings.
Sysfem desctiplion, covering:
design life of the system
.
.
.
.
design code or standard to be used
summary of the current densities and protection potentials proposed
whether an impressed current, sacrificial anode or hybrid system is proposed and
the pros and cons of each in relation to the subject structure
.
discussion of the cathodic protection requirements for each significant part of the
main structure and appurtenances.
102
MTD Ltd Publication 901102
a description of the anode materials considered and a discussion of their
performance characteristics
.
discussion of t h e requirements of appurtenances, particularly risers, for
electrical isolatiodcontinuity.
Special studies, covering:
desk studies, field surveys, testing programs, computer modelling programs
considered necessary or desirable to provide sufficient data to develop a safe,
efficient and reliable cathodic protection system.
Obviously, client approval of the Design Premise should be obtained prior to
proceeding with the design to an advanced stage. Until such approval is obtained,
design work is best restricted to caiculation of cathode surface area, estimation of
cathode current demand, determination of anode weight for the various sacrificial
anode materials considered, and acquisition of other basic design data.
6.4.2 Detailed design work
6.4.2.1 General
Much of the large body of data listed in Table 4 is unlikely to be available at the
commencement of the design work. Indeed, its acquisition forms an essential part
of the cathodic protection designers' responsibility as the design of the structure
proceeds. This serves to emphasise the desirability of the cathodic protection
designer being an integral member of the project design team, because the speedy
and accurate transfer of such technicaI information is essential in the design of
cathodic protection systems for offshore projects, which are almost always subject
to tight scheduling requirements.
6.4.2.2 Surfaces $0be prokckd
All external surfaces of fixed offshore steel structures below mean water level
require to be protected by cathodic protection. This includes the main structure
itself and all other metallic appurtenances.
Appurtenances with in-built corrosion allowances, or those which become
redundant after installation of the structure, also need to be included in the
cathodic protection design calculations, because they drain current from the main
structure for as long as they remain in piace and are electrically continuous with it.
It is not possible to list definitively each individual item which requires to be
protected on a "fixed offshore steel structure", because this term embraces a
multitude of different structure types and configurations. The following checklist
provides guidance on typical appurtenances which require to be included in the
cathodic protection design calculations:
Cathodic protection of offshore structures
103
.piles and associated guides
risers (see below)
riser clampdguides
conductors
caissons and associated clamp/guides
.
.
caisson pumps
J-tubes and associated clamps/guides
.
.
.
.
grout lines
electrical conduits and associated clampsiguides
boat bumpers
access ladders
temporary/permanent flotation chambers
mooring tethers
.
foundations for mooring tethers.
To prevent pitting and crevice corrosion of austenitic stainless steels, these should
be protected to the same potentials as prescribed for carbon steels, and their initial
current demand should be calculated on th e same basis as the latter. However, the
reduction of current requirement with time may, in some situations, be less than is
the case with carbon steels, for there is some evidence that calcareous scales form
less readily on stainless steels.
The cathodic protection of duplex stainless steels should be considered with care.
These steels are vulnerable to cracking when over protected if the metallurgical
structure is inappropriate. Welds and their heat-affected zones are the most likely
sites of such unfavourable structures (see Section 3).
6.4.23 Risers
Riser surfaces operating at or below approximately 25'C have usually been made
eIectricalIy continuous with the main structure, protection being afforded by the
main structure cathodic protection system or by sacrificial anodes located on the
risers. Risers operating above 25% are frequently electrically isolated from the
main structure, protection being afforded by the subsea pipelindriser cathodic
protection system.
The reason for differentiating between hot and cold risers is that it is common to
polarise hot surfaces to more negative potentials than cold surfaces, and to achieve
this higher values of current density may be required. It is extremely difficult (if
not impossible) to design a cathodic protection system to achieve radically different
localised current density and polarisation levels on a riser compared to the mass of
the main structure when electrical continuity exists between the two. The practice
has therefore arisen whereby hot risers are electrically isolated from the structure.
I04
MTD Ltd Publication 901102
However, a 1982 NTNF report(67)states that in aerated conditions adequate
protection of hot surfaces operating up to 4S0C is obtained at normal negative
potentials of -800mV wrt Ag/AgCl. No recommendation is made for surfaces
operating above 45'C, because this was the maximum temperature studied.
Work carried out for the UK Department of Energy(42)concluded tbat risers
operating up to 90'C in aerated conditions can also be satisfactorily protected at
negative potentials of -800mV wrt Ag/AgCl.
Also, although the current 1986 edition of DnV RPB401(9) recommends that
normal current densities (see Table 5), should be increased by 2mNm for every OC
by which the hot surface exceeds 25OC, no recommendation is made as to more
negative protection potentials being required for surfaces operating above 25OC.
It can be inferred from DnV RPB401 that it is sufficient only to increase the
current densities applied to surfaces operating above 25'C. Therefore, it follows
that, provided such an allowance is made in the current available td protect risers
(either From anodes located on them or from the main structure cathodic
protection system, as a combination of the two) there is no necessity to isolate
risers from the main structure, if the NTNF and DnV conclusions are correct.
The main provisos to this approach, equally applicable to cold risers, are that more
positive (less protective) potentials on risers and the main structure do not cause:
a shift in the potential of the subsea pipelines to a value more positive than is
required for their protection in an anaerobic environment. This is generally
100 mV more negative than the structure potential itself.
excessive current to be drained from the subsea pipeline anode system to the less
negatively polarised riser and platform, which would lead to an unacceptable
shortening of the life of the pipeline anode system.
No general recommendation can be given regarding the electrical isolation of risers.
The decision remains one to be made by the Corrosion Engineer (designer) in
conjunction with the Pipeline and Structural Engineer on a project by project basis.
6.4.2.4 Other items
In addition to the items listed above, cathodic protection is normally applied to
topside water storage tanks, and, on buoyant structures, to ballast water tanks. The
water boxes on some types of shell and tube heat exchangers are also suitable for
cathodic protection. For all these items, cathodic protection is usually applied in
conjunction with supplementary high performance coatings. For enclosed tankage
or storage vessels, containing essentially stagnant water and to which man-access is
possible, impressed current systems shouId not be used. This is because of the
possible build-up of hazardous chlorine gas, which is evolved at the surfaces of
impressed current anodes, and hydrogen, which is evolved at the cathodic surfaces.
For permanently sealed areas such as seawater flooded jacket legs, dosing of the
water with corrosion inhibitors and biocides is usually an effective and more
economic means of providing protection than by sacrificial anodes.
Cathodic protection of offshore structures
105
6.4.25 Calhode cuwenl requirements
The first basic step in assessing the required current capacity of the system is to
calculate the surface area of the items requiring protection. Although this is a very
straightforward arithmetical task, the importance of achieving a high degree of
accuracy cannot be overstated. To under estimate the surface areas inevitably
results in an undersized system, possibly resulting in a failure to achieve adequate
polarisation of the structure or a reduced system design life, or a combination of
these two defects. Although not sufferingthe same potentially disastrous
consequences as under estimating surface areas, an over estimate results in the
wasteful investment in an oversize system, to the financial disadvantage of the
owner.
Only the most up-to-date construction drawings, showing all amendments and
alterations to the items to be protected, should be used for the surface area
calculations. For retro-fit systems, the as-built drawings should be used,
supplemented with up-to-date diving survey reports which should identify the
presence of any steelwork additions not otherwise recorded. The diving survey
should also determine the condition of any protective coatings used or other factors
which may materially affect the current requirements of the retro-fit system.
To cater for the possible marginat increase in the sizes of structural members,
which sometimes occurs late in the final design of a structure, aIso to provide a
nominal safety factor in the system design, it is prudent to include a contingency
allowance in the surface area calculations or the current capacity of the total
system. A value of 5 to 7.5% is usually considered adequate for this purpose, and
it frequently obviates the need for a time-consuming re-working of the overall
system design. Naturally, design calculations need to be checked to ensure that the
contingency allowance and the distribution of anode current adequately cover any
change in member sizes, and that, if found to be inadequate, the system design is
amended to suit the revised requirements.
A methodicai and consistent approach is required when performing the surface
area calculation in order to design a system which provides adequate distribution of
anode current. This demands an accurate assessment of the surface area of each
individual structural member and appurtenant item, to which the appropriate
cathode current density can then be applied. This is of particuIar importance for
coated surfaces or when differing materials of construction may be used, because
differing current density values may then be required to obtain adequate
p olarisation potentials.
Differing localised environmental conditions demand &heapplication of different
current densities to achieve adequate polarisation potentials.
A typical tabulation of surface areas, current requirements, and sacrificial anode
weight requirements for part of an offshore jacket structure is shown in Table 9.
106
MTD Ltd Publication 90/102
TABLE 9 TYPICAL TABULATION OF SURFACE AREAS, CURRENT
REQUIREMENTS AND ANODE WEIGHT REQUIREMENTS
I
=ONE
CURRENT REQUlIED (A)
BARE
COATED
STRUCIURAL
I
L
MEMBER
LENGTH
DIA.
k OTY
Hark 10
4
(m)
(m)
0.45
.
0.60
14.0
S Z O M BARE
COATED
LENGTH AREA
AREA
(m3
(m)
17.5
TOTAL
(m3
-
24.14
No.
Ding I 1
8 No.
-
2639
COMBINED
I 4.95
I 4.95
I 0.11
hi 0.22
F 0.43
t 0.11
M 0.22
F 0.43
I
I 6.54
M 2.68
301 kg u c b
4No. = I##
26.16
10.72 10.48
tg
F 2.62
I
1
I
I
I
0.36
Dzng 16
4 NO.
Piles
13.50
15.27
10.70
12.10
15.7
20.23
I
.
MUD
1.22
13.69
MUD
.
-
0.92
6.0
17.35
9.3
26.88
Firewater
a92
6.0
17.35
caisson
Note I
13.30
38.44
I 2.42
I 2.42
M 1.00
117 kg u c h
4 NO. = 468 k6
F 0.97
M 1.m
F 0.97
I 4.0s
M 1.66
I F 1.62
I 4.05
M 1.66
F 1.62
I 2.74
I 2.74
M 1.13
F 1.10
MUD
MUD
t 8.76
M 4.38
F 3.29
I 8.76
M 4.38
F 3.29
M 0.15
F 0.28
I 5.38
M 2.21
F 2.44
1 5.45
M 2.36
F 2.16
I 0.07
M 0.15
F 0.28
t 7.69
M 3.16
F 3.08
I 7.76
M 3.31
-
caisson
148 kg cacb
4 No. = 5 9 1 kg
M 1.13
F 1.10
57
Disposal
I 3.m
M 1.26
F 1.22
II 11
1
12.1
I 3.M
M 1.26
F 1.22
I 0.07
1214
5.M
4.88
187 kg each
= 148 k6
4 NO.
127 t p u c h
5 0 8 kg
4 NO.
497 t1each
8 K O . = 3936 kg
276 kg Total
F 3.36
= Iniiial, M = Mean. F = Fmal
Cathodic protection of offshore structures
107
6.43 Sacrificial anodes
6.4.3.1 Magnesium aUoy anodes
There are two generic groups of magnesium alloys used in cathodic protection:
magnesium containing approximately 1.5% manganese as a deliberate addition, and
magnesium containing approximately 6% aluminium, 3% zinc and 0.15%
manganese as additions.
In both cases, incidental impurities naturally occurring in the magnesium need to
be controlled to limit polarisation and auto corrosion. Manganese is added to
sequester iron impurities and to make the anode potential more negative. The high
manganese alloy has a high driving potential, and it may be useful in higher
resistivity electrolytes than the Mg-Al-Zn alloys.
Magnesium alloys are used extensively where a high driving emf is desirable (e.g. in
soils and fresh and brackish waters). Because their corrosion products are nontoxic, they are suitable for use on potable water. Their low density and high
driving emf make them particuIarIy useful for suspended anodes and for temporary
protection where the number of anodes should be limited (e.g for ships which are
laid up or being fitted out). However, maguesium anodes can produce sufficiently
negative potentials to cause deterioration of paints, and they should, therefore, be
used with care in some applications.
Furthermore, hydrogen is evolved from magnesium anodes, and thermite sparking
may occur when magnesium is impacted on rusty steel. The possibility of explosion
hazards should, therefore, be borne in mind. Because of their high driving voltage,
magnesium alloy anodes are not normally used for the permanent protection of
offshore installations. However, they may be used in limited quantity as temporary
anodes to supplement the permanent anode system in order to achieve rapid
polarisation. Such anodes should be designed for a very short operational life with
a high surface area to mass ratio. Once the structure is satisfactorily polarised,
such temporary anodes should be removed to avoid the possible adverse effects on
the structure of hydrogen embrittlement caused by excessive negative potentials.
Magnesium anodes may be used for long periods as high current remote anodes in
specialised applications in which their high driving voltage does not result in the
cathodic polarisation potential exceeding the negative limit given in Table 6 (see
page 96).
6.4.3.2 Zinc anodes (see Figure 33)
Commercial purity zinc is of limited use as an anode material because of excessive
polarisation caused by iron present as an impurity. Anodes cast from high purity
zinc (BS Grade Zn selected to contain less than 0.0014% iron) perform
satisfactorily in offshore applications.
Alloys based on less pure zinc have been formulated in which the deleterious effect
of iron is negated by addition of aluminium, which sequesters the iron as an inert
intermetallic compound, or by addition of silicon which forms an iron-rich dross
which may be removed during t h e casting process. Cadmium may also be added to
promote formation of soft, non-adherent corrosion products on the anode. Typical
alloys may contain 0.5% aluminium with up to 0.1% silicon a n d o r cadmium.
108
MTD Ltd Publication 90,402
Figure 33
Offshore sfrudure zinc reference anode
AlIoys containing small additions of mercury, indium, calcium and lithium have
been proposed for application where higher driving potentials are desired, but none
of these has found commercial application.
At elevated temperatures, zinc undergoes a reduction in both open circuit and
driving potentials, and has been shown to suffer intergranular corrosion with
consequent loss of capacity. Alloys with very low aluminium contents have been
proposed to minimise intergranular attack, but zinc anodes are likely to be of little
practical use at temperatures exceeding 4OoC, because of their much reduced
driving potential.
The low driving potential of zinc is insufficient to cause cathodic disbondment of
high performance coatings used on offshore structures. It does not normally cause
hydrogen evolution at the cathode surfaces, nor is there a risk of thermite sparking.
6.433 Aluminium anodes
Aluminium corrodes to form an oxide film which is tightly adherent, and which
causes rapid polarisation when the pure metal is used as an anode. In chloridecontaining electrolytes, this breaks down to give very non-uniform attack by pitting.
The addition of an alloying element which leads to total breakdown of the oxide
film, or "activation" is necessary to make aluminium useful as an anode.
Cathodic protection of offshore structures
109
Early binary alloys of aluminium with about 5% zinc were only of any use if super
pure aluminium feed stock was used, and no commercially available alloys are now
available based on this combination alone.
There are three generic groups of aluminium alloys used for cathodic protection
anodes. All contain zinc in varying amounts together with tin, mercury or indium
as the activator.
In proprietary compositions, additional alloying elements such as silicon, bismuth,
manganese, magnesium, titanium and others may be included in the final
formulation.
Aluminium-zinc-tin alloys require heat treatment to achieve effective activation,
and electrochemical testing is used in addition to chemical analysis to ensure
adequate quality control. In general, these alloys have been superseded by the
aluminium-zinc-mercury and aluminium-zinc-indium alloys, which offer higher
electrochemical capacity.
Application offshore of aluminium-based anode alloys is restricted to sea water or
seabed muds, and particular compositions are favoured in specific applications. In
particular, not all alloys are suitable when operated at elevated temperatures,
particularly under heat transfer conditions. There are also indications that some
formulations (notably Al-Zn-Sn) suffer intergranular corrosion at lower (5% or
less) temperatures, and that they may be very slow to activate, with reduced
capacity to provide protection.
Some aluminium alloys containing a significant proportion of magnesium may be
liable to age hardening, resulting in serious cracking of the anode casting. Anodes
of this type suffered extensive cracking defects during the fabrication of a large
North Sea jacket, and they were replaced with anodes of an alternative aluminium
alloy composition.
Hydrogen evolution for aluminium alloys is generally very slight, but explosion
hazards should always be borne in mind, because of the risk of thermic sparking
from the impact of light alloys with rusting steel.
The two fundamentally important electrochemical parameters of sacrificial anode
materials are the ampere-hour capacity for a given alloy mass, and the
electrocbemical potential. Much research and development on aluminium
sacrificial alloys over the last 20-25 years has been devoted to increasing their
ampere-hour capacity, while maintaining stable closed circuit potentials in a variety
of operating conditions. This has resulted in the availability commercially of the
highly efficient Al-Zn-Hg and Al-Zn-In alloys which have gained virtually universal
acceptance and hence domination of the offshore sacrificial anode market for most
applications.
Table 10 shows typical ranges of values for closed circuit potential, current
capacity, and consumption rates for several sacrificial alloys. As the wide range of
values suggests, the performance of anode alloys can vary considerably, both within
a generic alloy type and between different generic types.
110
MTD Ltd Publication 90/102
Zn (US
Mil Spec)
Sea water
760-780
0.95
- 1.03
11.2
Zn (US
Marine
Sediments
750-780
0.95
- 1.03
11.2 - 11.7
Mg (High
Purity)
Sea water
I230
1.5
7.1
Mg (High
Potential)
Sea water
1230
1.7
7.1
Mil Spec)
- 11.5
Sediments
Sediments
1
It is therefore essential that the properties of any alloy to be adopted should clearly
be proven to fulfil the needs of each design condition applicable to its intended
use. Proof of the alloy properties should take the form of fully detailed alloy test
reports, preferably recording the results of long-term field testing triaIs, or at least
from long-term laboratory testing programmes.
When considering the use of anode alloys containing mercury, t h e designer should
ensure that the marine authority responsible for the area in which the structure is
to be located has no objections. Some authorities are concerned about the effect of
mercury on marine animals and plants, and they may restrict its use ahove certain
levels. Factors which affect in-service anode alloy electro-chemical properties
include:
.
electrolyte type (seabed mud, sea water, etc.)
electrolyte resistivity
electrolyte temperature
.
electrolyte oxygenation
.electrolyte pollution
anode temperature
.
anode current density.
Cathodic protection of offshore structures
111
As a general rule, increasing anode operating temperatures cause a decrease in
both anode ampere-hour capacity and driving potential. At temperatures
exceeding 5OoC,zinc alloys experience intergranular corrosion, and they should not
be used. At low anode current densities, the ampere-hour capacity of aluminium
alloys tends to decrease significantly. In order to realise t h e performance claimed
by anode manufacturers and thus to ensure the successful operation of the cathodic
protection system, it is imperative that strict quality assurance and quality control
of the anode manufacturing process should be achieved and maintained throughout
production. The subject of quality assurance and quality control is too complex to
discuss in detail here. Suffice it to say that the requirements contained in DNV
RPB401(9)are considered to set the minimum standards for offshore work,with
supplementary requirements for specific project applications to be determined and
specified by the designer.
6.4.3.2 Geometric shape
Sacrificial anodes are generally cast in three basic geometric shapes: long slender
stand-off type, flat plate flush-mounted type, and bracelet type. Typical examples
of the first two basic geometries are shown in Figure 34. Typical bracelet anodes
are shown in Figure 46 on page 156).
.
275
2 2 20 mean
I
I
1
215
,127.5,127-5,
mean
,
1
Anode
alloy
-Doubler
'
A
d
plate
( a ) Stand-off sacrificial anode
---
+-
49 0
I
12cover
450
Anode alloyl
450
I
-----------_--_-_____-_-_-_---_
----_-_
1
3.----
320
( b l Flush -mounted sacrificial mudmat anode
Figure 34
112
Typical stand-off and flush-mounted anode
MTD Ltd Publication 90/102
The most common anode shape used for offshore structures is the long slender type
of trapezoidal or circular cross section. The principal advantages of this anode
geometry are high current o u t p u t and good current distribution for a given mass,
simple fabrication and casting requirements, and highly efficient use of the
available anode alloy capacity (utihsation factor). Stand-off separation between the
underside of the anode alloy and the cathode surface in the range of 250 to 350 mm
has been found to be the optimum for most applications.
Flat plate anodes are generally best suited to complex fabrications where space
limitations prevent the use of larger stand-off anodes andor cathode current
densities are low. Examples are heavily reinforced mudmats and large flat plate
painted surfaces. The designer should determine if the chosen anode shapes can be
more economically chosen from a manufacturer's standard units or whether,
because of the large number required for a new structure, a preferred design could
raise costs. Anode manufacturers offer a large variety of standard anode and insert
core types, the choice of steel insert being usually between bar, tube or rod in
either straight lengths or prefabricated, weld jointed, shapes.
The insert should be structurally suitable for the anode weight and for the forces it
is likely to encounter during its lifetime, including impact, storm damage, wave
action and, possibly, ice. T h e insert should normally be made from weldable
structural steel. Typical grades of steel are BS4360 grades 40A, 43A or SOC, or API
5L grades B, X42 or X52 (See also Section 10).
If anode inserts are fabricated by welding, the latter has to be in accordance with a
recognised, quality controlled standard. Inserts should be pre ared by abrasive
cleaning to a minimum standard of SIS 05-59-00 1967 Sa 2%( 4 4. Zinc anode inserts
are normally zinc coated to BS729@') or to BS1706(70).NACE recommended
practice RP03-87(7') may be followed.
Aluminium anode steel insert specifications are similar to those for zinc, except
that the surface must not be zinc coated nor galvanised after cleaning.
Bracelet anodes are the most commonly used type for protection of submarine
pipelines and for which their wrap-around construction is ideally suited (see
Section 8). They are rarely used on new offshore platform constructions, because
of their low current output to mass ratio compared with long slender anodes.
However, bracelet anodes do lend themselves to retro-fitting on existing structures
to supplement or replace the original failed, deficient or end-of-life cathodic
protection systems.
6.4.33 Anode utilisarion factor
Anode shape and cordinsert configuration have a major influence on the utilisation
factor of any particular anode design, This factor represents the maximum volume
of cast anode alloy which can be consumed before the anode can no longer deliver
the current required, and it has to take into account the reduced size of the anode
alloy and disbondment of anode alloy from the core at the end of life.
Cathodic protection of offshore structures
113
Careful attention to detail in the design of the anode and its cordinserts should
enable the following utilisation factors to be realised:
long slender stand-off type : 0.9 to 0.95
.
.
bracelet type
: 0.8 to 0.85
plate type
: 0.75 to 0.85.
6.4.3.4 Weight of anode alloy required
Four basic design parameters are required to be known before the total weight of
anode alloy to protect a structure can be determined. These are :
total current based on mean cathode current density requirements
.
I
Ahkg capacity of the chosen anode alloy
utilisation factor for the chosen anode design
required design life of the structure.
Substituting these parameters into the foilowing formula provides the nett weight of
anode alloy required:
alloy weight
total current x design life in hours
-- Ah/kg
capacity x utifisation factor
6.4.35 Anode resistance
As indicated earlier, anode geometry has significant affect on anode current output.
This is because the anode to electrolyte resistance is a function of the anode
geometry rather than being directly proportional to the surface area of the anode
exposed to the electrolyte. Several theoretically derived and empirically derived
formulae for calculating anode to electrolyte resistance have been propounded, the
most commonly accepted ones being shown in the Appendix (see also Section 5).
6.4.3.6 Anode current output
Anode current output is determined using Ohms law as follows:
I = EIR
where I = anode current output in amps
E = anode to cathode closed circuit potential difference in volts
(electrochemical poten tia 1)
R, = anode to electrolyte resistance in ohms
Strictly, the total resistance of the electrical circuit should be considered in
calculating anode current output, I. However, for offshore structures, the resistance
of the structure can be assumed to be negligible, and it is therefore ignored. The
resistance of the electrolyte (sea water) is considered in calculating the value of the
anode to electrolyte resistance. Finally, the polarisation of the structure (effectively
the structure to electrolyte resistance) is allowed for in the calculation by using the
protection potential rather than the corrosion potential in calculating E (see also
Section 2).
To simplify the calculations. and to ensure that a consistent approach is taken when
calculating anode current output, the driving potential, €, is conventionally taken
114
MTD Ltd Publication 901102
to be the potential difference between the anode closed circuit potential and the
maximum positive protected potential of the steel cathode in the applicable
environment as shown in Table 6. The minimum cathode current density values
detailed in Table 5 are similarly based on polarising the steel to these same
maximum positive protected potentials.
Anode current output has to be determined for the new as-cast condition and for
the end of life condition in order to ensure that the anode design meets the initial
and final cathode current demand requirements.
6.4.3.7 Anode distribution and quantity
A sufficient quantity of anodes need to be deployed to provide adequate
distribution of anode current to meet the cathode current demand requirements,
and thus to maintain the desired level of polarisation potentials at all times.
The relatively Iow current output of sacrificial anodes usually ensures that, for most
structures, adequate distribution of anode current can be obtained by locating
anodes on structural members by simple numerical techniques. This is normally
accomplished by locating sufficient anodes on each structural member so that the
available anode output matches or exceeds the cathode current demand of the
member at all times. Anode spacing should be sufficient to ensure an even
distribution of current and to prevent mutual interference effects which would
materially reduce the anode output current. A greater concentration of anodes
around structural nodes is usually necessary to overcome shielding effects.
For items such as conductors, which are installed after installation of the main
structure, protection is normally provided by anodes mounted on the permanent
structural members located nearest to the conductors (i.e. conductor guide frames).
However, on structures with a large vertical separation between these frames or on
tension leg platforms which employ no intermediate guide frames between the
platform and sea bed, t h e use of supplementary coatings applied to the conductors
may b e necessary in order to maintain acceptable levels of protected potentials.
On more complex structures (particularly around complex structural nodes),
mathematical modelling techniques are beneficial (see Section 5 ) . If inadequate
current distribution cannot be overcome, the use of supplementary high
performance protective coatings should be considered.
Similarly, the assessment of adequate levels of polarisation on wellhead conductors
is a common problem, because of the mass of conductors bundled closely together
and which have to be protected by anodes mounted remotely on the platform
structural members. The application of a high-performance coating system to the
conductors minimises current drained by them, and it improves current
distribution,
Cathodic protection of offshore structures
115
The total quantity of anodes to be installed should ensure that the following criteria
are met:
the total net weight of anode alloy installed equals or exceeds the minimum
calculated requirement based on mean cathode current densities
the total initial current available from the installed anodes equals or exceeds the
total inifial cathode current required
the total final current available from the installed anodes equals or exceeds the
total final cathode current required
6.4.4 Impressed current anode systems
6.4.4.1 G e n e d
Impressed current systems need to be designed to satisfy statutory electrical and
hazardous area requirements. Because operating voltages are large compared to
those available with sacrificial anodes, fewer, larger capacity anodes are utilised.
For jacket protection, sacrificial anodes are designed with current outputs typically
in the range 1 to 5 A, whereas corresponding impressed current anodes are
designed with typical current outputs in the range 10 to 100 A. System
performance is thus generally less susceptible to changes in electrolyte than is the
case for sacrificial anodes.
An impressed current cathodic protection system is an active system requiring an
external source of power incorporating the following major components:
anodes which transmit the direct current from the transformer rectifier into the
electrolyte and thence to the structure to be protected
a transformer rectifier which acts as a source of controllable direct current
reference electrodes which measure the structure potential and control the output
of the transformer rectifier
power cabling which carries the direct current from the transformer rectifier to
the anodes
I signal cabling which transmits potential signals from the reference electrodes to
the transformer rectifier
cable routeing components which facilitate the safe passage of cables between
anodes, electrodes and the transformer rectifier.
The selection of cathode design parameters for an impressed current cathodic
protection system is identical to that for a sacrificial anode system.
Depending on anode material, impressed current anodes operate at relatively high
voltages, typically 8 to SO V, and high output currents, typically 10 to 100 A. At
these output levels, the hazard to divers posed by electric shock can be significant.
The system design has to take this into account, preferably by the localised deenergising of the anodes in areas of diver activity. This usually necessitates t h e
installation of supplementary sacrificial anodes at critical structural nodes to
prevent their depolarisation when a nearby impressed current anode is temporarily
de-energised.
116
MTD Ltd Publication 90/102
Another significant consequence of the relatively high current output of impressed
current anodes is that highly negative potentials can be induced on the protected
structures by incorrectly designed systems, particularly in areas immediately
adjacent to the anodes. This can lead to the highly detrimental effects of hydrogen
embrittlement of the steel cathode and cathodic disbondment of protective
coatings.
In order to avoid hydrogen embrittlement and cathodic disbondment problems, it is
essential for anodes to be physically separated from the protected structure by
cantilevering them away from the structural members on which they are mounted,
or by locating flush-mounted anodes in the centre of a dielectric shield.
The problem of determining the dimensions of physical separation requires
particular attention. Until recent years, relatively simple empirically derived
formulae of questionable accuracy were frequently relied upon to do this, such as
those referenced in CP1021c72). However, with the advent of powerful computing
facilities, much greater accuracy can be obtained at reasonable cost and in an
acceptable time frame by the use of mathematical modellin methods. This was
demonstrated in the work carried out €or the Hutton TLP(6$, and it is
recommended that such computer modelling techniques be used for this task in
future.
Further applications for mathematical modelling techniques are the determination
of anode to electrolyte resistances and the relation of anode current distribution to
cathode polarisation potentials. Indeed, the use of this design tool for these tasks is
increasingly becoming recognised as an essential requirement for the design of
successful offshore impressed current systems, and it is highly recommended(73).
Section 5 gives a more detailed discussion ofthis subject.
6.4.4.2 Anode design
Essential requirements for the anode design are as follows:
#
Anode material has to be sufficient to meet the design life requirements based on
proven anode wear rates.
Anode design has to possess a sufficiently low electrical resistance in the
electrolyte to emit the maximum design. current without exceeding allowable
anode voltages.
At maximum design current output, the allowable anode current density should
not be exceeded.
#
At minimum design current output, the anode current density exceeds the
minimum value required to prevent the anode material passivating (e.g. lead
anodes).
Throughout the designed current output range, the anode does not induce
excessively negative cathode potentials which may cause damage to the cathode
or cathodic disbondment of protective coatings.
Cathodic protection of offshore structures
117
Anodes can be installed in appropriate locations to provide efficient distribution
of current to maintain complete polarisation of all cathode surfaces, and to
minimise damage caused by falling objects, dragging lines, etc.
The design employs simple reliable construction techniques to permit partial
replacement of critical parts of the entire assembly by divers.
6.4.43 Anode wuateriak
Materials used for general seawater applications include the following:
piatinised titanium
platinised niobium
niobium wound with platinum wire
I
lead-silver-an timon y
lead with platinum bielectrodes
I
high silicon iron-chrome
I
magnetite.
Of these materials, only platinised titanium and platinised niobium have found
general acceptance as suitable anode materials for offshore applications. Properties
possessed by these materials (and which make them the favoured candidates for the
highly demanding operating conditions offshore) can be summarised as follows:
low rate of corrosion
low anode polarisation
good electrical conductivity
high reliability
high mechanical strength
good resistance to abrasion and erosion
readily fabricated into useful forms.
The one significant disadvantage of platinised anodes, particularly platinised
niobium, is their high unit cost relative to most other candidate materials.
However, this is Iargely offset by their ability to operate at high current densities,
typically 500 A/m2 of platinised surface, thereby minimising their physical size and
hence material costs for a given current output.
The production of precious metal anodes requires specialised manufacturing
equipment and techniques, and it demands the implementation of stringent quality
assurance and quality control standards to ensure that the predicted in-service
performance is achieved. Supply of anodes manufactured by companies without a
satisfactory long-term track record in this field should be viewed with considerable
caution.
t 18
MTD Ltd Publication 90/102
Typical performance characteristics of anode materials are given in Table 11.
Materials used in the manufacture of anode assemblies, including dielectric shields,
should not suffer a deterimental Ioss in their physical, mechanical or electrical
properties when subjected to permanent immersion in sea water or when exposed
to gaseous chlorine which is evolved at the active surfaces of the anode.
Furthermore, dialectric shield materials need to possess excellent resistance to
cathodic disbondment when subjected to the maximum negative structure to
electrolyte potential predicted for the specific application.
TABLE 11 IMPRESSED CURRENT ANODE PERFORMANCE
CHARACTERISTICS
Anode
material
Typical consumption
wear rate (UA-a)
Typical design
Typical anode
operating voltage (V) current density
Wm2)
Pt-Ti
I
Pt-Ni
500
8
1
1
1000
0.008
1
0.008
Pb-Ag-Sb
150
90
Pb-Pt
500
90
75
500
Si-Fe-Cr
* Voltage limit governed by statutorylsafety constraints
6.5
ENGINEERING
6.5.1 General
The engineering of cathodic protection systems applied to offshore structures needs
to consider the severe marine environments involved and the large and inaccessible
surface areas to be protected, as well as tbe effectof safety regulations and other
statutory limitations on the power sources, cable conductors, and instruments
installed on the operating platform.
Because the greater part of the structure is immersed on installation, anodes, cables
and monitoring devices have to be installed during or immediately after structure
fabrication. These components need to be engineered to withstand the handling
and environmental forces resulting from transportation from the fabrication yard,
and installation and operation of the offshore location (see also Section 10).
Damage to any part of the installation during structure fabrication or instahtion is
unacceptable, because loss of anodes or monitoring devices may impair the
structure corrosion protection or corrison monitoring systems.
Particular consideration has to be given to cables for impressed current anodes and
monitoring systems which are fixed to the structure before its final installation, so
Cathodic protection of offshore structures
119
that they are available for connection to the platform power supplies and
instrumentation.
Electrical equipment also has to be protected from damage at the dockside, during
transportation and installation on the platform, as well as from the marine
atmosphere during service.
6.5.2 04iectives
Engineering of the cathodic protection system is the work of converting the
theoretical design requirements into a fully detailed, paractical working system.
Engineering documents to be produced include comprehensive specifications and
drawings which fully define requirements for in-service performance, material
properties, quality assurance and control, dimensional details, etc.
Essential information required to perform the engineering work will have been
determined during the design phase. It includes the following:
type of system required (sacrificial, impressed current or hybrid)
.
.
.
.
.
design life of system
total current required to protect the structure, based on initial, mean and final
current density values
current required to protect each individual structural, item based on initial, mean
and final current density values
general arrangement and detailed drawings of the structure, showing optimised
locations of all anodes, and, if required, reference electrodes
anode quantity, material and, for sacrificial anodes, net and gross weights
.
areas covered by splashzone coatings and supplementary coatings, and the
cathodic disbondment characteristics of each
extent of use of the various types of metallic construction materials.
Additional necessary information, specific to the engineering of impressed
currenthybrid systems, includes the following:
means of powering the impressed current system (i.e. platform ac power supply,
solar energy or other means)
maximum continuous power available from the platform ac power supply in kW,
and characteristics of the supply in terms of voltage, frequency, single- andor
three-phase
predicted reliabiIity of the platform ac power supply
planned availability of the platform ac power supply before and after installation
of the structure
available locations for siting the system power source (e.g. transformer-rectifiers,
solar energy arrays, and available cahle routes for anodes and reference
electrodes)
120
MTD Ltd Publication 901102
hazardous area classifications (if any) where the system power source uoit(s) and
ancillaries are to be located and through which cables will be routed
maximum heat dissipation and ventilation requirements of t h e system power
source unit(s) and ancillaries.
.
6.5.3 Sacrificial anode systems
6.53.1 INinuanon wnsidemtbns
The total quantity of anodes required, and their distribution around the structure,
are determined by the designer (see Section 6.4.3). An essential requirement for all
sacrificial anode types is that they are securely fixed to the structure via very low
resistance electrical connections. Furthermore, the anodes, their W n g s and fming
methods should not induce unacceptabIe levels of stress in the structural members
to which they are attached. Anode attachments to structures which are to be fixed
to the sea bed using driven piles, and where the pile driving operations induce
considerable vibration in the structure, require particular attention.
Loss of anodes with resulting damage to the structure has occurred on a number of
platforms as a result of excessive pile driving forces experienced during installation.
To minimise the risk of damage to the structure, it is recommended that massive
stand-of€anodes should be welded in place via doubler plates. A more detailed
discussion of the structural implications of anode installation is given in Section 10.
Minor adjustments to the location or geometry of individual anodes may be
necessary to take into account the following:
maximum anode weight loading on any structural element
.
.
avoidance of welding anodes on, or too close to, structural welds
.
.
avoidance of specific structural items of ancillary equipment
to ease future ROV or diving activities.
The extent to which such adjustments can safely be made depends on the details of
each individual system design. However, any adjustments should always remain
within the design tolerances for the system.
Wherever possible, all anodes on a given structure shouId be of a common design.
The use of anodes of differing size and mass on a single structure Can rarely be
justified in economic or technical terms, and it may lead to the anodes being
incorrectly located on the structure. Specific exceptions to this general rule are :
anodes required to be Iocated on mud mats, which frequently have to be flat
plate type rather than the massive stand-of€type most commonly used for the
main structure
anodes Iocated around complex node structures designed to overcome
"shadowing" effects and which by necessity are required to be compact because
of space limitations
.
Cathodic protection of offshore structures
121
b
anodes for areas employing supplementary coatings, which are required to exhibit
output current characteristics substantially different from those protecting bare
areas
anodes located on risers.
.
6.5.3.2 Anode JpeciJication
The anode specification, based on the system design (see Section 6.4.3), should
comprehensively define the performance required of the anodes under all
operational conditions, As a minimum requirement, the specification should detail
the following:
design Iife of the system
applicable British or other internationally recognised standards or codes of
practice governing the design and manufacture of materials and equipment
environmental conditions at the offshore site such as electrolyte type (sea water,
mud); electrolyte temperature and resistivity range; known type and
concentration of any pollutants present in the electrolyte
anode alloy type required (aluminium, zinc, magnesium)
operating temperature of the anode, if different from the electrolyte temperature
range (e.g. anodes located on hot risers)
predicted range of current densities within which the anodes are required to
operate, and A h k g capacity required of the anode alloy over this range
closed circuit potential, wrt a silverhilver chloride half cell, required of the anode
to alloy when operating over the predicted range of operational current densities
net and gross anode weights and dimensions, and anode current rating
.
.
anode insert (core) and doubler plate material type and grade in accordance with
an internationally recognised standard (e.g BSI, API, ASTM, etc.), stating any
supplementary requirements for chemical composition, mechanical properties,
weldability, etc.
0
anode insert surface preparation prior to coating
6
packing and shipping requirement
quality assurance and quality control requirements in terms of certification for
anode requirements in terms of Certification for anode inserts and doubler plates,
and manufacture of the anodes.
Specific requirements for the following should be included:
alloy compositional control by spectrographic analysis of all heats, stating
frequency of analysis throughout casting
control of casting defects such as cracks, laps, shuts, void, porosity, bowing,
twisting, shrinkage, etc., including permitted tolerances for each
dimensional and weight tolerances
.
.
.
6
122
welder and welding qualification requirements where applicable
MTD Ltd Publication 90/102
laboratory testing of production alloy samples to prove electrochemical properties
correct location of anode insert within the anode alloy, including destructive tests
to prove compliance with specified requirements
Additional information required of the cathodic protection contractor in order to
properly assess the acceptability of his proposals shouId include the following:
successful track record of undertaking similar projects
.
documentary evidence, including long-term test or operational data,
demonstrating that the equipment offered complies fully with the project
specification
a project-dedicated quality plan with corporate quality manual covering all
equipmeo t to be supplied
qudity documents showing that quality assurance and quality control activities
are controlled by a dedicated quality management structure functioning
independently from the production management structure.
It is preferable that the cathodic protection contractors' quality system should be
assessed and approved as complying with a recognised internationaf standard for
quality systems, for example the relevant parts of BS 5750(74).
6.5.4 Impressed current systems
As discussed in Section 6.3.5, the term "impressed current" can be misleading,
because most such systems are generally "hybrid" systems. Engineering of the
sacrificial anode element of these systems is covered in Section 6.5.3. The
impressed current elements are covered in tbe following Sections.
6.5.4.1 Installalion considerations
Similar considerations apply regarding structural and mechanical integrity of the
impressed current equipment to those discussed in Section 6.5.3.1 for sacrificial
anodes. However, the greater complexity of impressed current systems has more
far-reaching consequences than that encountered in sacrificial systems, one of the
most significant being the routeing of cables feeding the anodes and reference
electrodes. A soIution to this problem, attractive to the cathodic protection
engineer, may be routeing cabies within the members of the structure. For buoyant
structures, this is logical and relatively easy, because of the cavernous construction
of most (if not all) parts of the vessel hull. However, for a jacket structure it may
be impossible because of the small diameter of many of the tubulars, particularly
horizontal and diagond brace members. Where diameters are sufficiently large, the
serious structural implications of providing through-wall penetration of tubulars to
allow passage of cables and their connection to anodes need to taken into account.
Cathodic protection of offshore structures
123
Additional problems requiring resolution if cables are routed within structural
members are :
through-wall penetrations carry an inherent risk of water leakage and consequent
flooding, leading to corrosion damage of structural members
cables so routed are probably entirely inaccessible for repair or replacement
during the operational life of the platform.
.
The utmost care has to taken to avoid these problems, particularly those of the
water sealing arrangements of structural penetrations. Electrical circuit
penetrations should pass through fully pressure-rated steel cofferdams employing
water-tight cable glands. To ensure water-tightness of the penetration, it may be
necessary to fill the cofferdam with an inert, water-proof, sealing compound.
The most common means of routeing cables to anodes (and reference electrodes)
on jacket structures is to enclose them in heavy duty pipe conduits which are
welded or bolted to the exterior surfaces of the structural members. Cable conduits
should always be located in-board of the jacket framing plan, and where possible
on tbe underside of the structural members, in order to minimise risk of
mechanical damage during fabrication, installation, and operation of the structure.
The conduits should be stood-off a sufficient distance from the structure surface to
allow inspection of the members under them, and they should be robustly designed
and fabricated as appurtenant structural members in their own right, capable of
withstanding, without damage, the severest conditions envisaged for the offshore
location. In the event of seawater leakage into metallic pipe conduits, provision
should be made for adequate corrosion protection measures consistent with
achieving the required design life of the conduit.
To facilitate replacement of defective anodes, cables should terminate in pressurerated couplings or connectors which can be easily disconnected by divers, and they
should be constructed of materials inherently resistant to corrosion in sea water at
the maximum voltage available at the termination.
Anodes mounted close to, or flush with, the structure should be mounted on
prefabricated dielectric shields. A material successfully used for this type of
dielectric shield and its fixings is hot-pressed epoxy resin impregnated glass fibre.
This type of dielectric shield usually needs to be bedded onto a secondary
dielectric shield applied in the form of a high build solvent-free coating direct to
the structure. The dielectric coating should completely cover and extend beyond
the structure surfaces under the prefabricated dielectric shield, and it should
prevent the ingress of water behind the prefabricated dielectric shield.
Dielectric shields for cantilevered anodes are applied to the structural steeiwork
supporting the anode assembly and to adjacent structural members, usually in the
form of a high-build, solvent-free coating similar to the dielectric coating described
above. To improve its resistance to mechanical damage, these coatings may be
reinforced with glass flakes or fibre glass mat.
Surface preparation of steelwork to receive non-prefabricated dielectric shields has
to be carried out to very high standards. This normally entails degreasing and
124
MTD Ltd Publication 90/102
rinsing the surfaces prior to abrasive blast cleaning, using chilled iron angular grit
to obtain a degree of cleanliness equal to grade Sa 3 to SIS 05 59 00.The resulting
peak-to-trough surface profile obtained should be in accordance with the
recommendations of the manufacturer of the dielectric material.
At installation, all dielectric shields should be thoroughly inspected to ensure they
are undamaged, free from discontinuities, and satisfactorily bonded to the structure.
65.4.2 Equipment specification
The principal components of an impressed current system are described in general
terms in Section 6.4.4. The specification governing these component parts should
comprehensively define the performance required of each under all operational
conditions. As a minimum, the specification should cover the following:
General
-design life of the system
- applicable British or other internationally recognised standards or codes
governing the design and manufacture of materials and equipment
- environmental conditions as defined in Section 6.5.3.2) supplemented by
information relevant to the rating of electrical equipment and its materials of
construction.
Anodes
- anode assembly description, including materials of construction for active anode
surfaces and suhstrates, anode carrierhousing, connectorskouplings in supply
circuits, h n g s , dielectric shields, ancillary items
- anode current and voltage ratings
- anode assembly sketches defining minimudmaximum dimensional and weight
limits.
Transformer-rectifier equipment
- ac power supply information, including number of phases, voltage, frequency
and whether 3- or 4-wire feed, and, if applicabIe, the maximum kVA and k W
power consumption available
-hazardous area classification of each location, if applicable
- degree of environmental protection to be afforded by tbe cubicle, referenced to
BS 5490(”), and the cooling medium for the equipment
- output voltage and current rating of each transformer-rectifier installation, and,
if comprising a number of modular units, the output voltage and current rating
of each module
-maximum R M S ripple content permitted in the dc output to anodes
-number and rating of anodes fed from each transformer-rectifier installation and
each modular unit
- operating range from zero to full rated output required, taking into account
back emf of the anode material
- method of regulating output current to be automatic control, governed by
structure potentials as monitored by the reference electrodes, with manual
control back-up - rectifier stack materials (e.g. thyristors, silicon diodes,
selenium, etc.)
- electrical protection requirements for ac input, dc output, rectifier elements,
transformer secondary circuit, etc.
-current and thermal overload protection facilities to be provided
Cathodic protection of offshore structures
125
-the number of reference electrodes controlling the output of each transformer
rectifier module
- monitoring and control circuits to detect and reject spurious reference electrode
signals
- monitoring and control circuits to detect faults in anode circuits
-facilities for metering of total and individual anode output currents, anode
circuit voltages, and reference electrode Potentials
-sensitivity of the automatic control circuitry to maintain structure potentials at
the preset protection level
- data logging facilities for monitoring critical operational parameters of the
system (e.g. reference electrode potentials, anode circuit current and voltage,
operation of protection devices, ac input voltage, etc.)
- local and remote alarm facilities triggered by operation of protection devices or
mal-operation of the equipment detected by the monitoring circuits
- termination requirements for all incoming cables
- labelling of instruments and controls
- protective plating and coating requirements for equipment and its component
parts
-spares required, particularly fuses.
Reference electrodes (see also Section 9)
- reference electrode assembly description, including materials of construction for
the reference cells, housing, connectordcouplings to monitoring circuits, fixing,
ancillary items
- reference electrode sketches defining minimum/maximum dimensional and
weight limits
- current drain limits from the electrode to ensure they do not transmit spurious
signals as a result of passivation or polarisation.
Cabling
Note :The detailed specifications for cabling and its ancillaries are normally
defined within the bulk cable order for the whole project. However, the
engineer should ensure that cables for the cathodic protection system
adhere to the following requirements:
- cable volt-drops do not exceed the maximum value as determined by the
cathodic protection engineer in order to ensure satisfactory operation of the
system
- all cabling should be steel wire armoured, and reference electrode and control
circuit cables have to be adequately screened
- reference electrode and control circuit cables should be segregated from anode
and other power cables
- cables and ancillaries subject to immersion or installation in hazardous locations
need to be suitably rated and approved for these environments. Wherever
possible, jointing of discontinuous cable lengths in these environments should be
avoided. Where this is not possible, jointing materials have to totally
encapsulate the conductor(s) and armouring to provide complete water tight and
inert protection to them.
MTD Ltd Publication 90/102
Structure cathodic circuits
The connection between the power source negative pole and the structural steel
work should be designed and installed with adequate current carrying capacity.
Multiple connections should be made between the negative pole and structural
members, to limit current amphtude in individual cables and members. Cables
should be terminated on substantial fixings such as threaded terminals securing
cable lugs, which then need to be protected by waterproof coatings.
Consideration should be given to area safety classification when locating
cabldstructure connecting points.
.
Electrical isolation
The design of the cathodic protection scheme is based either on isolation of the
structure from pipeline risers or on their continuity. When isolation is required,
the specification of the isolating joint has to reflect the electrical requirements of
the cathodic protection design, the mechanical requirements of the pipeline
design, and the environmental conditions.
ElectricaI isolation may be achieved by providing insulating gaskets, bolt sIeeves
and washers to a normal flanged coupling, or by installing a prefabricated
monolithic isolating joint. In either case, the units need to be located as high
above the waterline as possible, and they have to be adequately coated for
corrosion protection. Test facilities should be provided to both sides of the joint
at a location suitable for testing the effectiveness of the insulations.
Electrically isolating joints should be the subject ofa separate detailed
specification.
.
Quality assurance and control
The following aspects should be covered :
- certification for specific materials of construction or equipment to show
compliance with specified standards or codes
- dimensional and weight tolerance requirements of all major components
- comprehensive works testing and as-installed testing of transformer-rectifier
equipment. These tests should include full fictional and heat-run testing,
including simulation of fault conditions to ensure satisfactory operation of fault
detection and alarm systems, and the data logger
- calibration of reference electrodes
- quality assurance and quality control documentation for anodes as described in
Section 6.5.3.2.
Cathodic protection of offshore structures
127
6.6
OPERATING MANUAL
The cathodic protection system should be provided with an operating manual. The
importance of this manual for monitoring and surveying the system is discussed in
Section 9. The manual should provide the following information :
.
the parameters used in designing the system.
.
installation details
fault-finding procedures
maintenance procedures.
The above topics may be set in greater detail as follows :
design parameters
-structure surface areas in sea water
- structure surface areas below mudline
- drill casing surface areas
- riser surface areas
-temperature, environmental and at risers
-water resistivity
-water current,velocity and direction
- design current density, including allowances for piles and wells
- design life of anodes, power source and monitoring devices
- coatings used in the cathodically protected zone, particularly at odes and
risers and the current density values used
- design current output of anodes, maximum anode output of impressed current
anodes
- maximum power source current and voltage
- circuit resistances for each impressed current anode
anode current and voltage operations limits
- monitoring system details.
-
.-
installation details
drawings and schematics of all installations.
- materials used for:cables, anodes and reference cells.
.
fault finding procedures
-chart to guide technical and non-technical staff in the location and correction of
faults.
maintenance procedures
- provided by each manufacturer for their equipment, which should include :
operation a1 limitations, lubrication requirements, painting schedules and
calibration procedures. The manufacturer’s own documents should be bound
into the manual.
MTD Ltd Publication %/lo2
Section 7 Cathodic protection systems
for concrete offshore structures
Cathodic protection of offshore structures
129
7. Cathodic protection systems for concrete offshore
structures
7.1
INTRODUCTION
Unlike steel offshore structures, which are exposed to the various zones (under
water, tidal, splash, atmosphere) of the marine environment, the steel in a concrete
offshore structure is exposed to a "concrete" environment, which, in, turn, is
modified by the conditions to which it is exposed. Concrete can be regarded as an
aqueous environment in which the resistivity is relatively high (say lo3 to 106 ohm
cm, compared with 30 ohm cm for sea water) and the diffusion of gases and ions is
severely restricted.
On a bottom-mounted concrete offshore structure standing above sea level, &he
concrete forms a continuous electrolyte with varying properties spanning several
environmental zones. The way in which all these factors influence the corrosion of
reinforcing or pre-stressing steel in the structure, and the way in which this
steelwork interacts with otber steel components on the complete installation have
to be considered before the performance of any cathodic protection system can be
discussed. This Section therefore describes the special features which characterise
the electrochemical corrosion of steel embedded in concrete, and the influence of
various environments, before turning to the current state of knowledge about
protection criteria and the installation and monitoring of cathodic protection for
steel in concrete structures. Operational experience on such structures in the
North Sea is then discussed. At the present time, cathodic protection is applied
only to the "barel' steel components attached to underwater parts of concrete
structures, although electrical continuity (either deliberate or accidental) between
the "bare" steel and the reinforcement can allow some of the protection current to
be diverted to the Iatter. The influence of existing cathodic protection systems is
confined Iargely to reinforcement in the underwaterltidal zones of the structure, but
the possible need to develop impressed current cathodic protection for the
splasWatmospheric zones is also considered.
7.2
"HE CONCRETE ENVIRONMENT
The chemical and physical properties of concrete are determined by the properties
and proportions of the materials of manufacture, the way in which the concrete is
made and cured, and the way in which it is subsequently altered by reaction with
its environment. The principal components of concrete are usually Portland
cement, coarse and fine aggregates, and water. Concrete can also be made with
part of the Portland cement content repIaced by other cementitious materials such
as blast furnace sIag, or pozzolanas (reactive silica) such as pulverised fuel ash.
Often, small amounts of other additives are used to facilitate the mixing and
placing of the concrete andor to improve its final properties. However, the basic
features of the structure and the chemical properties of the concrete remain very
much the same.
Cathodic protection of offshore structures
131
Offshore structures in the North Sea are made to very high standards with concrete
of good overall "quality", including careful control to ensure correct thickness of
cover to the reinforcing steel, correct mix proportions, good compaction, and
proper curing procedures. Such concrete represents the best which can be
achieved economically to ensure adequate durability in a large reinforced concrete
structure exposed to a severe marine environment.
7.2.1 Chemical properties
Concrete sets within a few hours of mixing, but it continues to gain strength over
weeks (or even years) as chemical reaction between the cement and water
continues. The principal phases of ordinary Portland cement (OPC) are tricalcium
siIicate, 3CaO.Si0, (usually abbreviated in concrete terminology to C,S) dicalcium
silicate (C,S); tricalcium aluminate, 3Ca0.A1,03 (C3A) and an aluminoferrite,
4Ca0.A1,0,.F+03 (C,AF). On hydration, the silicate phases, which make up 70
to 80% of the cement, form a poorly crystalline microporous calcium silicate
hydrate gel plus crystalline calcium hydroxide. These, together with the hydration
products of the other components, make up the cement paste matrix. Concrete
always retains an aqueous phase which at first is very strongly alkaline (typically
pH 13 to 14) as a consequence of a small alkali metal content in the cement,
although it is likely that processes such as leaching eventually reduce the pH
somewhat, to that of a saturated calcium hydroxide solution (i.e. pH about 12.6).
The relatively large quantity of solid Ca(OH), produced by hydration of the cement
effectively buffers the pH at this value, but several other chemical changes continue
in concrete throughout its life, depending on the environment and the extent to
which the environment can penetrate. Three of these may influence corrosion
significantly.
7.2.1.1 Carbonation
The reaction of the aIkaline pore solution with acid gases diffusing into the
concrete can eventually neutralise the calcium hydroxide and allow the pH to fall.
This is known as carhonation, because the gas primarily involved is carbon dioxide.
Carbonation can occur locally along cracks, but more generaIly its influence
penetrates only slowly into the concrete, forming a "front" which is readily detected
by the colour change of the indicator phenolphthalein, As is discussed later,
penetration of carbonation to reinforcement depth can result in corrosion, but
carbonation is usually a very slow process in immersed or very wet concrete, and is
unlikely to affect the concrete at reinforcement depth.
7.2.1.2 Ingress of chloride
Sulphate and chloride from sea water diffuse into concrete, and both can react with
any residual C,A phase. Sulphate ions react to form calcium sulphoaluminate
hydrates which occupy a very much larger volume than the reactants, and this
could disrupt concrete with a high C3A content. Consequently, sulphate-resisting
Portland cemeat (SRPC), which has a very low C,A content, is often specified for
concrete likely to be exposed to environments high in soluble sulphates. Although
there is n o evidence that the small amount of sulphate in sea water warrants the use
of SRPC, cements with a very high C,A content are generaliy avoided for concretes
exposed to marine environments. Chloride ions react with C,A to form calcium
132
MTD Ltd Publication 90/102
chloraluminate hydrates, but these do not damage the concrete structure. Indeed,
C3A is generally considered to be beneficial in reinforced concrete, because it is
believed to immobilise chloride ions which might otherwise cause corrosion.
Although there is little actual evidence that the formation of chloroaluminates
significantly retards the ingress of free chloride ions into hardened concrete, an
OPC with an intermediate C3A content seems to be widely preferred for concrete
exposed to sea water.
7.2.13 Formation of surface deposik
In sea water, alkali leached from concrete reacts with dissolved magnesium and
calcium ions,and this may form a surface deposit of brucite (magnesium
hydroxide) and aragonite (calcium carbonate). Seawater conditions (e.g. flow rate,
temperature, micro or macro biofouling) can influence deposition, but if a surface
"skin" forms, it may be less permeable than the concrete itself and may therefore
influence the overall permeability of the cover to the steel.
7.2.2 Physical properties
Concrete contains pores and voids wbich vary greatly in size, possibly ranging from
large voids resulting from incomplete compaction to minute pores within the
bydrated cement gel. Tbe size of pores and the interlinking of "capillary" pores in
the cement paste depend on the waterkement (wk) ratio of the original mix and
the subsequent long-term curing, during which continued hydration may reduce
and segment tbe pore structure. A typical pore size distribution for OPC pastes is
illustrated in Figure 35. There should be few voids resulting h o m incomplete
Metal-
I
ioo
Concrete
Ioist
air
io
Pore diameter ( nm 1
Figure 35
Figure 36
Typical pore size distribution in
cement paste (volume intruded by
mercury under increasing pressure)
Schematic of pore system in concrete
Cathodic protection of offshore structures
133
compaction or bleeding (separation of water and solids before the concrete has set),
but a small number of voids up to several millimetres in diameter can be expected,
even in a good quality concrete.
Physical properties of the concrete which are most important in corrosion are given
below.
7.2.2 .I Permeability
This is not a simple function ofporosity, for the size and continuity of the
interconnecting pores affects the rate of penetration of moisture, gases and
dissolved species in different ways. For example, in the pore system illustrated(76)
diagrammatically in Figure 36, air-filled pores act as barriers to ion diffusion, hut
nearly all the resistance to oxygen transport is in the water-filIed capillary system
(Figure 37). Typical diffusion coefficients for chloride ions in saturated cement
cm2/s, increasing in an approximately
pastes lie in the range 5 x lo-* to
exponential manner with increasing w/c ratio and temperature. Diffusion
coefficients are also markedly higher in pastes cured in air compared with similar
pastes cured under water. Page et alo7) reported a diffusion coefficient of
lom7
cm2/s at 25°C for an OPC paste of w/c 0.5 cured in saturated air, compared
with 4.5 x
for a paste ofthe same w/c ratio cured in saturated Ca(OH),
solution.
Metal I
I Moist
Concrete
air
c
m
L
Figure 37
Effective oxygen concentration profile
through concrete cover, showing
relative elfects of water-filled and airfilled porosity (after Tuutti (7ej)
Waterfilled
filled
7.2.2.2 Penetrability
Transport through concrete is generally greater than can be accounted for by
permeability of the cement matrix, because of microcracking, and perhaps coarser
fissures because of incomplete compaction. In concrete exposed to alternate
wetting and drying (as for example in the upper parts of a marine structure), t h e
rate of penetration of ions can exceed that expected from diffusion, at least near the
surface, as a result of intermittent flow of bulk solution into partly dried concrete.
It was shown that a chloride Concentration of 0.06% by weight of concrete
(approximately 0.32% by weight of cement) was reached at a depth of 30 mm in a
good quality concrete after only 2.5 years exposure to splash zone conditions(7R).
More generally, it has been observed that chlorides penetrate the concrete of
marine structures to reinforcement depth within a few years in most
circumstances(79). The greater depth of cover, and the screening afforded by ducts,
should isolate prestressing tendons from the external environment very effectively,
provided the ducts are completely filled with cementitious grout.
134
MTD Ltd Publication 901102
722.3 Ekcmcal
- condrrcriviry
The conductivity of concrete is consistent with diffusion and ionic conduction
through liquid electrolyte in the interconnecting capillary pore system. The
presence of ions,such as chloride and its associated cations, increases the electrical
conductivity of the pore solution, but in practice the most important influence on
conductivity (more usually expressed as its reciprocal, resistivity, in ohmxm) is the
amount of water in the capillary pore network. Typical values range from a few
thousand ohm.cm in wet concrete with a fairly extensive interconnecting capillary
pore system, to many hundreds of thousand ohm.cm if the concrete is dry. Typical
resistivities for a high quality (70 MPa) concrete immersed in sea water range from
less than 7000 ohm.cm if air-cured to 23 000 ohm.cm if water cured(77). The
resistivity of sea water is around 30 ohm.cm,
7.2.2.4 lhznqwrt of oxygen
The rate at which oxygen penetrates through concrete may be the determining
factor in the corrosion of embedded steel, and it is certainly important in
determining the current demand of cathodically polarised steel in concrete. If the
samples have cut surfaces, both concrete and cement paste show apparent diffusion
coefficients for oxygen which may be('') several orders of magnitude greater than
those for ion transport, probably on account of gas-filled voids acting as "shortcircuit" paths for oxygen diffusion. Concrete specimens with cast faces show a
much lower rate of oxygen transport(B0) (Figure 38), and it is likely that in sea
water the barrier which controls oxygen trans ort lies in the modified outer surface.
Comparison of results from various sources(BR has shown that there is indeed a
fairly close relationship hetween concrete surface area and oxygen flux. Oxygea
transport is therefore best described as flux per unit concrete surface area rather
to 2 x
than by a bulk diffusion coefficient. Values range from about 3 x
mol Odcm2/s.
tfaces
Figure 30
Effecfof surface on transport of
oxygen through cement paste
(after Gjorv ef a/ @@)
Cathodic protection of offshore structures
0
20
60
Thickness ( mm 1
135
7.3
CORROSION OF STEEL IN CONCRETE
In strongly alkaline solutions, steel can develop a thin film of corrosion products
which, for all practical purposes, prevents further corrosion. Steel in this condition
is said to be "passive", and it is generally accepted that reinforcing steel becomes
passive as a result of alkaline conditions within the concrete. Reinforcement
corrosion can only become a problem when conditions lead to sigmficant
hreakdown of passivity on the embedded steel. In a well-made concrete, this only
occurs through the ingress of "depassivating species" from the environment. If
depassivation is delayed until these species reach the steel in sufficient
concentration, there is an induction period hefore corrosion starts. The subsequent
rate and form of corrosion depend on the depassivating species, other
environmental factors, and the properties of the concrete. In practice, the
depassivation which leads to significant corrosion is almost always the result of
either carbonation or to the presence of chloride ions.
Even widespread depassivation does not necessarily lead to serious corrosion if the
supply of oxygen is restricted, as is often the case for concrete which is permanently
under water. This highlights the fact that the corrosion of steel in concrete is an
electrochemical process, in which restriction of either the anodic or the cathodic
reaction can limit the rate of corrosion. It is therefore convenient to separate the
anodic and cathodic reactions, and to consider the way in which they combine to
produce the overall corrosion reactions.
7.3.1 The anodic reaction
A modified potential-pH diagram (Figure 39) broadly illustrates the conditions
under which corrosion may be expected to occur(82). It shows the increased
likelihood of corrosion as pH is reduced, and the depassivating effect of chloride
ions, even a t pH 12.5 and above. It also shows a region of corrosion which always
occurs at very negative potentials and high pH. In Figure 39 and elsewhere
throughout this Section, potentials are quoted with respect to a silver/silver chloride
electrode immersed in normal sea water (see Section 9.4.1 for a detailed discussion
of reference electrodes).
Under normal circumstances, penetration of a carbonation front into concrete is
SIOWand the depth of cover required by design codes should allow an adequate
margin to avoid depassivation of the steel during the design life of the structure.
Given the standards of quality and construction required of concrete offshore
structures and the very humid conditions to which they are generally exposed, it
should he possible to discount carbonation. Consideration of the effect of t h e
anodic of behaviour steel in an offshore structure can therefore be confined to
concrete with and without chloride ion contamination, as contrasted schematically
in Fignre 40.
136
MTD Ltd Publication 901102
*1000
+600
*200
-200
d
U
2 -600
3
\
>-
E -1000
4
ID
.-
Immunity
c
c
al
c
2 -1400
1
1
\
1
1
1
I
6
8
10
12
1
I&
Figure 39 Inthence of solution pH and potential (with and without presence of chloride
ions) on corrosion of steel (after Pourbaix (83))
*200
1
-100
-400
-
-700
d
U
P
\
0
a -loo(
>'
-.E
m
-
Immunity
c
C
c
al
2 -1 30(
Log current
[ a ) No chloride
-
Immunity
Log current
( b l Chloride present
Figure 40 Schematic of the effect of chloride on the anodic polarisation of sleet in
concrete
Cathodic protection of omhore structures
137
7.3.2 The cathodic reaction
The principal cathodic reaction which "fuels" the corrosion of steel in concrete is
oxygen reduction. The availability of oxygen at the concrete surface and its rate of
penetration through the concrete may be important in determining the rate of
reinforcement corrosion. It is certainly important in the behaviour of reinforcing
steel under cathodic polarisation, in other words when cathodic protection is
applied. A schematic cathodic polarisation curve is sbown in Figure 41. Hydrogen
is evolved on the reinforcement if it is polarised to a sufficiently negative potential.
0
-200
- 600
-600
-800
F
U
0
5
P
s- -1ooc
d
Q
.t
C
al
c
B -1200
Log cu went
Figure 41
138
-
Schematic of the effectof Iimited oxygen availability on the cathodic
polarisation of steel in concrete
MTD Ltd Publication 90/102
733 The composite reaction
The effect of oxygen availability on the behaviour of steel in concrete (ignoring
resistivity and without chloride ion contamination) is illustrated schematically in
Figure 42. For free corrosion to occur, the net rate of the anodic reaction has to
be electrically equivalent to the net rate of the cathodic reaction. This condition is
represented by the intersection of the anodic and cathodic curves in the
illustration. There is a transition from a stable passive condition (P) to a stable
non-passive (active) condition (A) when the availability of oxygen is greatly
reduced. Nevertheless, the corrosion represented by (A) is negligible. In
intermediate conditions, two stable potentials (P') and (A') are possible, but
impaired passivity at voids and other defects at the concretdsteet interface, tend to
favour development ofthe active condition represented by point (A'). Here the
corrosion rate is greater than at A. It can robably be assumed to be uniform, with
penetration not more than 1 pm per year($1 .
When oxygen is more plentiful, ingress of chloride ions (which is variable from
point to point), tends to break down passivity locally. This, and the development
of diffusion gradients in the concrete, favour pitting corrosion. Many attempts have
been made to define a critical chloride concentration below which corrosion does
occur, but it is now recognised that the threshold value depends as much on the
(variable) steeljconcrete interface conditions as on the actual chloride ion
As pitting develops, corrosion occurs on small areas of the
concentration
reinforcing steel, and the nearby passive steel becomes the cathode in a galvanic
"macro-cell",
In reinforced concrete exposed to the air, the size ofthe macro-cell will be limited
by the resistance path through the concrete (Figure 43(a)), and numerous pits may
develop. Underwater, the resistance path is short-circuited (Figure 43(b)), and it is
possible for a pit to draw upon a much larger cathodic area, so that pits tend to
occur less frequently. In theory, localised underwater corrosion can be very
intense, but usuaily the restricted rate of transport of oxygen from water much
reduces the effect of a given area of underwater cathode,
Cathodic protection of offshore structures
139
~300.
*loo.
-100.
-300,
-500,
-
-700,
d
U
<-? -900,
d
.-m
f
2 -1100.
,g current
-
Figure 42 Schematic of potentials of steel in concrete (free corrosion can on/y occur at a
rate corresponding to the current where curves intersect)
Air
Concrete (mode)rate
( a ) I n air(current f l o w restricted t o the concrete)
Sea water (very low
( b l I n sea water (current can flow through the sea water,
greatly affecting the cathodic area 1
Figure 43
140
Current flow through concrete in air and in sea water
MTD Ltd Publication 901102
7.3.4 Concrete in the sea
It is known(86to
that the reinforcement in concrete which is fully immersed in
hesh water or sea water can eventually reach potentials more negative than -800mV
AglAgCl, sometimes reaching values as low as -1000 mV Ag/AgCl, which
correspond to non-passive potentials in regions A or A' in Figure 42. This is
attributed to depletion of oxygen, and it is assumed that the steel is then corroding
at a very low, almost negligible, rate. When another part of the concrete is exposed
to air, the reinforcement in this region takes up a less negative potential, and a
partly-submerged concrete structure thus develops a large differen tjal oxygen
concentration cell.
As shown in Figure 43,the underwater area is not significantly restricted by
electrical resistance, but above water the resistance of the concrete limits the
effective cathodic area of the cell to a "galvanic interaction zone", probably
extending some way above the high-tide level. Oxygen may aIso be relatively freely
accessible to reinforcement near the inside concrete surface of air-filled legs on
some offshore structures. In this case, galvanic interaction is limited only by the
resistance of the concrete through the thickness of the "hollow leg".
The effects of differential oxygen concentration on a concrete structure in the sea
depend on a number of factors, such as concrete resistivity, ratio of immersed to
air-exposed concrete, and whether or not chloride ions have penetrated to the
reinforcement, either by diffusion or more directly through cracks in the concrete.
Furthermore, in most structures, there is some steel directly exposed to sea water,
either reinforcing steel in areas of damage or, more important in offshore
installations, other steelwork which is in direct electrical contact with the
reinforcement.
T w o possible situations may accordingly arise in a structure partly immersed in the
sea:
.
With a large oxygen-depleted underwater zone, the reinforcing steel develops a
general active corrosion condition, at a very negative potential. There is a
tendency for the potential of reinforcement in a limited above-water "galvanic
interaction zone" to be held in the passive range (below the pitting potential),
and any small amounts of bare steel are protected (Figure 44).
Cathodic protection of offshore structures
141
-500
Key
( Potenfial o f embedded steel
C'=Potential of embedded steel coupled
to bare steel
5
Potential o f bare steel
S'=Potential of b a r e steel coupled
t o embedded s t e e l
E =Potential drop through concrete
-600
Cathodic current diverted
to embedded steel
of b a r e steel
---am5
-700
=
-a00
u
-z
embedded steel
cn
a
>E
v
c
1
c
al
c
9-900
Log current
Figure 44
Schematic of couple between steel in sea water and steel in oxygen-depleted
concrete (the embedded steel corrodes sacrificially and reduces the corrosion
rate of fhe bare steel)
Alternatively, the "galvanic interaction zone" (including perhaps the inside of a
"hollow leg") may be able to polarise the underwater reinforcement into the
passive potential range. There is increased corrosion of any parts of the
immersed steel which are bare, inchding reinforcement exposed by damage or at
wide cracks (Figure 45).
However, it has been shown(89)that cracks in the concrete Iess than about 1 mm
wide at the surface can be tolerated without causing localised corrosion under
water, even though chloride ions have penetrated immediately to the steelkoncrete
interface. Corrosion is therefore unlikely to initiate at the fine cracks which
normally form in all reinforced concrete.
142
MTD Ltd Publication 90/102
C
-300.
= Potential
of embedded steel
C’= Potential of embedded steel coupled
to bare steel
S = Potential of bare steel
S’= Potential of bare steel coupled
to embedded steel
E = Potential drop through concrete
Cathodic current diverted
-500,
I
of baresteel
d
U
01
U
\
IL D ~ D O - = D - - ~ D D - D O D ~ +
o
l
a
>-
\
’
E
-.- -700.
I
m
c
1
\
1
\r\
I
c
1
W
c
0
a
Log current
Figure 45
Schematic of couple between sleel in sea water and passive steel in concrete
(the embedded steel cafhode increases the corrosion rate of the bare steel)
Whether or not pitting corrosion of reinforcement occurs in concrete in the upper
parts of the structure, heyond the galvanic influence of the underwater zone,
depends only on the properties of the concrete cover, chIoride penetration,
moisture content, etc.
Cathodic protection of offshore structures
143
It can broadly be concluded that for isolated reinforcement in relatively
undamaged concrete, the first situation probably occurs in most bottom-mounted
offshore structures in deep water, because they have a very large submerged surface
area and a comparatively small tidaysplash zone area. In particular, steel
prestressing tendons may be expected to be isolated from oxygen ingress and from
direct galvanic interaction with other steelwork.
The second situation could possibly develop in structures in shallow water, and it is
conceivable in a deeper water structure if it is hollow and air-filled. However, in
practice, there is the additional factor that the reinforcement in almost all marine
concrete structures is connected, either deliberately or accidentally, to quite large
areas of submerged bare steel. Because attached steelwork on concrete offshore
structures in the North Sea normally has cathodic protection, there is a further
interaction between the cathodic protection system and the rebars in the submerged
concrete and in the "galvanic interaction zone".
7.4
CATHODIC PROTECTION OF STEEL IN CONCRETE
The application of cathodic protection to steel in concrete presents several practical
problems. A general requirement is that the embedded steel should be electrically
bonded to ensure effective distribution of protection and to prevent possible "straycurrent" corrosion within the steelwork system. Normally, electrical continuity
within reinforcing steel is adequate, but it may be necessary to consider the
desirability (or the practicability) of either deliberately bonding or isolating the
reinforcement from prestressing steel and attached steelwork, In considering
related problems (distributing the protection current through relatively high
resistance concrete, deciding a suitable protection criterion, and monitoring the
achievement of protection), a clear distinction needs to be made, depending on
whether the concrete is immersed or is normally exposed to the atmosphere.
7.4.1 Immersed concrete
There is little evidence that cathodic protection of underwater reinforced concrete
is necessary, but it is not easy to isolate the reinforcement from other steel, which
usually is protected. The most common criterion for protection of bare steel in sea
water is polarisation from its corrosion potential of around -650 mV to at least
-800 mV with reference to a Ag/AgCl electrode in the sea. Steel in fully immersed
concrete can reach potentials as low as -1000mV Ag/AgCl and therefore,
depending on resistivities, it could be sacrificial to adjacent bare steel. However, it
seems unlikely that this circumstance would arise in any but a fully immersed
structure. General corrosion is anticipated when reinforcement reaches these
negative potentials, but it is likely to be very slow on account of oxygen depletion.
However, if steel in contact with concrete is polarised to -750 to -850 mV Ag/AgCl
in sea water, pitting has been reported("). Pitting has also been reported on steel
encased in cement mortar at similar potentials, but the pits were generally small,
except at very negative potentials, in the region of -1200 mV Ag/AgCI(91). Sucb
corrosion is believed to be the result of the formation of a soluble corrosion
product, accompanied by hydrogen evolution, corresponding to the bottom righthand active region of Figure 39.
144
MTD Ltd Publication W1M
The possible effect of hydrogen evolution on the bond at the steeVconcrete
interface, and the risk of hydrogen embrittlement are also points of concern. In
laboratory tests, no deleterious effect on bond was found after either olarisation
for 1 year at -1360 mV(m) or for nearly 2 years at -1550 mV Ag/AgCl$3) (nor was
any significant corrosion remarked in either test). However, embrittlement at very
negative potentials remains a concern, particularly if prestressing tendons might be
affected.
In summary, all the implications of cathodically protecting a combination of
reinforcement and bare steel in offshore structures, particularly with impressed
current which might result in local over protection, have not yet been fully
investigated. In any structure where negative potentials are not reached naturally
(perhaps because of galvanic interaction from a tidaysplash zone or a “hollow-leg”
effect as described above), it may be prudent to follow a recommendation(=) that
the system should be polarised to a potential 50 to 100 mV more negative than the
normal level for steel in sea water (i.e. -900 to -950 mV AdAgCl). However,
because it might also be prudent to avoid potentials much more negative than say
-1000 mV Ag/AgCl, the practical difficulties of achieving such a narrow potential
range are formidable.
At present, the only guidance relates to an additional current allowance to account
for drain to the reinforcement, but until galvanic interaction with air-exposed
concrete surfaces is better understood, the current estimates will have to be made
on an empirical basis. Suggested allowances based on the surface area of concrete,
range from 0.5 to 1.0 mA/m2(”) to 3 to 5 rnA/m2(”). Espelid et uZ?’~) suggested that
the allowances applied so far for concrete platforms in the North Sea seemed to
under estimate the total current drain to the reinforcement by a factor of 2 to 3 if
the current drain to the tidaVsplash zone and the actual area of embedded steel are
not taken into account. This uncertainty seems to have been incorporated in the
1987 revision of the NPD Guidelines(97),which call for 0.2 to 4 mNm2 of
reinforcement area. If oxygen access is restricted sufficiently to bring the steel to a
free corrosion potential more negative than -800 mV Ag/AgCl, no current should
be required unless the cathodic protection system actually polarises the steel to a
more negative potential, The current densities required to polarise even to
-1000 mV Ag/AgCl are then very small.
7.4.2 Concrete above water
Whereas corrosion is rarely a problem under water, there is always a risk that
reinforcement will corrode in concrete exposed to air, and this risk is particularly
great if the concrete is contaminated by chloride. Once corrosion has been
initiated because of chloride penetration, it can prove difficult to prevent its
Cathodic protection of offshore structures
recurrence without drastic repairs which often entail cutting away and replacing all
the concrete to behind the reinforcement. Cathodic protection, which in principle
can control corrosion even in the presence of high levels of chloride, is an
attractive alternative or supplement to repair. The very serious problems with
corroding reinforcement in concrete bridges affected by chloride from de-icing salt
has provided the incentive to develop the necessary technology. The very high
quality of the concrete used in offshore structures should greatly reduce the rate of
penetration of chloride to rebar depth. To date there is no indication that
significant corrosion has occurred in the splash/atmospheric zones of such
structures. However, it is prudent to consider whether cathodic protection could
be used, if required, on the above-water parts of concrete offshore structures.
Cathodic protection of reinforcement in concrete exposed to air poses two major
problems:
7.4.2.1 The anode
Because of the relatively high resistivity of concrete, it is necessary to place the
cathodic protection anode close to the reinforcement. This can only be achieved
by distributing the anode over the surface of the concrete. Several impressed
current systems have been successfully developed, based either on conductive
coatings or conductive overlays, or on anodes fully or partly embedded in the
concrete surface. Prolonged operation at too high a current density could result in
deterioration of the concrete at the concretelanode interface, althou h it is believed
that limiting the sustained current density to below about 100 mA/m will
minimise such damage. Long-term reliability of the chosen "extended anode"
would be of paramount importance for any system used in an offshore application.
4
7.4.2.2 Measurement of potential
There are practical difficulties in measuring potentials of steel in concrete, and
these have not yet been entirely overcome. For example, it is not easy to achieve a
reliable and reproducible junction between a reference electrode and the concrete,
and the exact position of the electrode in relation to the reinforcement and the
anode may be important. Reference electrodes designed to be embedded in
concrete are available, and measurements may also be made using electrodes
placed on the concrete surface. But there are obvious difficulties in using either
method on the splash zone of on a offshore structure.
The reliability and longevity of the present generation of cathodic protection
systems for air-exposed reinforced concrete have probably not yet been proved
sufficiently for the technique to be considered for offshore use. However, this is a
very active field of development, and if the need should arise in the future,
cathodic protection of reinforcement in the splash/atmospheric parts of an offshore
structure could be a viable option.
146
MTD Ltd Publication 901102
7.5
NORTH SEA EXPERIENCE
7.5.1 Design of concrete structures
There are 20 concrete structures in oil and gas service in the North Sea. The oldest
of these is the Ekofisk Centre in the Norwegian Sector, which has been in service
for 17 years. There are 12 other structures which have been in service for more
than 10 years. Table 12 lists the structures and their installation dates.
The use of concrete structures for oil and gas roduction has prompted
considerable research into concrete
Large-scale research
programmes included the "Concrete in the Oceans" programme in the UK and
several DnV research ro rammes in Norway. The results were incorporated into
several design Codes(9Q95,fbO,lOl,
The design Codes reflect a consensus among the certifying authorities, designers
and operators of concrete structures in the North Sea that steel which is embedded
in high quality concrete with adequate cover does not require any other corrosion
protection. The Codes require concrete compositions which should, according to
laboratory results, have low permeabilities to oxygen and chlorides.
All offshore Codes require a minimum cement content of 400 k g h 3 and a
maximum w/c ratio of 0.45. The Norwegian Standards also require cements to have
a minimum C3A content of 5%, on the basis that this may further reduce the rate
of chloride penetration through the concrete by chemically complexing the
chlorides,
The Codes do not require pulverised fuel ash or blast furnace slag to improve
concrete properties. There have been no reported uses of these additives in
Norway, but at least one structure built by Howard Doris included 20% pulverised
fuel ash in its cement composition. No operators have reported using galvanised or
coated reinforcing steel, and none have reported using coatings on the concrete to
reduce permeability.
The design Codes specify a minimum concrete cover of 50 mm for reinforcing steel
and 70 mm for prestressing steel. Some designers specify a minimum cover of
75 mm for reinforcing steel and 100 mm for prestressing steel.
Prestressing elements can tolerate much less corrosion than the reinforcement, for
they are made of high strength steels which may suffer hydrogen embrittlement as
well as tensile failure through loss of cross section. Therefore they should not be
either over protected or under protected. Prestressing elements run in steel ducts
which are cast into the concrete, and after tensioning the ducts are filled with
cement grout. In order to reach the prestressing steel, oxygen and chlorides have to
pass through two additional interfaces as well as the duct itself.
Cathodic protection of offshore structures
147
TABLE 12 NORTH SEA CONCRETE STRUCTURES
--PLATFORM
OPERATOR
DESIGN
___.-
INSTALLATION
WATER
DEPTH
(m)
WEIGHT
(tonne)
BRITISH SECTOR
Beryl A
Brent B
Brent C
Brent D
Cormorant A
Dunlin A
MCP-01
Maureen ALC
Ninian Centr
Mobil
Shell
Shell
Shell
Shell
Shell
Total
Phillips
Hamilton
Norwegian Contr
Norwegian Contr
McAl y ine/Seatank
Norwegian Contr
McAlpineISeatank
ANDOC
Howard-Doris
Howard-Doris
Ove Arup
July 75
Aug 75
June 78
July 76
May 78
May 77
July 76
July 82
1990-Planned
117
139
141
139
150
94
93
143
50
2000000
165000
280000
178000
295000
150000
8000
345000
22000
C.G. Doris
Howard-Doris
McAlpindSeatank
Norwegian Contr
Norwegian Contr
Norwegian Contr
Norwegian Contr
Norwegian Contr
Norwegian Contr
June 73
Sept 75
June 76
June 77
May 77
Aug 81
Aug 84
Aug 86
Aug 87
73
98
104
104
145
145
145
135
142
200000
150000
130000
164000
200000
300000
300000
340000
173000
Norwegian Contr
1988-Planned
105
C.G. Doris
C.G. Doris
Aug 83
Aug 83
NORWEGIAN SECTOR
Ekofisk Centr
Frigg CDP-1
Frigg TP- 1
Frigg TCP-2
Statford A
Statford B
Statford C
Gulfaks A
Gulfaks B
Oseberg A
Phillips
Elf
Elf
Elf
Statoil
Statoil
Statoil
Statoil
Statoil
NorsM
Hydro
GERMAN SECTOR
Schwendeneck A
Schwendeneck B
Texaco
Texaco
25
16
7.5.2 Cathodic protection design
Cathodic protection is applied to exposed steel on North Sea structures, usually
with some allowance for current drain to the reinforcing steel. The design codes
provide a wide range of allowances and they have changed over time. The 1977
revision of the DnV Rules required 0.5 to 1 mNm2 of steel area in the outer layer
of reinforcement. The 1986 DnV Recommended Practice RPB401(’) has no
requirements for cathodic protection drain to reinforcing steel, while the 1987
revision of the NPD Guidelines((n)requires 0.2 to 4 m u m 2 of reinforcing steel
area.
148
MTD Ltd Publication 90/102
BS6235("'), now withdrawn, contained a recommendation that reinforcing steel and
exposed steel be electrically insulated. The 1985 revision of the Germanischer
Lloyd Rules(") requires positive electrical bonding between the reinforcement and
external steel with an allowance of 3 to 5 m N m 2 of steel in the outer layer of
reinforcement. The surface area of the outer layer of reinforcement can, for
practical purposes, be estimated to be equal to the surface area of concrete.
The exposed steel on concrete structures may include pipeline risers, distribution
pipes for oil and ballast, drilling conductors, and riser and conductor support
structures. The surface area of exposed steel on concrete structure varies from 1 to
10 % of the total concrete surface area, although the oldest concrete structure has
no exposed steel at all. Cathodic protection for the exposed steel is normally
supplied by aluminium anodes, although one platforni has impressed current
systems inside two flooded legs.
The exposed steel may be attached to the outside of the structure and be exposed
to the open sea, or it may run inside a flooded leg or shaft. Flooded legs may be
static or open to the sea. Design current density for external steel and steel inside
legs open to the sea varies from platform to platform in the range of 120 to
160 mA/m2. Design current density for steel inside static legs ranges from 50 to
80 mA/m2.
The exposed steel on many of the structures is coated to reduce the number and
weight of anodes required. The available current in these designs depends on the
coating breakdown factor allowed. These factors range from 6 to 20 %, depending
on the type of coating specified and the designer of the structure. Use of coatings
and the above breakdown factors leads to design current densities on coated steel
of 10 to 30 mA/m2.
Some operators and designers of concrete structures specified the deliberate
bonding of external steel to reinforcements, others specified the deliberate isolation
of all or some external steel, and still others assumed that the external steel is in
electrical contact with the reinforcement. Most designers used a design value of 1
to 2 m N m 2 of concrete surface area as an allowance for current drain to the
reinforcement, regardless of whether or not isolation was specified, This allowance
was used for platforms built in the mid 1970s, and it is still used on recent designs.
7.5.3 Inspection of concrete structures
The main methods of inspecting concrete structures are visual inspection of the
concrete surface and potential surveys of exposed steel. In the atmospheric zones
of structures, including inside dry shafts, visual inspection is carried out by
inspection technicians. In the submerged zones, visual inspection is carried out by
divers or remotely-operated vehicles (ROVs).
Visual inspectors look for cracking, spalling or rust staining of the concrete as an
indication of deterioration, also for evidence of mechanical damage.
Cathodic protection of offshore structures
149
Cathodic protection potential surveys of external steel are made by divers or ROVs.
Potential surveys on steeI inside flooded legs are made by dipping a half cell into
the leg. This method is more limited in terms of accuracy, but it is the only
method available, because divers are not permitted to work inside flooded legs for
safety reasons.
Cathodic protection surveys concentrate on pipeline risers and riser supports,
because these are critical to the operation of the installation. In most cases, the
surveys consist of potentia1 measurements on the risers and riser supports and
visual inspection, supplemented by physical dimension measurements of sacrificial
anodes.
A number of supplementary methods of inspection have been tried on concrete
structures. These include core sampling of the concrete and reinforcement,
electropotential mapping of the concrete, current density measurements on the
steel and the concrete, and electrical continuity checks where electrical bonding is
part of the design.
7.5.4 Field experience - corrosion of reinforcement
Field experience with all concrete structures in North Sea oil and gas production is
cumulatively more than 200 years, although the maximum exposure of any
individual structure is 17 years. Service experience reported for all structures is
similar. No operator has reported any evidence of corrosion of the reinforcement
in any concrete structure. There is no reported evidence of cracking or spalling of
the concrete because of corrosion in the dry, submerged or splash zone areas.
There are several instances of mechanical damage to concrete structures which
exposed reinforcing steel in the submerged zone or the splash zone. Examination
of the reinforcing steel revealed no evidence or corrosion prior to its exposure. In
cases where the damage was in the submerged zone, the exposed steel typically
exhibited potentials in the range of -850 to -950 mV Ag/AgCl, and it did not show
any signs of corroding.
This was typically regarded as an indication that the reinforcing steel was drawing
on the external cathodic protection system, although this could also be evidence of
the type 1 behaviour of the reinforcing steel described on page 141 and illustrated
in Figure 44. A DnV research programme commenced in 1976, in which
electrochemical measurements were conducted on reinforced concrete specimens
over a 3-year period("). The specimens were representative of concrete and
reinforcement used in the construction of offshore structures. The reinforcing steel
typically had corrosion potentials of -200 to -500 mV Ag/AgCl when the specimens
were first immersed in sea water. Over a period of months, the corrosion
potentials declined to about -900 mV Ag/AgCI. The DnV researchers concluded
that this was because of restricted oxygen availability at the steel after the oxygen
originally present in the concrete had been consumed.
150
MTD Ltd Publication 90/102
Core samples are reported to have been taken on four structures. In three of the
cases, cores were taken from the inside of dry legs, and in one case from the inside
of a leg which was permanently flooded with stagnant water. The periods in service
prior to coring ranged from 5 to 10 years. The reinforcing steel showed no
evidence of corrosion in any of the four cases.
The evidence from field experience of concrete structures indicates that the
specification of high quality concrete with good cover is sufficient to prevent
deterioration and to protect the reinforcing steel from corrosion at this stage in the
lives of the structures.
-
7.5.5 Field experience cathdic protection
Adequate levels of cathodic protection to prevent corrosion of steel attached to
concrete structures have been achieved in all cases. In general, experience is
consistent with the expected polarisation behaviour of the reinforcing steel, where it
draws current from the exposed steel in the early life of the structure while in a
passive state, and then ceases drawing current or even contributes to the cathodic
protection of exposed steel as oxygen becomes depleted and settles in an active
state at a potential of about -900 mV Ag/AgCl.
It is common experience that initial anode consumption on concrete structures is
much higher than expected for the protection of the attached steel alone. Several
operators report adding additional sacrificial anodes to risers because of high anode
consumption.
In these cases, anode consumption stabilised at low levels within a few years, and
although anode consumption is still monitored, there appears to have been no
instances of anode consumption remaining at high levels. Surveys indicate that
potentials in the range of -900 to -1000 mV Ag/AgCl are common on older
structures.
There is one platform in the North Sea which has impressed current systems
operating inside the flooded legs to protect risers and drilling conductors. The
steel on this structure is electrically isolated from the reinforcement. The
impressed current systems polarised the steel to approximately -950 mV AgIAgC1,
based on permanent reference cells and annual surveys with a dipping half cell.
After approximately 10 years of operation, the average current densities required
by the steel in these legs is about 10 to 25 mA/rn2. This is consistent with a good
calcareous film on the steel and the low water velocities in the legs. Impressed
current systems on steel jackets require current densities of 30 to 50 mA/mZ after
similar periods in service, reflecting the influence of water velocity.
Cathodic protection of offshore structures
151
7.6
CONCLUSIONS
Two important conclusions can be drawn from experience of reinforced concrete
structures in the North Sea:
There is no evidence that there has been any significant corrosion of reinforcing
steel in any structure to date.
In most cases, cathodic current has been diverted to reinforcement in the
underwaterkidal zones from systems installed to protect bare or coated steel
immersed in sea water, This current was appreciable only during the early life of
the structure, and after a year or so the current flow either to or from the
reinforcement became insignificant.
.
.
However, these conclusions are based on information from very limited inspections,
and therefore it is also concluded that:
More information is required, particularly quantitative data on the early current
flow to reinforcement and its subsequent decay. It is also significant that no
means exist to confirm the absence of corrosion on prestressing tendons.
.
From a consideration of the corrosion processes involved, it is further concluded
that:
It is unlikely that cathodic protection is necessary to prevent corrosion of
reinforcement in the underwaterkidal zones, However, provided an allowance is
made for the early current drain on a cathodic protection system, there is no
reason to attempt the difficult task of isolating it from the reinforcing steelwork.
The absence to date of corrosion in the splash/atmospheric zones is no guarantee
that it will not occur in the iuture. Technology to provide cathodic protection for
reinforced concrete exposed to the air is developing rapidly, and it could perhaps
be adapted to control reinforcement corrosion on the above-water parts of an
offshore structure should it occur.
.
152
MTD Ltd Publication 901102
Section 8
Cathodic protection systems for subsea
installations and pipelines
Cathodic protection of offshore structures
153
8 a Cathodic protection systems for subsea installations and
pipelines
8.1
OVERVIEW OF SYSTEMS FOR SUBMARINE PIPELINES
In theory, the cathodic protection system for a submarine pipeline could take
numerous forms, using all sorts of shapes, sizes, and locations for sacrificial or
impressed current anodes.
In practice, most submarine pipelines are protected by sacrificial anode bracelets of
the half-shell or segmented type (Section 8.6) attached to the pipeline at regular
intervals (generally 25 to 200 m), and pre-installed to the pipeline before it is laid
on the sea bed. Submarine pipelines protected in any other way are very much the
exception, although even for the simplest pipeline the cathodic protection system
may become a little more complex at its extremities (see Section 8.7).
The reasons for the preference for the bracelet anode system are as follows:
a It provides a sacrificial anode system designed to last for the service life of the
pipeline, so that it should be maintenance free.
No separate subsea or offshore operation is needed to install the cathodic
protection system, because anodes are in place during pipe laying.
The use of bracelet shapes places no restrictions on the methods of pipeline
installation (laying and trenching), and it has minimal (or no) impact on the
speed of pipe laying. Delays in this operation are usually the most costly of the
entire pipeline project, to either the client or the contractor.
a Once installed on the sea bed, the anodes are not vulnerable to accidental
damage in the same way that remote anodes sitting proud of the sea bed might
be, nor are there any cable runs vulnerable to weather damage in the splash/tidal
zones.
a Cost penalties, if any, in choosing this type of system over any other are
insignificant compared with the total installed cost of the pipeline.
The arrangement is well tried and tested, with no evidence to date of any
shortcomings if the system is well designed.
.
.
.
There are some circumstances in which the conventional sacrificial bracelet anode
system may not be the most appropriate:
Short pipelines emanating from shore or from offshore platforms, or portions of
longer pipelines adjacent to landfalls or platforms, may be protected by an
impressed current system sited on land or at the platform primarily for the
purpose of protecting onshore or platform facilities. (Note: Few North Sea
offshore platforms are in fact protected by an impressed current system.)
.
Cathodic protection of offshore structures
155
a
a
Bracelet-type anodes may not be suitable for certain configurations of nonenclosed pipeline bundles. In this context, it should be mentioned that a bundle
of pipelines enclosed within a carrier pipe is not usually protected against
corrosion by cathodic protection. The carrier pipe itself may be protected by
bracelet anodes, but as a non-critical item it may instead be furnished with a
generous corrosion allowance to maintain a minimum wall thickness and
submerged weight during installation.
If the cathodic protection system on an existing pipeline has to be augmented insitu (e.g. if the pipeline design life is to be increased), considerations of
installation cost may militate against installing new, relatively frequent, bracelets
rather than fewer, higher current and capacity, and larger anode designs.
The text which follows deals primarily with the design of bracelet anode system
which culminates in the selection of an appropriate anode size and spacing along
the pipeline, as shown in Figure 46. Where appropriate, however, the merits and
demerits of other types of systems are brought into the discussion. For instance, if
the pipeline diameter is small or if some installation techniques, in particular
reelbarge installation, are employed, the use of bracelet anodes may have a
significant impact on pipelaying timescales.
96
84
anode Length and s
72
60
48
-- 36
E
.-
24
u
m
$12
W
U
0
SO
0.25
0
0.50
0.75
1.00
1.25
Anode length ( m 1
Notes: 1. S, = maximum spacing of anodes to satisfy material mass requirement
5,=maximum spacing of anodes to satisfy end-of-life c u r r e n t requirement
2. Shaded area represents allowable and feasible combinations of
anode l e n g t h and spacing
3. Assumed t h a t anode dimensions other than l e n g t h a r e held constant
Figure 46
156
Typical optimisation of bracelet anode system
MTD Ltd Publication 901102
8.2
TIiE PLACE OF CATHODIC PROTECTION IN PIPELINE DESIGN
8.2.1 Bracelet anode systems
The design of a sacrificial bracelet anode type of cathodic protection system can be
finalised only when all of the following information is known:
a the pipe material
pipeline outside diameter
type and thickness of anti-corrosion coating(@
.
.
.
.
a
thickness of concrete coating, if any, required for stability purposes
design of pipeline insuIation materials, if any
method of pipeline installation and trenching
anticipated pipeline burial status througbout design life
pipeline product inlet temperature, and temperature gradient along the line
a
type of soil (if buried), resistivity, SRB activity
a
location, water depth and ambient seawater temperature
a
the design of interfaces at each end of the pipeline.
Consequently, in the most straightforward instances, the design of the cathodic
protection system may be delayed until the design of the pipeline proper has been
finalised, although it is naturally considered good practice for cathodic protection
and coatings to be designed in tandem in order to achieve a consistent and
economic approach to external corrosion prevention measures.
Apart from the interrelationship between anodes and anti-corrosion coatings (see
Section 8.3.4), the cathodic protection system design may in certain circumstances
of necessity become more interactive with other aspects of pipeline design, these
being:
8.2.1.1 I m p m on method of pipeline installation
Where a choice of pipe laying method is available, usually conventional laying (i.e.
welded on a lay barge) or laying from a reel, the anode design and the practicalities
of anode attachment (particularly on the reel barge) have to be fully investigated
prior to a technical choice between laying methods. This is eIaborated further in
Section 8.6.
8.2.1.2 Elevated temperature pipelines
The practicalities of external corrosion prevention need to be addressed at an early
stage when considering the options available for dealing with transportation of
"hot" (50°C or above) products. In particular, the ability of sacrificial bracelets to
protect effectively determines the technical feasibility of an entire design option.
Cathodic protection of offshore structures
157
This ability to protect may be compromised of:
very large current density requirements for bare steel surfaces at elevated
temperatures in sea water (i.e. unburied pipeline) - see Section 8.3.6
deterioration of anode material electrochemical properties at elevated
temperatures (i.e. when effectively insulated on a buried pipeline) - see Section
8.4.1
for an insulated pipeline, the feasibility of a compatible design incorporating
anodes, anti-corrosion coating and insulation materials.
In some cases where a minimum pipeline temperature is mandatory, several
pipeline options may be available, namely an insulated or non-insulated pipeline,
buried or not buried. Each option has a different temperature gradient along the
pipeline length, thus impacting on pipeline hydraulic (and possibly internal
corrosion) design. The choices may be further complicated by the availability of
different installation options, as discussed earlier.
Consequently, for an elevated temperature pipeline, insulated or not, the ability to
design a reliable coating/ cathodic protection system (using anodes other than
bracelets if necessary) to protect the pipeline throughout its design life is a primary
requisite for the technical acceptability of each design option. Restrictions imposed
by the anti-corrosion design may have implications for such basic design
parameters as pipeline sizing (through hydraulic design or installation
consid er a ti o11s) or pipe ma teria1 (from internal corrosion consid er a ti on s).
8.2.2 Other types of anode systems
From the foregoing discussion, it is clear that the design of cathodic protection
systems which include anodes remote from the pipeline tends not to impinge as
directly on other aspects of pipeline design or construction as does the use of
bracelet anodes (or other shapes mounted on the pipe itself), other than in terms of
compatibility with anti-corrosion coatings. For example:
The presence or absence of weight coating or insulation material is irrelevant
except in as much as rate of breakdown of the anti-corrosion coating may be
affected.
Pipeline installation techniques are largely irrelevant, because anodes are
installed separately at a later stage.
Anode performance is unrelated to pipeline burial status (although current
requirement and capacity of anodes will be affected).
Requirements for the physical design of interfaces with the pipe and all types of
coating are much less onerous, because only cable tails are attached to the pipe.
Instead, there are a numher of factors unconcerned with pipeline design, but very
much concerned with what is a separate sub-system, which have to be considered at
the initial stages offormulating the externaI corrosion prevention philosophy.
158
MTD Ltd Publication 90/102
These are:
the ability to install the anode(s) on the sea bed, to make reliable cable
connections to the pipe, and to maintaidreplace as necessary. This may be
dependent on water depth and seabed topoFaphy.
the availability, for impressed current systems, of convenient electrical power
supplies and the ability to install an adequately protected submarine power
supply cable to each anode location.
the ability (particularly for impressed current systems) to make satisfactory
interfaces with other existing or planned cathodically protected installations,
without detrimental interference effects (see Section 8.7).
Given that a non-bracelet system is considered only in fairly exceptional
circumstances, these factors should not be insurmountabIe even if, for a particular
pipeline installation, the solution is relatively expensive or a certain amount of
development work is found necessary.
8.3
DESIGN REQUIREMENTS FOR PIPELINES
8.3.1 General
As outlined in Section 6, the primary objective of the cathodic protection system
designer is to deliver a sufficiently high level of current to protect areas of bare
steel on the pipeline at all stages in its d e s i p life. An associated objective is to
ensure that there is sufficient consumable anode material for the necessary
protective current to be provided without interruption, for the full duration of
pipeline service life.
The preparation of a Design Basis is the first task in the design of a submarine
pipeline or any other subsea installation. That Design Basis will include
environmental data and other assumptions upon which the design of the cathodic
protection system will be based. Section 2 of DnV RPB401(') may be used for
guidance on design requirements, being the most recent standard produced for the
North Sea environment, although with the imminent issue of Part 2 of CP 1021 this
may change for UK waters. Adherence to the recommendation of RPB401 is, in
practice, usually mandatory when designing cathodic protection systems for use in
the Norwegian Sector of the North Sea, while adherence to most of its
recommendations is the current norm when dealing with other locations on the
Northwest European Continental Shelf.
8.3.2 Environmental conditions
The environmental parameters which impinge on cathodic protection system are
the electrical properties of the electrolyte(s) in which the protected structure is
immersed or buried. The relevant, experimentally-derived recommended values
for Northwest Europe are presented below:
Cathodic protection of offshore structures
159
Electrolyte
Resistivity (ohm.cm)
(Approximate values)
Sea water
Sediments :
Soft clay (mud)
Clay
Sandstiff clay
30
60-75
75-110
110-160
It is not usual for field measurements of resistivity to be taken, so the above values
are used as a guide. The choice of value to use when a range is sugested is
influenced by the information on soil types which is invariably collected during
early surveys of the pipeline route. However, in every case, it is good practice to
err on the side of conservatism if uncertainty exists. For example, for a buried
pipeline, the design ignores patches of low resistivity sediments within extensive
higher resistivity sediments, An anode produces less current as the electrolyte
resistivity rises, all other factors being equal.
However, field resistivity measurements are necessary at landfalls where the
pipeline is buried in soils not forming part of the sea bed, and which tend to have
resistivities much higher than those tabulated above.
8.33 Protective potentials
Current codes for cathodic protection design indicate the following protective
potentials:
Environment
Sea water
Sea bed sediments
Protective potential (Volts wrt Ag/AgCl)
Positive limit
Negative limit
- 0.80
- 0.90
- 1-05
- 1.05
L
where the positive limits are those defining the minimum potential level to prevent
corrosion, and the negative limits are those beyond which damage to carbon and
low alloy steels through over protection is a risk. The assumption is made that,
when buried, a pipeline is always in an environment which is, for practical
purposes, anaerobic and subject to activity by bacteria from rotting animal and
plant matter. Hence the lowering of the positive limit.
It is argued in some quarters that the need to shift the buried steel potential to
-0.90 V is too onerous a requirement, and that it is not a proven necessity.
Consequently, outside the Norwegian Sector of the North Sea, a positive limit
(buried pipeline) of -0.85 V is often used, taken from Table 1 of CP 1021(72).Some
operators raise the limit still further to -0.80 V, based on their own research work.
160
MTD Ltd Publication 90/102
The negative limit, at -1.05 V, is one which should not be exceeded when the
cathodic protection system utilises zinc or aluminium-zinc-mercury anodes, but it is
in danger of being exceeded with some aluminium-zinc-indium alloys. That value,
too, is regarded by some as too restrictive, and a value of -1.15 V has been
proposed, this to be applicable only to steels susceptible to hydrogen cracking.
Although hydrogen embrittlement caused solely through over protection in sea
water is not considered a problem for normal pipeline steels (say up to API
Specification SL Grade X 75), cracking is considered a possibility for these
materials when the steel is beyond the negative potential limit and additionally
subjected to low frequency fatigue, such as might occur in sea currents at risers or
pipeline spans. Because regular wave and current forces are always likely to
impinge on subsea pipelines, when not deeply buried, and on structures, a uniform
negative limit tends to be adopted.
However, negative potential limits effectively limit the "throw" of protection along
a pipeline from remote anodes which have highly negative open circuit potentials
(i.e. all impressed current anodes and magnesium sacrificial anodes - see Section
8.5.5).
8.3.4 Current requirements
The current required to protect an area of coated steel is given by:
Current = (Current density for protection of bare steel)
x(% of coating breakdown)
x(area of coated steel).
The initial, mean and final currents required to protect subsea installations with
thin film coatings, such as marine grade paints, may be calculated using the values
recommended for current density and coating breakdown in Tables 2.3 and 2.4,
respectively, of DnV R P B ~ O ~or
( ~similar
)
tables in other Codes.
However, the same Code has different recommended values of current density
(Table 2.5) for thick film coatings, with or without concrete weight coating, on
buried or exposed pipelines, valid for design lives of up to 30 years. This Table in
effect combines protection requirements for bare steel with the anticipated coating
breakdown into a single set of values giving protection requirements for coated
steel. The Table may be followed when dealing with a pipeline having a
"conventional thick film coating" (i.e. a reinforced coal tar, bitumen or asphalt
enamel), even though most existing pipelines have performed quite satisfactorily
with a less generous allowance for coating breakdown.
Most current Codes give no guidance on how to deal with pipelines having more
"high performance coatings", particularly under concrete weight coating, such as:
fusion bonded epoxy (FBE)coatings, which are strictly thin film, but which suffer
far less deterioration and breakdown than do solvent-based (even epoxy-based)
paints
polychlorprene (neoprene) and other similar elastomeric coatings, which are
thick film but which appear to suffer less from coating breakdown than do
conventional thick film coatings.
Cathodic protection of offshore structures
161
Quite often the above coatings are used specifically to reduce coating breakdown
and hence to reduce current requirements for cathodic protection systems.
8.35 Burial status
Burial status (i.e. buried or non-buried pipeline) affects the fundamental design
parameters for a cathodic protection system, principally the current density
requirements and t h e size of potential shift specified. For each section of the
pipeline, the anticipated most likely burial status, on average throughout the life of
the pipeline, has to be established. Two factors should be taken into account:
The designed as-laid condition (after all planned backfilling, etc.)
The mobility of the sea bed, and the tendency for change from the as-laid
condition (i.e. natural burial or deburial).
If there is a significant probability of both burial and non-burial occurring, the
adopted design should reflect the most onerous consistent set of conditions (i.e the
cathodic protection system should have sufficient current output capacity and
material to satisfy either scenario).
In general, it can be stated tbat:
Burial of a pipeline decreases current density requirements considerably and
consequently, for a sacrificial anode system, reduces the minimum mass of anode
material required over the design life. This makes the requirements less onerous.
Burial increases electrolyte resistivity around the anode, and hence it effectively
reduces driving voltage hetween anode and pipeline. Because of the more
negative potential shift specified, this acts to reduce t h e current supplied by a
given size and shape of anode. This Ieads to reduced spacing, and thus tends
towards more onerous requirements.
On elevated temperature pipelines, burial tends to reduce the performance of the
sacrificial anode material (see Section 8.4), making the requirements more
onerous.
Note:
The design of pipeline burial is generally a response to considerations of
pipeline stability or physical protection. Burial may occasionally be used
as a means of insulation. The corrosion engineer is therefore usually faced
with a fait accompli. However, it is possible that the cathodic protection
design will influence burial decisions, particularly at elevated temperatures,
if the system promises to become impractical or prohibitively expensive
under the proposed burial scenario.
8.3.6 Temperature effects
If a pipeline is operating at an elevated temperature (because it is carrying hot
product), the demands on the cathodic protection system may he increased in two
ways:
162
MTD Ltd Publication 901102
.
.
increased current density requirements for protection of the steel surface
in certain circumstances, deteriorating anode performance (see Section 8.4).
Standard ractice for increasing current density requirements is reflected in DnV
RPB401(9Pas below, but different values are sometimes currently recommended:
.
.
For bare steel surfaces (Tables 2.3 and 2.4), 2 mNm2 and 1 mNm2 per "C over
25OC and up to 65"C, for steel surfaces in sea water or buried, respectively
For coated surfaces of pipelines (Table 2.5), 0.2 mNm2 per "C over 25°C
irrespective ofburial status.
The relevant temperature is the operating (i.e. product) temperature, although the
outer surface of the pipe may be cooled a little by the surrounding sea water.
Incidental shutdown or upset temperatures are not usually taken into account.
The large increase in protection requirements with increase in temperature is
reflected in the following example:
Current densities to protect pipeline
(Reinforced coal tar enamel + weight coating, not buried)
Pipeline
temperature
Current densities (mNm2)
Initial
Mean Final
<25OC
2
4OoC
6OoC
8OoC
5
9
13
9
12
16
20
15
18
22
26
Clearly, as the temperature increases, the mean current density increases at a
higher rate than the final current density. In other words, for sacrificial systems,
mass requirements are increasing faster than final current requirements. At BOOC,
the mass requirement is hvice that at <25"C. If the line were buried, the mass
requirement could be further multiplied because of deterioration in performance of
the sacrificial anode material.
Hot pipelines are usually encountered:
downstream of platform-mounted compression facilities on gas export pipelines.
on flowlines carrying hot, unprocessed produces direct from wellheads.
With the growth of subsea production systems, the latter case has become much
more common, and it has instigated pressure to find less temperature-sensitive
materials for sacrificial anodes and coatings less prone to breakdown.
8.3.7 Temperature gradients
The design temperature for a pipeline is usually taken to b e the operating (i.e.
product) temperature at the upstream end of the line. The designer may be able to
make use of the temperature gradient as the pipe contents cool in traasit to
Cathodic protection of offshore structures
163
decrease t h e performance required of the cathodic protection system away from the
upstream end of the line. In particular, the temperature drop from top to bottom
of an exposed riser and expansion offset may be considerable, allowing sub-systems
for the riser and pipeline to be developed using different temperature criteria.
Further, it may be desirable to split the pipeline into a number of subsections, each
with a nominal operating temperature, corresponding to the upstream end of each
subsection, for cathodic protection purposes. For example, a long flowline from a
wellhead may be divided up as follows:
Subtiom
I Temperature drop
1
Operating temp
I
The delineation of these subsections is left very much to the judgement of the
corrosion engineer. Some sub-sections may, for convenience, correspond to
sections of buried pipe within an unburied line, or vice versa. Overall, his aim is to
minimise the mass and number of anodes required, subject to the following
constraints applicable to bracelet anodes:
for ease of manufacture and construction, a single design of anode may be used
for the entire pipeline, disregarding risers. Only the spacing between anodes is
changed between subsections
the number of subsections should not be too great, otherwise anode spacing
changes too frequently and the risk of errors during construction is introduced.
In exceptional circumstances, there is no real reason why one or both of the above
constraints should not be ignored, but at the cost of complicating construction
activities.
8.3.8 Effectof water depth
Analysis of ocean waters indicates that oxygen content, and hence corrosivity of the
water, decreases with increasing water depth. However, it is known that the
relatively shallow developed offshore waters of North Western Europe are well
mixed through interaction of countervailing currents, resulting in fairly uniform
oxygen content and thus allowing depth-independent current density requirements
to be applied.
As suhsea structures and pipelines are installed in deeper waters off t h e
Continental Shelf or in the Norwegian Trench, there may be a case for amending
current density requirements, Preliminary work indicates that any such changes
may have to be location-specific as well as depth-related. Nevertheless, DnV
RPB401(’) recommends an increased initial current density, which in the case of
well-coated pipelines and subsea structures (initially with very little bare steel)
should have no effect on the maximum current output demand of the cathodic
164
MTD Ltd Publication 90/102
protection system. Therefore the key criteria on which the cathodic protection
system is designed remain unchanged from those employed in shallower North Sea
waters.
8.3.9 Requirements for high alloy steels
A number of high alloy pipelines, specifically austenitic-ferritic (duplex) stainless
steels, have been installed in the North Sea. These materials are specified for their
resistance to corrosion by the product stream, and it may be wondered why any
external corrosion protection should be needed.
However, it cannot be guaranteed that corrosion in sea water will not occur,
particularly in crevices for example at minor surface welding flaws or at the
extremities of coating disbondment, or where parts of &hesteel surface are in an
anaerobic environment as a result of partial or total burial, or again at parts which
have been "contaminated" by rubbing against low alloy steel. Cathodic protection,
along with anti-corrosion coatings, is necessary to protect any such vulnerable
areas, and because current is also drained from the anodes to non-vulnerable
exposed surface areas, the cathodic protection system should he designed to protect
all bare steel on a "duplex" pipeline.
Consequently, the common approach has become to design a system for duplex
pipelines using much the same parameters as for low alloy steel pipelines. In the
absence of conclusive data to the contrary, it is felt that this is a conservative
approach, and, to date, it has been found to work in practice. However, there are
arguments based on recent testing put forward for further limiting the negative
potential limit for duplex steels to -1.0 V wrt Ag/AgCI, to prevent hydrogen
damage. This is a matter which is still the subject of much debate within the
industry (see also Section 3).
8.4
ANODEMATERIALS
8.4.1 Bracelet anode materials
The most common material used for bracelet anodes was originally zinc to US Mil
Spec 18001J(61),but more recently aluminium alloy materials have been developed
which compete on a technical and economic basis with zinc. Leaving aside
technical considerations, discussed below, the cost of protection using zinc or
aluminium alloy can be compared directly, taking into account:
the relative current capacities of the two materials: roughly 780 AWkg for zinc
and over 2300 M k g for aluminium alloys in sea water
the price per cast tonne of the two materials,
This rule-of-thumb comparison may indicate a clear economic advantage. If the
two materials work out at roughly equal cost, the design employing fewer anodes is
recommended. It can be deduced that, because aluminium alloys have at least
three times the current capacity of zinc (i.e. the zinc is consumed three times
faster), zinc has to be considerably cheaper than aluminium alloys if it is to be
competitive.
Cathodic protection of offshore structures
165
Zinc is unsuitable for use as a bracelet anode when it is required to work a t an
elevated temperature: a limit of 5OoC is usually applied for saline mud service. At
elevated temperatures, zinc suffers intergranular corrosive attack which can lead to
unconsumed (unoxidised) metal being detached from the anode proper (and thus
losing electrical contact with the pipeline), or, in some cases, the zinc surface may
passivate (i.e. to cease being consumed). Consequently, the anode is not consumed
entirely as intended, and its current capacity is severely and unpredictably reduced.
The insulating effect of seabed mud dictates that zinc should not be used on buried
elevated temperature pipelines. Moreover, its use on nominaIly unburied lines at
high temperature, as a close-fitting bracelet, is not recommended, because partial
burial and local insulation through natural soil movement is common. US Mil
Spec 18001J(6') zinc maintains its 780 AWkg capacity over the range from ambient
to SO'C, and its open circuit potential is usually taken as -1.0 V wrt Ag/AgCl.
Several aluminium alloy compositions have been developed for use in marine
cathodic protection, but those overwheImingly used for pipeline cathodic
protection are the aluminium-zinc-indium (Al-Zn-In) or aluminium-zinc-mercury
(Al-Zn-Hg) group, which were developed specifically to minimise the deterioration
of anode performance when buried, compared to immersion in sea water (see
Section 6.4.3.3 for a discussion of alloy composition and properties). Some earlier
alloys bad shown a tendency to passivate when working at a low output current
density when buried. Submarine pipelines are frequently either deliberately buried
or prone to natural burial, and so aluminium alloys are a natural choice for
bracelet anodes.
The performance of aluminium alloy material does deteriorate with increasing
temperature, but intergranular corrosion is not a problem in the range of pipeline
operating temperatures so far encountered. With that risk removed, it is safe to
assume that, even for bracelets on an unburied elevated temperature pipeline, the
anode material is consumed as at ambient temperature in a seawater environment.
Assumptions made concerning burial have to be consistently applied to all design
parameters and anode properties, so that, for example, high anode current capacity
(in sea water) is offset by high current density requirements for protection (in sea
water in comparison to burial).
The open circuit potential of Al-Zn-InaIloys is usually taken to be -1.1 V and that
of Al-Zn-Hg alloys to be -1.05 V wrt Ag/AgCI at ambient temperature.
Current Codes all put forward guidelines on the deterioration of t h e current
capacity and open circuit potential of Al-Zn-In alloys with increasing temperature
in saline mud. But manufacturers' data fromfield tests suggest that these
recommendations are too pessimistic. It is recommended that fully justified
manufacturers' data should be used as the basis of cathodic protection design, with
a small margin for safety included. Al-Zn-Hg alloys are not permitted in
Norwegian waters, but they are not disallowed in UK waters, and current evidence
suggests that they may not have the detrimental environmental effects originally
suspected. The effects of raising the temperature are to change the open circuit
potential in saline mud from -1.05 V towards -1.00 V, and to decrease current
capacity as shown in Figure 47.
166
MTD Ltd Publication 901102
Notes: 1 Data obtained from testing by
Schreiber and Murray at Freeport,Texar
( presented at Corrosion 79 1
2800
D o w Galvalum I1
2 Galvalum and Alanode are registered
trade names of aluminium alloy
anode material
2600
2000
4 Dashed line represents guidance for
AL-Zn-In alloys f r o m
Figure 3.1 of DnV RP8k01
- t o be used if no other
documented data
available
0
1600
1200
2 eoo
Mitsubishi Alanode
rn
Y
-
z1
c-
U
400
U
al
W
0
s o
I
1
10
20
Tern peratu re 1 'C 1
Figure 47
1
I
30
40
r
50
I
I
60
70
I
80
I
90
1
100
Performance of sacrifkial anode materials in hof saline mud
Whereas the above discussion has focused on bracelet anodes, it applies equally to
other anode types directly mounted onto the pipe or integral with the pipe coating,
such as long thin strips.
8.4.2 Other sacrificial anode materials
Anode materials used for protection of subsea structures are normally zinc or an
aluminium alloy, chosen in line with the discussion on the protection of jackets
elsewhere in this publication. If the use of bracelets on a pipeline is not feasible,
one alternative is to use a "remote" sacrificial anode or array of anodes on (or
buried in) the sea bed close to the pipeline. Suitable materials may be high purity
zinc, one of the aluminium alloys, or a magnesium alloy (Mg-Al-Zn).
Magnesium anodes have a current capacity of approx. 1100 Ah/kg and an open
circuit potential wrt AglAgC1 of -1.5 V. The resulting comparatively high driving
potential allows the anode to be buried (i.e. placed in physical safety though in a
higher resistivity electrolyte), while returning a current output comparable to or
greater than zinc or aluminium anodes exposed on the sea bed. If a negative limit
on pipeline potential is imposed, resistance may need to be built into the cabIe
connecting the magnesium anode to the pipe so that the drain point on the pipe is
more positive than -1.15 V.
Cathodic protection of offshore structures
167
Because magnesium alloys are consumed more rapidly than aluminium (around
1200 Ah/kg, assuming 50 to 60% efficiency), and are more expensive by weight, it is
not surprising that they are rarely chosen for subsea use. At landfalls, remote
anodes have to be buried in a groundbed, and the great superiority of magnesium
over other sacrificial materials in soils makes it t h e most suitable competitor to
impressed current systems. Further, at iandfalls a pipeline may be buried very
deeply or even bored by directional drilling through the surf zone. If it can be
shown that there is no likelihood of cyclic loadings in this area or further inland,
the negative limit on the potential of the pipeline may be extended (though
remaining compatible with the maintenance of pipeline coating integrity - see
Section 8.4.4), thus extending the length of line which may be protected by a landbased anode groundbed.
8.4.3 Impressed current anode materials
Remote impressed current anodes suffer in the same way as magnesium anodes in
that distance of potential throw (and hence minimisation of anode sites) is severely
restricted by negative limits on pipeline potential. Additionally, impressed current
systems are of necessity tied to power sources, which for the purposes of pipelines
and subsea installations means proximity to offshore platforms or to a landfall.
If close to the platform, the system is likely to form an integral part of an impressed
current system protecting the subsea structure itself. Consequently, the choices of
materials for anodes are identical to those discussed in Section 6.
At landfalls, impressed current anodes, if used, are in a groundbed, normally
forming part of a more extensive cathodic protection system such as might be
required for an oil terminal or tank farm, a gas processing plant, or a land
transmission pipeline. The choice of anode and groundbed materials is not
necessarily totally governed by the protection requirements of the submarine
pipeline. Buried impressed current anodes are usually high silicon cast iron or
graphite, with a carbonaceous backfilled groundbed to reduce resistance of the
electrolyte immediately surrounding the anode, unless the groundbed is deep
enough for the anodes to be in wet soil below the watertable.
8.4.4 Cathodic dhbondment of coatings
The cathodic reaction on an area of bare steel, at coating damage, tends to lift the
surrounding coating, a process called cathodic disbonding (see Section 4).
Assuming correct surface preparation of the steel, each type of coating exhibits a
threshold steel surface potential, below (more negative chan) which cathodic
disbonding is likely to occur. FortunateIy, for commonly used submarine pipeline
coatings, the threshold potentials tend to be around -1.5 V wrt Ag/AgCl.
In other words, if the negative potential limit for the pipeline is set at around
-1.1 V, to avoid hydrogen cracking through fatigue, or if normal bracelet anode
systems are used, cathodic disbonding is not a problem which normally need worry
the designer of the cathodic protection system.
168
MTD Ltd Publication 90/102
Coating disbonding may establish the negative limit for pipeline potential when
magnesium or impressed current anodes are used and hydrogen cracking is not a
foreseen problem (e.g. a t deeply buried or sheltered locations not subject to fatigue
loadings).
8.5
SYSTEM DESIGN CALCULATIONS
8.5.1 General
The designer of a cathodic protection system has to satisfy three fundamental
criteria:
The system needs to be able to deliver the maximum current required to protect
the subsea installation, whenever that maximum might occur during design life.
This is the current output requirement.
The system needs to have sufficient consumable material to enable the require
protective current to be delivered throughout design life. This is the anode
material mass requirement.
For pipelines, the anode installations need to be sufficiently closely spaced for
voltage attenuation not to leave parts of the pipeline at potentials more positive
than the target protection range.
For sacrificial bracelet systems on pipelines and for sacrificial system on subsea
structures, only the first two criteria above are normally taken into account,
because spacing between anodes is usually relatively small. As a rule of thumb,
where calculations indicate that bracelet anodes may be placed more than 12 pipe
joints (150 m) apart, a voltage attenuation check should be made.
Remote sacrificial anode and impressed current systems are designed to fulfil all
three criteria listed above, but for the impressed current system the precise anode
output capacities, including contingency factors and design of consumable
materials, are performed in conjunction with the anode manufacturer, who will
often have proprietary materials and designs. The design of impressed current
system hardware is covered in Section 6.5.4 and in CP 1021(n).
In cases where a pipeline is divided into sub-sections for reasons of differing
operating temperatures and/or burial status, the criteria should be satisfied for each
and every sub-section.
Pipelines consist of simple geometrical structures. The design process assumes that
no parts of the surface are shielded from anodes mounted around the pipe
circumference or from anodes placed in remote positions. No modelling of current
distribution is undertaken, except perhaps where risers are protected by jacketmounted anodes.
The process of design for cathodic protection systems to protect subsea structures,
such as wellheads, manifolds, protection structures, templates, etc., as opposed to
pipelines, is identical to that used to design systems to protect steel jackets,
described in Section 6, inasmuch as stand-off sacrificial anodes are usually
employed. The chief difference is that subsea structures are usually 100% coated,
Cathodic protection of offshore structures
169
thus demanding maximum current at end of their design life, whereas jackets are
usually bare steel and demand most current at the initial polarisation stage.
8.5.2 Satisfying current output requirements
In order to decrease the amount of sacrificial anode material required for
continuous protection, submarine pipelines and subsea installations are almost
invariably coated with a high quality anti-corrosion system. The initial polarisation
of bare steel surfaces at areas ofdamage to the coating is thus restricted to a very
small current requirement, although current density is high.
The coating deteriorates with time, exposing an increasing area of bare metal, so
that the maximum current to be delivered by the cathodic protection system occurs
at the very end of the design life of the installation.
The final (maximum) current to he delivered by the cathodic protection system, If,
is:
where
If = C&A(A)
C, = final current density for protection of bare steel (A/m2)
b,
A
(81)
= final coating breakdown factor (%)
= area of coated steeI (m2)
For a pipeline,
where
A
= XDL(~~)
D
L
= pipeline outside diameter (m)
= pipeline (or part of pipeline) length (m)
Also from Ohm’s law,
where
If
-
Vp
= positive limit for adequate pipeline protection
4
N
(A)
(V)
= anode material closed circuit potential (V)
R, = resistance of each anode (ohms)
N = number of anodes
V,
Combining Equations (8.1), (8.2) and (8.3), a relationship between R, and N is
obtained:
4 is derived from the resistivity, p, of the electrolyte surrounding the anode, and
the dimensions (and spacing, if in an array) of the anode, using formulae
170
MTD Ltd Publication 90/102
dependent on anode shape. Equation (8.4) is applicable to all coated subsea
pipelines and stmctures which are to be protected by any number of anodes. The
anode system needs to be of a low enough resistivity to deliver the specified design
currents, even at the end of life when anodes are depleted and coatings degraded.
For bracelet anodes on pipelines,
4 =
where
A
0.315 p /
a (ohms) (McCoy’s Formula)
(8.5)
= outer curved surface area of the bracelet (m2) at the end of life.
Examples of other commonly used resistance formulae for anode output
calculations are:
(a)
R
= p/2nqln(4Ur)-l] (modified Dwight)
where
p
= sea water resistivity (ohm cm)
L
= effective anode length (cm)
= effective radius (cm)
r
This formula is generally used for long slender anodes mounted approximately
30 cm from the structure.
(b)
R
=
where
u
= width
= length (cm)
b
II
+
b (Lloyds)
This formulae is generally used for thin plate anodes attached to flat surface.
For a pipeline (or section of a pipeline), from Equations (9.2), (9.4) and (9.5):
where
AV = ( Vp - VJ = the driving voltage
or
fi , AV
s, = 0.315~ C.bfmD
where
Sl
=
for bracelet anodes
(8.7)
uniform spacing of bracelet anodes (m) along the
N pipeline or sub-section of the pipeline.
Equation (8.7) therefore gives the relationship between the exposed area and
spacing of bracelet anodes, S,, necessary to satisfy the maximum current
requirement, at end of design life.
Cathodic protection of offshore structures
171
8.5.3 SatisqZng anode materiel mass requirements
As stated earlier, initial current demand for coated steel is small, while the final
demand is large. Consequently, the mean demand lies somewhere in between, and
it is this value which is used to calculate the mass of sacrificial anode material
required.
The mean current requirement from the cathodic protection system, I,, is
Zm
where
C,
b,
-
C,.b,.A(A)
(8.8)
= mean current density for protection of bare steel (A/m2)
= mean coating breakdown factor (%)
The required net mass (kg) of anode material, W,is given by:
where
--
w
,IT
M
T
= consumption rate of anode material (Ah/kg)
= design Iife (h)
= utikation factor, dependent on anode shape, design,
u
M U
in particular, core design.
Combining Equations (8.8) and (8.9) gives the generally applicable relation:
(8.10)
For pipeline bracelet anode systems the mass per anode, w,is:
w
(8.1 1)
=
Combining Equations (8.2),(8.10) and (8.11) and rearranging:
which gives the relationship between anode mass and spacing, S,
satisfy the mean current requirement over design life.
(8.12)
necessary to
8.5.4 Optimisation of bracelet anode system design
Equations (8.7) and (8.12) give expressions relating anode spacing to exposed anode
area at the end of life and anode net weight, respectively. These expressions define
minimum protection requirements, which are exceeded if the actual anode spacing,
S, is equa1 to or less than both S, and SM.
Also, for a particular design of bracelet anode, there is a relationship between its
net mass,w, and the exposed area, A, at end of life, involving the anode dimensions
and material density.
172
MTD Ltd Publication 90/102
As an example, consider the simplest form of half-shell bracelet anode of uniform
thickness, to be installed on a concrete coated pipeline (Figure 48).
Anode net weight, w = (n/4 [ ( D + 2 1 ) * - 9 ] - 2x1)la.d
=
t =
x =
d =
where ,Z
(8.13)
lengthofanode(m)
thickness (unconsumed) of anode (m)
gap between half shells (m)
density of anode material (k#m3)
Note: This calculation is approximate, and it ignores the volume of steel insert
material and the increase in pipe diameter because of anti-corrosion coating,
both of which are taken into account in the final dimensioning of the anode.
Also, anode surface area (end of life)
(8.14)
It is usual, for concrete-coated pipelines, for the anode outside diameter to equal
the outside diameter of the concrete coating. Thus, t can be held constant in this
case. Also x is usually minimised, consistent with the ability to join the half shells
and attach them to the pipe, and it can be considered a constant.
Thus, both w and A, and hence S,and S
, are functions of a single variable 1, for a
bracelet anode in a particular material for a particular concrete-coated pipeline.
Consequently, it is possible to plot S,and ,
S against 2, to obtain an acceptable
combination of anode length and spacing (see Figure 46 on page 156).
Sacrificial material
A
Segment of sacrificial material
r ~ t e e L tug
extensions
f r o m insert
rnaterial,to be
welded after
positioning
{ a ) Half-shell bracelet
Figure 46
welded after
welded to bands
( b ) Segmented bracelet
Bracelet anodes lor concrete-coated pipelines
Cathodic protection of offshore structures
173
The preferred combination of spacing and length is chosen taking into account:
.
If the chosen combination lies on the S, line (in Figure 46), the weight of anode
material required is being minimised. Conversely, there is a weight penalty on
long, widely-spaced anodes for which the end of life current requirement is the
determining factor.
.
.
The fewer the anodes the better (i.e. the wider the spacing the better) in terms of
anode fabrication and installation costs.
Practical limits on anode length are imposed by casting technology.
.
The necessity, for conventionally laid pipelines, for the spacing to be an integral
multiple of the average pipe joint length (usually 12 m). This requirement may
be unnecessary for pipelines laid by reeling or towed methods.
The desirability of maintaining a single type (i.e. length) of anode throughout the
length of pipelines divided into sub-sections for cathodic protection design
purposes.
On pipe without weight coating, and to a certain extent on weight-coated pipe,
provide the anode is not proud of the concrete, anode thickness, t, is also a
variabIe, up to the limits imposed by casting techniques and the practicality of pipe
handling. Varying t as well as 2, allows the designer to fine-tune his design even
further.
8.5.5 Attenuation of potential
Anti-corrosion coatings are intended to form an electrically insulating barrier
between the pipe wall and the surrounding electrolyte. Where this barrier breaks
down, there is a flow of current to the pipe surface. This influences the rate at
which voltage attenuates with distance from the maximum (most negative) potential
at the drain point to the anode. This attenuation therefore increases as coating
quality deteriorates.
The “throw“ of protection from an anode array is derived from the expression:
(8.15)
where
V,
V
4
G
= open circuit potential of unprotected steel (V)
= positive limit for adequate pipeline protection (V)
= pipe potential at drain point, i.e. the point of connection to the
anode (V)
= distance from drain point to which pipe is protected (m)
= longitudinal electrical resistance of pipe waH ( o h d m ) , which is a
function of steel cross section area and the resistivity of the steel
= electrical conductance of p i p e h e coating (ohm”/m).
The pipe potential at the drain point (V,)is either the open circuit potential of the
sacrificial anode material, for remote sacrificial anodes made less negative by
voltage drop along the connecting cable, or the negative potential limit for the steel
imposed by design requirements, whichever is the more positive.
174
MTD Ltd PubIication 901102
Thus for a given pipeline, the length of pipe protected by a single anode is 2lP,for
the anode throws current in both directions along the pipeline. It is a function of
the conductance of the coating, all other factors being constant, and I,, decreases as
conductance increases. Conductance of the coating is a maximum, and l,, a
minimum, at the end of pipeline design life, when coating breakdown is at its
maximum.
End of life conductance G =
-1
( V o - VJ
(8.16)
= final current (A) to be delivered to pipeline, From Equation (8.1)
= total length of pipeline (m).
I,
where
L
L
Thus i,, can be calculated from Equations (8.15) and (8.16). Figure 49 illustrates
the effect of attenuation on pipe surface potential, assuming protection by a single
anode.
-m
.-
c
c
c
W
0
n
(Y
.-a
a
/
I
I
I
I
I
I
/
Distance from drain point
l
1
I
I
w
Pratected length
no negative potential limit 1
1
I
Protected l e n g t h [ w i t h
negative potential Limit I
8
1
I
I
,I
I
I
I
I
I
I
I
OAnode
Figure 49
Attenuation of pipe material
Cathodic protection of offshore structures
175
8.5.6 Positioning of remote anodes
The pipe potential at the drain point changes as compared to the anode potential,
because of two effects:
.
Voltage drop along the cable and its connections at either end, which, for a given
cable size, increases with distance of anode array from the pipeline.
The "tension hill" effect, which changes the electrolyte potential close to the
anode array, with magnitude given by
where
I = anode array current output (A)
p
= electrolyte resistivity (ohm. m)
a
= distance of array from pipeline (m)
Clearly, the voltage drop from the tension hill effect decreases with increasing
distance of the remote anode from the pipeline.
The positioning of a remote anode is therefore a compromise baiancing these two
effects.
8.6
ANODE DESIGN AND ATTACHMENT
As well as satisfying the current output criteria and (for sacrificial anodes) the mass
criteria for the cathodic protection system, the design of anode adopted for a
particular pipeline and the proposed method of attachment o the pipe have to be
compatible with:
pipeline coating materials (anti-corrosion, weight-coating, nsulation coating),
dimensions and application sequencdmethods
.
.
onshore prefabrication, if any
offshore and subsea construction activities.
The discussion here is confined to pipeline cathodic protection systems. Anodes
for subsea structures are similar in design and attachment methods to those
described in Section 6 section for jacket structures.
8.6.1 Bracelet anode shapes
The two principal types of bracelet anode are the twin half-shell anode and the
segmented bracelet anode, shown in Figure 48. Either is technically acceptable for
most applications, but the simpler segment shapes tend to be more easily cast than
are half shells, particularly in Iarge diameters and thick sections. However,
segmented bracelets tend to include more insert and support steel sections, and
hence more complex steel fabrication, than do half shells. The latter therefore tend
to be preferred when casting them is straightfonvard. Maximum lengths of half
shells tend to be around I. m, but individual segments may be cast in longer lengths
if desirable. Alternatively, anodes may be "doubled up" in areas requiring greater
mass.
176
MTD Ltd Publication 901102
if desirable. Alternatively, anodes may be "doubled up" in areas requiring greater
mass.
Steel insert material for braceIets consists of welded cages of bar, strip and plate
intended to provide an electrical connection to the pipe and to hold the consuming
anode material in a cohesive mass while maintaining its electrical continuity to the
pipe. The utilisation factor, u, is that proportion of anode material which is
consumed, uniformly, before electrical continuity of the remainder with the steel
insert (and thus with the pipeline) is lost.
Bracelet anodes for weight-coated pipelines are usually designed to be flush with
the outer surface of the concrete, so that &hepipeline presents a uniform diameter
to the pipe tensioners, stinger and other handling equipment on lay barges. An
anode proud of the concrete is likely to suffer considerable physical damage during
pipelaying. Occasionally, the anode surface may be recessed from the concrete
surface if it is found necessary to increase the ratio of current output to net weight.
In order that consumption is restricted to the outer surface of the anode, the gaps
between segments and half shells are filled with a mastic material, as is the gap at
either end between anode and weight coat. T h e inner and edge surfaces of each
segment or half shell are also often coated, with coal tar epoxy or similar, again to
prevent consumption of these surfaces. This is important because the utilisation
factor, u, used in the design calculations, and the placement of steel insert material
to achieve that value of u, both assume consumption only from the outer radial
surface.
Typical placement of a half shell anode over a coal-tar enamel and weight-coated
pipe is shown in section in Figure 50.
---7\
!!
1
Mastic infill
Figure 50
Pipe w a l l
Anti-corrosion coatinq
Anode material
,Concreke
coating
Typical alfachment of bracelet anode to concrete-coaled pipe
Cathodic protection of offshore structures
177
Bracelet anodes for non-weight-coated pipes may be ofa similar design to those
described above, but more usually they are tapered at either end in order to ease
their passage through handling equipment, stinger and trenching machine without
damage. In addition, because it is the small size pipelines which tend to have no
weight coat, their bracelets are often hinged on one side for ease of installation.
This may be particularly important if pipe handling (e.g. reeling of pipe) dictates
that the anodes should be installed on the pipe quickly on the lay vessel,
immediately before passage through the stinger. In order to preserve the utilisation
factor, the inside and edge surfaces of the anode should be coated. A typical anode
is shown in Figure 51.
Bolts
Sacrificial material
Elevation
Figure 51
Section
insert
material
Typical clamped, tapered anode bracelets
8.6.2 Bracelet anode attachment
Attachment of the bracelet anode to the pipe has to fulfil two functions:
provision of a permanent, low resistance electrical connection between anode and
Pipe
prevention of anode movement, which may rupture the electrical connection,
while the pipe is being handled, laid and trenched.
On concrete-coated lines, bracelet anodes are invariably installed in a factory or
yard environment onshore. Each anode is positioned in two halves around the
pipe at the centre of the pipe joint, The halves are then fixed in position by
welding or bolting to each other. Movement along the axis of the pipe is prevented
by the adjacent concrete coating.
'
178
Conversely, anodes on pipe without weight coating need to be prevented from
movement by firm attachment to the pipe. This may be achieved eitber by the two
anode halves forming a hinged clamp which can be bolted on firmly, or by fillet
welding of the anode insert material to the pipe, or, preferably, to a doubler plate
pre-installed on the pipe joint. The former solution, being quicker, should be used
if the anodes have to be installed offshore during the pipe laying operation. Weld
runs along the longitudinal a x i s of the pipe should be avoided.
MTD Ltd Publication 90/102
wire
=/-
'Strip of
anode
Polychloroprene
Figure 52
material
Proposed pipeline sirip anode
If an anode is welded to tbe pipe or doubler plate, tbe mechanical artacbment
doubles as the electrical connection. In other cases, short length electrical cables
(at least two per anode, for contingency in case of damage) are run between the
anode insert material and the pipe wall. The cable connection to the steel insert
material is made by brazing andor welding while that to the pipe wall uses thermit
welding.
Thermit welding is chosen because, thanks to its low penetration and heat input
into the pipe wall, it has minimal effect on material properties, and because of its
repeatability: once the procedure has been qualified and the weld parameters set
there is little scope for operator error or other variability. Slack is left in the cables
so that minor axial movement of the anode does not cause one or both electrical
connections to break. A typical cable arrangement is shown in Figure 50.
Alternative methods of attachment are becoming available which are all claimed to
have technical benefits over thermit welding. These include pin brazing, explosive
spot welding and friction stud welding.
During and after anode attachment, any coating damage or other bare steel areas
on pipe, doubler plate and anode insert material are re-coated using a compatible
repair material, in order to maintain the initial high integrity of the pipe coating.
Thermit welding to duplex or other stainless steel pipelines is not desirable because
the process induces an undesirable microstructure in the steel. ConsequentIy, the
attachment of bracelet anodes to duplex pipes inevitably involves the use of welded
doubier plates, which may themselves be duplex material or an austenitic stainless
steel such as AISI 316L. One of the alternative processes discussed above may be
used.
Cathodic protection of offshore structures
179
8.6.3 Strip anodes
An alternative close-fitting anode design which is under development is the strip
anode integral with a thick film anti-corrosion corrosion coating (specifically
poIychloroprene): see Figure 52. The attachment of each strip to the pipe is by
thermit welding, and because anode attachment is part of the coating process it can
be carried out only at the coating plant. Anode strips can be extruded, so the
length of these anodes may in theory approach that of a pipe joint. One of the
advantages claimed for the strip anode system is that, unlike bracelets, it is flexible
enough to be used on a reeled pipeline.
8.6.4 Remote sacrificial anodes
A remote array of sacrificial anodes has a specified minimum current output and a
minimum mass of consumable anode material. Other than that, the anodes may
take any form consistent with being mounted on some sort of structural steel sled,
the cathodic protection requirements of which need also be taken into account
during the design of the anode array. The sled should be easily installed offshore
by crane, and robust enough to withstand accidentai impact from trawls, etc.
Computation of anode array current output should take into account interference
effects from the close proximity of anodes, the totaI output being much Iess than
the sum of the notional outputs of each anode. An array could consist of a
structural frame covered with bar or plate anodes, or a length of tubular with
several bracelets mounted on it.
A sled may be specially designed to resist self buriaI, if burial were to reduce
current output unacceptably. Conversely, it may be deliberately buried for extra
physical protection if the reduction in current output is sustainable.
The cable running to the pipeline may need to be buried and/or armoured for
protection, and two cables may be run to gwe 100% redundancy. Connection to
the pipe may be achieved most easily by using a clamp incorporating several sharp
set screws tightened hard into the pipe wall. Such a system has to be regularly
checked by divers for tightness. The use of welding under wet or hyperbaric dry
conditions to make these connections is feasible, but it needs to be carefully
qua1i fied .
8.6.5 Impressed current systems
On land, anode installations take the form of conventionally designed groundbeds,
with buried cable runs to transformer rectifiers and thence to the pipeline, where a
thermit welded or other type of welded connection is made. Groundbed current
output and capacity are determined by the size, shape, mass and spacing of
individual anodes, and the size and resistivity of the groundbed material.
Offshore, remote impressed current anodes are cabled back to transformer
rectifiers located topsides on the offshore platform, the drain connection to the
pipeline being located either above water, or possibly beIow water on the riser, the
cabling baving been instalIed at the jacket fabrication yard. Offshore impressed
current anodes could in theory be groundbeds similar to those at landfalls, but
180
MTD Ltd Publication 90/102
subsea excavation is time-consuming and expensive, and the anodes are difficult to
maintain. Instead, platinised niobium or platinised titanium anodes, suitable for
operation in sea water (but not when buried) have ia the past been used, mounted
in arrays on sleds designed to resist natural burial. Alternatively, some protection
to a pipeline might be afforded by jacket-mounted impressed current anodes.
8.7
PIPELINE INTERFACES
8.7.1 Risers (see also Section 6.4.2.3, page 104)
The subsea portions of risers are protected according to the same criteria as
pipelines, but their unique nature, often in terms of coatings and fabrication
method, and sometimes temperature, demands that a cathodic protection subsystem should be designed specifically for them. It is common for the operator to
isolate the riser electrically from the offshore platform by insertion of an isolation
joint (see Figure 53) in the riser, above water, and to line all riser clamps beIow
that joint with poIychloroprene in order to eliminate the possibility of metal-tometal contact. The purpose is to prevent the pipeline and riser cathodic protection
system from being drained through any inadequacy in the platform cathodic
protection system, and vice versa. As a further precaution against drainage, it is
common to over supply bracelet anode material on risers, and on tbe pipeline
within 0.5 km of the platform, by a factor of 1.5 to 2.
Conversely, an alternative approach is to ensure good electrical contact between a
riser and steel jacket, and to protect the riser b y using the jacket cathodic
protection system. Xf current distribution is good, interference effects between riser
and jacket should be eliminated, but there is a risk of poor current distribution and
drainage from pipeline to jacket or vice versa.
Interference effects at risers are practically non existent if both pipeline and jacket
are protected by zinc or aluminium-alloy sacrificial systems. If an impressed
' .
Figure 53
.
Typical monobloc isolation joinf for pipelines
Cathodic protection of offshore structures
181
current system is present, considerable care and fine tuning are needed to prevent
interference in the form of accelerated corrosion resulting from electrical shorting
through sea water. The imposition of a negative potential limit around -1.1 V (wrt
Ag/AgCl) on steel in sea water may be beneficial in this respect.
8.7.2 J-tubes
J-tubes form part of the secondary structure of an offshore platform, and their
exteriors are protected by the platform cathodic protection system.
Cathodic protection of pipelines within J-tubes is usually not considered possible,
because:
Anodes mounted on the pipe are probably sheared off during pull-in.
.
PipeIine anodes outside the J-tube cannot "see" inside the J-tube, and so are
unable to throw protective current inside.
Anodes within the J-tube have limited throwing power (say five times the tube
diameter at the most) because of the confined space (c.f. cathodic protection
inside seawater piping).
The pipe inside the J-tube is likely to have suffered coating damage during pull-in,
and so it is potentially exposed to a highly corrosive environment, particularly in
the tidal zone. The most permanent solution is to block the lower end of the Jtuhe and treat the contents with corrosion inhibitors.
Nevertheless, at a J-tube pull-in, the pipeline remains unavoidably electrically
continuous with the J-tube and hence with the platform.
8.7.3 Flanged joints
Properly mated flanges in pipelines are normally assumed to provide adequate
electrical continuity. Nevertheless, it is considered good practice when designing
sacrificial systems to ensure that each flanged spool, perhaps including valves or
other equipment, has sufficient anode material to protect it independently of the
adjoining sectors of the line,
8.7.4 Wellheads and templates
It is common to wish to isolate a pipeline from a wellhead or template structure,
which has its own cathodic protection system, to avoid drainage of the pipeline
cathodic protection system, particularly to underprotected piles and well casings.
This desire must often come secondary to the design of the pipeline tie-in system,
which may not usually allow for isolation. The high pressure flexible pipes often
used for tie-ins are all electrically continuous from end fitting to end fitting, and so
do not act as isolation joints. As a result, continuity at these interfaces should be
assumed, and, as a precaution, the pipeline cathodic protection system should be
overdesigned in the vicinity of the wellhead.
182
MTD Ltd Publication 90/102
8.75 Single point moorings
Low-pressure submarine hoses installed between a pipeline end manifold and a
single point mooring (SPM),or other loading systems, may be provided either
electrically continuous or discontinuous. It is up to the owner’s safety approach
and loading procedures whether or not he wishes the S P M to be isolated from the
pipeline.
8.7.6 Landfalls
The interface between the cathodic protection system of a submarine pipeline and
that of a land pipeline, provided the submarine pipeline is not designed to be
protected by the land system, is usually marked by an isolation joint suspended in a
dry inspection pit.
8.7.7 Pipeline crossings
Crossing submarine pipelines should be vertically separated by at least 0.5 m,
according to pipeline design codes, using bituminous mats or similar if necessary.
This requirement is principally intended to eliminate physical damage through
contact during installation and operation, but the separation is sufficient to
eliminate any possibility of interference effects hetween two pipelines protected by
a zinc or aluminium alloy cathodic protection system.
If one of the pipelines is protected by impressed current, consideration should be
taken of the desirability of providing a cable earthing strap between the two lines,
or else the potentials of the two lines should be regularly monitored at the crossing
point and equalised if necessary.
8.7.8 Dissimilar materials at interhces
Where duplex stainless steel risers are mounted on low alloy steel jackets in low
alloy steel clamps, or duplex piping is present on subsea templates, it should not be
assumed that the provision of cathodic protection to all components by the same
system eliminates problems of galvanic corrosion between dissimilar metals. The
duplex stainless steel pipe should be rigorously isolated from surrounding low alloy
steelwork, using high integrity coatings, polychloroprene lining of clamps and
supports, etc., while retaining electrical continuity with the cathodic protection
system.
8.8
MONITORING OF PIPELINE CATHODIC PROTECTION
8.8.1 Introduction
As applied to pipelines, monitoring of cathodic protection systems (see Section 9
for genera1 discussion) can take many forms:
the monitoring of impressed current anode outputs, which is an activity for landbased or platform-based instrumentation only
Cathodic protection of offshore structures
183
the monitoring of steel potential through permanent hard-wired reference cells
placed adjacent to the risedpipeline (feasible only at platforms) or through hardwiring back to conventional test posts on land
measurement of steel potential, current density or anode current output through
periodical subsea intervention by divers, remote-operated vehicles (ROVs) or
towed instruments
remote permanent steel potential monitoring devices.
Of these possibilities, only t h e last two are relevant to locations away from landfalls
and offshore platforms
8.8.2 Monitoring by divers
The monitoring of steel poten tial by divers using handheld contact probe reference
cells is discussed in Section 9. This method may be used on subsea structures and
pipelines wherever bare steel is accessible, but in practice this is restricted by:
water depth. Limits are imposed by human physiology and operator intervention
philosophy
efficient use of vessel resources. Because pipelines are more cheaply inspected
using ROVs than divers (in terms of time and inspection spread costs), divers
tend to be used for inspection and maintenance only around platforms and the
more complex subsea structures such as wellhead templates, valve assemblies, or
in the vicinity of landfalls where water depth is too shallow for ROV deployment.
Should ROV inspection reveal deficiencies in the cathodic protection system, it is
common practice to employ divers (water depth permitting) to perform a more
detailed inspection programme prior to establishing the need for any remedial
action.
8.8.3 Monitoring by ROV
Inspection of cathodic protection systems at pipelines and risers is frequently
carried out using ROVs. Suhsea riser inspection is usually carried out
simultaneously with platform inspection by small-sized “eyeball” ROVs employing
proximity-type reference electrodes to measure steel potential. Proximity-type
instruments are used in preference to contact instruments because the precision
required to place the latter on a small area of bare steel is difficult to achieve for a
free-swimming ROV, always assuming that marine growth permits access to the
bare metal, and also because contact probes are frequently damaged on impact.
Use of a proximity-type probe requires a hard-wire connection to the platform
topside at a point electrically continuous with the riser. Thus if the riser is isolated
from the platform, the hard-wire connection for jacket inspection is inappropriate
for riser inspection, and it has to be moved to a point on the riser below the
isolating joint.
MTD Ltd Publication 90/102
Inspection of the cathodic protection system of a pipeline is normally carried out as
part ofa regular programme of video and sonar inspection of pipelines by ROV, in
h e With certifymg authority requirements. Several techniques are available which
may be used siogly, sequentially or, ROV capability permitting, concurrently, to
measure pipeline potential and (via measurement of electrical field gradients)
anode output. These techniques, illustrated in Figure 54, are:
The use of a proximity-type reference cell mounted on the ROV, with a hardwire connection from the support vessel to one end of pipeline (i.e. to the
offshore platform). This is directly analogous to the measurement of potentials at
risers mentioned earlier. O n all but the shortest pipelines, however, the duration
of the inspection, a matter of days, makes the trailing wire a hazard, and hence
this technique is seldom used with ROV.
Remote reference Celt
Remote reference cell
on umbilical
( a ) Potential survey using remotely-operated vehicle with remote reference Celt
Sea bed
To support
ve sset
Re rn otely - operated
Twin reference cells
.,Pipeline
Sea bed
( b l Field gradient measurements using remotely-operated vehicle
Figure 54
Monitoring of pipeline cathodic protection systems, using remotely-operated
vehicle
Cathodic protection of offshore structures
185
The use of two reference cells, one ROV mounted as before but the second
immersed "remote" from the pipeline, usually suspended from the support vessel
or attached to the ROV umbilical. Pipe potential is indicated by the difference in
readings obtained from the two reference cells, but frequent check calibration of
the system (usually against sacrificial anodes on the pipeline) is necessary.
The use of two reference cells mounted on the ROV, the relative positions of
which are known precisely, allows the potential field gradient adjacent to the pipe
or an anode to be measured. Pipe potential, current densities being applied to
the pipe, and current densities being emitted from anodes may all be calculated,
using various data processing techniques, from these measurements. This
technique can be combined with the first two techniques to provide direct pipe
poten ti aI measurements offshore.
The above techniques are suitable for use when the pipeline is either exposed o r
buried.
8.8.4
Monitoring b y towed instruments
The hard-wire and "remote" reference cell techniques used with ROVs may also b e
implemented using an unpowered instrument or "fish" towed along the pipeline by
a surface vessel. The speed of inspection is much increased over the use of ROV
but because the distance of the towed unit from the pipeline is known only
inaccurately (under normal circumstances it is much further from the pipeline than
an ROV-mounted instrument), the measured potential is in effect a value averaged
out over an appreciable pipeline length. Very local effects therefore tend to be
suppressed, so that a coarse measurement of pipe potential is all that can be
obtaincd. The techniques are shown in Figure 55.
8.8.5
Remote permanent monitoring devices
These devices consist of one or more reference cells attached to the pipe from
which the pipe potential is obtained hy means of a n acoustic transponder, which
transmits when interrogated by a transducer immersed from a platform or vessel.
[ a ) P o t e n t i a l survey using towed instrument and trailing hard wire
Pipeline
reference cell
Towed reference Celt
Ib ) Potential survey using towed instrument and remote reference cell
Figure 55
186
S e a bed
S e a bed
Monitoring 01 pipeline cathodic proledion systems, using towed instruments
MTD Ltd Publication 901102
Such devices have been used with some success on offshore pIatforms where
frequent interrogation is possible. Fitment to subsea pipelines and structures is
much rarer, principally because the devices cannot be pie-assembled to the
pipeline as they can to a jacket, because they would be destroyed during pipeline
installation and because a vessel needs to be mobilised to interrogate them.
Acoustic monitoring devices may be particularly useful on subsea structures which
are helow the commercial saturation diving range, and to which ROVs can gain no
earthing cable contact for proximity-type reference cells.
8.8.6 Response and uncertainties
As discussed in Section 9, cathodic protection surveys are likely to be subject to
uncertainties and anomalies. Every observation which may help to explain usual
potential readings should be reported and, in the evaluation of results, corrosion
specialists should be consulted when irregular values are recorded.
At potentials less negative than -0.80 V (AgIAgCl), the steel is under protected.
Potentials less negative than -0.83 V should be considered as an early warning that
under protection may occur. If &hereadings show potentials less negative than
-0.83 V, additional measurements should be performed for each 0.5 m away from
this point untii potentials more negative than -0.83 V are read. These
measurements should be carried out in all relevant directions. The readings and
the extent of the area should be reported. Potentials on uncoated steel, measured
more than 3 m from sacrificial anodes, should normally not be more negative than
-1.02 V. Readings which are more negative may be erroneous, and they should be
repeated. I€ the second reading gives the same value, the measuring equipment
should be checked, and the reference electrode should be calibrated. All the
readings should be reported.
Potential readings of aluminium sacrificial anodes more negative than - 1.10 V, and
readings of zinc sacrificial anodes more negative than -1.05 V, may be erroneous.
If such negative values are measured, the measurement should be repeated. If the
second measurement gives the same value, the measuring equipment should be
checked and the reference electrode shouId be calibrated. All readings should be
reported. When their current output is high, anodes may be polarised to relatively
positive potentials. If the potentials of sacrificial anodes (any alloy) are less
negative than -0.95 V, the steel potentiai close to the anode should aiso be
measured.
Cathodic protection of offshore structures
187
Section 9 Operation, monitoring and surveying of
cathodic protection systems
Cathodic protection of offshore structures
189
9. Operation, monitoring and surveying of cathodic
protection systern
9.1
INTRODUCTION
Cathodic protection systems for fixed steel offshore platforms require significant
survey, monitoring and, in certain cases, control activities in order to ensure their
effective performance throughout life.
Section 2 indicates how factors such as weather conditions, caicareous deposits,
marine fouling and coating deterioration result in time-dependent changes in
cathodic protection requirements for a particular platform.
Sections 6 and 11 indicate that the design parameters for cathodic protection of
fixed steel offshore platforms have been changing during the past 15 years as our
knowledge of system performance has improved. Each such design change brings
the probability of improved performance but also the possibility of unforeseen
problems. The introduction of novel offshore facilities, tethered floating drilling
and production platforms, and subsea control and manifolding facilities brings new
design requirements for cathodic protection.
New locations for oil and gas development with modified environmental conditions,
such as extreme northern latitudes or locations such as Morecombe Bay, will result
in cathodic protection requirements different from those established by trial and
error for the southern basin and the northern sector of the North Sea.
These factors combine to present cathodic protection systems for fixed offshore
structures with demands for current and mechanical properties which vary with
geographic location and with time.
Overlaid on these dynamic requirements is the indication from Section 12 that the
quality of original cathodic protection designs is variable. Even recently executed
and relatively simple sacrificial cathodic protection systems are deficient in respect
of anode and current distribution. Those impressed current systems which are
successful reflect the highest standards on the part of both designers and operators
and the benefit of past experience. Without these advantages, the problems with
impressed current systems have been considerable.
It is these geographic, time-dependent and design variables which determine the
necessity for properly planned, executed and interpreted monitoring and surveying
of cathodic protection performance.
Throughout this Section "monitoring" refers to the collection and assessment of
data from permanent fixed location sensors, while "surveying" refers to data from
periodically deployed mobile sensors.
Cathodic protection of offshore structures
191
9.2
DESIGNREVEW
It is evident from Section 12 that a significant proportion of the deficiencies in
existing cathodic protection systems could have been identified and corrected prior
to installation of the platforms. The high costs of surveys, and particularly of
remedial action in repairing corrosion damage or retrofitting additional,
replacement or modified cathodic protection systems, dictate that these events
should be minimised. It is recommended that each cathodic protection design for
fixed offshore structures should be subjected to a design review, external to the
desigdconstruction project team prior to finalising the design and approving it for
construction.
An important feature of the design review should be to ensure feedback of past
operating experience with new designs.
The design review should in particular include, but not be limited to, current
distribution in complex and congested areas, electrical continuity provisions for
components such as piles, risers, caissons and clamps, provisions for repolarisation
after storms, and sufficient monitoring provision to provide data during and
immediately after storms.
9.3
INITIAL OR COMMISSIONING SURVEY
The initial or commissioning survey should be undertaken as soon as is possible
after placement of the offshore structure. Det norske Veritas(') calls for these
initial surveys to be carried out within:
.
.
1 month for impressed current systems
3 months for sacrificial anode systems, uncoated installations
12 months for sacrificial anode systems, coated installations.
The requirement to undertake a cathodic protection survey within 1 month of
pIacement of an offshore installation may be impractical, depending upon the type
of installation and other essential work. However, it does properly reflect the
importance of an early survey to detect areas of under protection or generally slow
polarisation. Both of these deficiencies are best rectified urgently, because the cost
of rectifying even quite minor corrosion damage at critical areas can be
disproportionately high compared with the cost of the original cathodic protection
system and of surveys or limited retrofits.
The initial survey should extend to 100% of the structure and its cathodic
protection systems. It should include:
A 100% visual inspection of the cathodic protection system
A representative steeYsea water potential survey of each vertical and each
horizontal frame of the main structure
A detailed potential survey of representative nodes on each vertical and each
horizonta1 frame of the main strncture
192
MTD Ltd Publication 901102
A detailed potential survey
of known areas of high complexity and/or high
surface area density, for example:
- conductor bays
- conductors (if installed) at mid points between frames
- pile guides
- pile sleeves
- a n y partly closed compartments or shielded areas
- inside uncapped piles.
A detailed potential survey, specifically using Iocal point contact for the
measurement (Figure 56), of areas of possible electrical discontinuity from the
main structure, for example:
- piles
riser, caisson and J-tube clamps.
A 100% visual and potential calibration of any fixed monitoring system.
-
A properly
designed monitoring system should meet the second of the above
requirements and, at least partially, the fourth requirement.
Priority should be given to items requiring high operational integrity, areas of higb
stress and any areas where steels of high strength (yield strength > 700 MPa) are
used.
The initial survey should determine any areas of deficient protection, a n y areas of
damage to the cathodic protection or monitoring system, and any items such as
piles or clamps which require electrical continuity bonding to prevent accelerated
corrosion.
The initial survey of the structure normally incorporates the equivalent survey of
the riser(s). It is important that these riser surveys should be undertaken with the
metallic connection of the measurement circuit connected to the t i w , on the
seaward side of any electrically isolating joint, to ensure that the measurements are
accurate even if the riser is intentionally or inadvertently electrically isolated from
the structure. The survey and monitoring requirements of pipelines and risers were
addressed in Section 8.
9.4
FMED MONITORING SYSTEMS
The Department of Energy "Offshore Installation: Guidance on Design and
Construction M(le) states that:
"6.1.2.4.(vi) The regular and frequent monitoring of steeYsea potentials preferably
by a permanently installed system is a minimum performance monitoring
requirement. In addition, anode current and/or cathodic current density
monitoring may be advantageous. The location of monitoring instruments is
important and should take into consideration:
Cathodic protection of offshore structures
193
vehick video
remoteiy-operated
vehicle video
I
Figure 56 Potential
194
survey
I
electrode
Circuit diagram
- local point contact
MTD Ltd Publication 90/102
areas least likely to receive full protection in terms of either potential or current
density
items requiring high operational integrity
highly stressed regions
areas on high strength steel structures most likely to suffer from hydrogen damage
if over-protected
sufficient representative locations to indicate general trends of levels."
Both Det norske Ventas(') and Lloyds(lm) mandate permanent monitoring systems
for impressed current systems, and both recommend them for sacrificial anode
systems.
Despite this guidance from the Department of Energy and others, plus the
experience reviewed in Section 12 revealing that fixed monitoring systems can be
reliable and that operators consider the data to be valuable, a significant proportion
of new structures are designed and installed without fixed monitoring.
In part, this is probabIy the result of operators' incomplete knowledge of the value
of the data available from such systems. The poor reliability of early systems has
prejudiced some operators, and many are now conditioned to more expensive,
extensive, periodic surveys than would be necessary if fixed monitoring were
instarled. A complaint of some corrosion engineers in the offshore operations
groups is that potential savings arising from better design of cathodic protection
systems (made possible by data from well monitored systems in the same
environment) are not achieved, because the project design teams for one
development do not incur additional expense for the henefit of a future
development project.
Valuable early confirmation of system performance can be obtained from a fixed
monitoring system. These data can be collected in the early weeks after system
placement (particularly if an acoustic linked system is employed with portable
interrogation, prior to the commissioning of any long-term fixed interrogation
system). It gives a record of polarisation (i.e. the time during which the structure is
under-protected while calcareous films are being formed).
Data collected from a fixed monitoring system during storms indicate the extent to
which the depolarising nature of storms results in under protection of the structure.
The magnitude of this storm depolarisation should be determined, in order that all
data from surveys by divers or ROVs (in good weather) can be assessed to
determine if any areas of the structure are liable to corrosion during and after
storms.
It is recommended that fixed monitoring systems, complying with the Department
of Energy guidance, should be provided for:
all systems with impressed current systems
-
all structures in new environments
Cathodic protection of offshore structures
195
all structures with designs significantly different from those previously used by the
operator in the proposed environment.
It is recommended that similar fixed monitoring systems should be given serious
consideration for:
representative structure(s) in all field developments
all structures for which the cathodic protection design parameters have been
modified from those previously used in that locality.
The available fixed monitoring systems fall into two categories: hard-wired systems
rely upon cables run from the submerged sensors to the topside measurement,
display and recording systems; acoustic-linked monitoring systems use local
measurement, with transducers to send coded messages through the sea water from
the submerged sensors to a transponder near the water surface. This is connected
to the topside interrogation, display and recording systems. The acoustic-linked
system transponder and interrogation unit may be portable, giving some advantages
in early deployment and flexihility. A disadvantage of such portable systems is the
difficulty of their deployment in storms and their intrinsic dependence upon
operator usage.
Hard-wired systems bave the advantages of simpticity. Section 12 confirms their
reliability if properly engineered. Most failures of early systems were because of
damage to cable conduits. However, in deep water, the high cost of properly
engineered cable conduits renders hard-wired systems more expensive than
acoustic-linked systems.
The acoustic-linked systems have the advantage of avoiding long cable conduits.
Early systems were unreliable, but recent systems appear to be satisfactory,
although placement of transducer/transpoaders in structurally complex areas has
resulted in less than ideal communication response. This is exaggerated by "noise"
generated by storms. Some systems have a memory capacity enabling significant
volumes of data to be collected and then interrogated intermittently. These may
have particular attractions for sea-bed facilities or small unmanned structures
where intermittent interrogation from an adjacent ship would be appropriate and
cost effective.
9.4.1 Reference electrodes
Irrespective of the system used for data transmission, fixed cathodic protection
monitoring systems utilise zinc or silver/silver chloride reference electrodes to
provide a standard connection to tbe sea water and to enable the steeYsea water
potential to be measured (Figures 57 to 60).
196
MTD Ltd Publication 901102
Figure 58
Acoustic-linked reference electrodel
transponder for acousfic-linked
monitoring system (courtesy Global
Cathodic Protection Lfd)
Figure 57
Hand-wired dual reference electrode
zinc and silverlsilver chloride
elements (courtesy Global Cathodic
Protection Ltd)
Figure 59
Hard-wired monitored anodes
(courtesy Global Cathodic
Protection Ltd)
- 0 . 5 - 0 - 4 - 0 . 3 -0.2 -0-1
Zn
I
I
I
I
I
I
0 *O 1 +0.2 +0'3 +0.4 +O-5 + 0 4 *O 7 + O 8 ~ 0 . 9+1.0 +I 1
I
I
I
I
I
I
1
1
1
I
I
I
I
I
I
I
I
I
1
l
l
1
I
-1.5 - 1 4 -1 3 -1.2 -1 1 -1 0 -0 9 -0.8-0.7 -0-6-0 5 -0 6 -0 3 -0.2 -0.1 0
Volts
Ag/AgCl
Ag/AgCI
1
Cu/CuSOL
I
1
I
-1.5 -1.6 -1.3 -1.2 -1-1 -1 0 -0 9 -0.8 -0.7 -0.6-0.5 -0 4 -0 3 -0.2 - 0 1 0
1
I
I
I
I
I
I
I
I
1
I
I
r I II II I I I I 1
-1.6 -1.5 -1.4 -1.3 -1.2 -1 1 -1.0 -0 9 - 0 4 -d.7 -0.6 -0.5 -0.4 -0 3 -0.2 -O.?
1
0
Volts
Note: Extracted f r o m CP1021"2! Apptles only at 20"C and in clean undiluted sea water
Figure 60 Approximate comparison of potentials using zinc, copperlcopper sulphate and
silverlsilver chloride reference electrodes
Cathodic protection of offshore structures
197
9.4.1.1 Zinc electrodes
These are normally machined castings of either high purity (99.99%) zinc to ASTM
B418 Type 2(@) or,with Iess accuracy, of lower purity zinc alloyed with aluminium
and cadmium in accordance with the US military standards for zinc sacrificial
anodes, MIL-A-18001J(61). Both types should he provided with a current drain to
ensure metal dissolution so that they remain active. The current drain shouId be
switched off (open circuit) prior to and during measurements.
Reference electrodes using ASTM €3418 Type 2 material with current drain
provision should be accurate to 2 20 mV for many years.
9.4.1.2 Silverfsilver chloride electrodes
A wide variety of silver/silver chloride reference electrodes is available. Their life
and performance characteristics vary significantly, and caution is recommended in
the selection of the appropriate type. It is important in hoth fixed monitoring and
mobile surve appkations that the correct Ag/AgCl cell is selected, recorded and
calibrated (1037.
The original "Admiralty pattern" Ag/AgCl cell (72) comprises a silver wire
surrounded with silver and silver chloride in a finely divided powder held within a
porous pot. These cells and more recent variations of them incorporating silver
chloride powder may be adequate for portable surveys (if of sufficient stability and
accuracy) but are not suitable for permanent use. Marine fouling, contamination,
and loss of the silver chloride powder result in early failure and/or inaccuracy.
More recent Ag/AgCl cells comprise a silver wire surrounded by a porous adherent
coating of fused and subsequently electrolysed siIver chloride. These modern
Ag/AgCl cells require no current drain provision and should be accurate to & 5mV
for more than 5 years.
Both old and modern Ag/AgCl cells rely for their accuracy upon the fairly constant
chloride ion concentration of fresh undiluted open sea water. In environments
where the sea water is diluted, such as estuarine locations or those enclosed seas
where the salinity is higher than normal, corrections are necessary to ensure
accurate measurements (Figure 61).
Another form of cell, often called the Ag/AgCl cell, is widely used in portable
surveys and in permanent monitoring of reinforced concrete structures above
ground. This cell contains the silver and fused silver chloride of the modern cells
described above, but these are surrounded by a solution of fixed chloride ion
concentration within a porous plug. These cells are not affected by variations in
salinity and are suitable for surveys, but they are of unproven longevity for fixed
monitoring applications in sea water, Depending upon the chloride concentration
in their internal solution, they may not conform with the conversion values in
Figure 60.
9.4.1.3 Dual electrodes
It is increasingly common practice for each permanent sensor to incorporate two
reference electrodes of zinc or Ag/AgQ, often one of each, so that cell to cell
accuracy checks can be incorporated into each measurement (Figures 57,58, and 59)
198
MTD Ltd Publication 90/102
Potential
i d s for steel in sea wafer of 25ohm cm
h respect t o the commonly used
reference cells are :
-0.80volts AglAgCL Ref.
- 0.85volts CulCuS0,Ref
- 0 - 7 9 volts SCE Ref
+0.24volts Zn Ref,
This is shown i n t h e Nomogram,which can be used t o express
potentials referred to one standard with respect to either of
two other standard half cells. As
Resistivity(ohm c m ) this relationship varies with water
10000 resistivity, the Nomogram is used by
constructing a line orrginating at the
water res 1st iv it y applicable through
the potential referred to the half
2000,
c e l l being used.The equivalent
potentials with respect t o the other
half cells are then at t h e intersection
1000
of that line and t h e respective scale
An example is shown for
25ohm cm a t -0 8volts AglAgCl Ref.
-
200
Figure 61
Correction for salinity when
-
using silverlsilver chloride electrodes in sea water (lo4)
9.4.2 Monitored anodes
All impressed current anodes should have their current monitored at their topside
power supplies. It is of value for representative sacrificial anodes to be monitored
in order to give data regarding current output and thereby their life. Data
collected under a variety of weather conditions enable better and often less costly
design parameters to be selected for subsequent cathodic protection systems on
other structures in the vicinity.
Sacrificial anodes are monitored by fabricating isolating joints in their support or
stand-off tubulars and installing a low resistance shunt across one of the isolating
joints. The current-related shunt voltage is relayed to the surface using either hardwired or acoustic-link techniques (Figures 58 and 59).
Cathodic protection of offshore structures
199
9.43 Monitoring of isolated items
Risers, caissons and J-tubes may be intentionally electrically isolated from the main
structure, but they require incorporation into any fixed monitoring system.
Reference electrodes placed on the main structure, but adjacent to an item such as
an isolated riser, can be used to monitor both structure and riser potentials, but
only if special cable connections are provided from the riser to the measurement
system. For hard-wired systems, a cable connection is required between the
topside measurement, display and recording unit to the seaward side of the
isolating joint. For acoustic-linked systems, in which measurement (but no display)
takes place in the subsea unit, a local cable connection is necessary between the
monitoring unit and the riser. In such cases, it may be more practical and reliable
to duplicate monitoring units, and to have one on the riser and one nearby on the
main structure.
9.4.4 Location of fixed monitoring sensors
Fixed monitoring is not intended to replace periodic mobile surveys but rather to:
supplement survey data, generally taken in good weather, with a measure of the
exteat of depolarisation during storm or winter swell conditions
reduce the exteat of mobile surveys to representative areas and areas indicated by
fixed moaitoring to be less we11 protected than other parts of the structure
provide a record of the early polarisation history of the structure against which
visual surveys of corrosion damage can be judged
.
provide, at low cost, repetitive data collection from fixed points in order to
enable trend analysis to disclose, at an early stage, any problems with the
cathodic protection
.
provide routine and coatrol data for any impressed current system.
In order to meet these requirements, the fixed monitoring sensors, reference
electrodes and monitored anode should provide representative steeYsea water
potentials from each vertical and each horizontal frame of the main structure (see
pages 191 and 192) as well as representative potentials at known areas of high
complexity andor high surface area density such as:
conductor bays
conductors
.
.
pile guides
pile sleeves
any partly closed compartments or shielded areas
.risers, caissons and J-tubes.
a
200
areas of high strength steels (yield strength > 700 MPa).
MTD Ltd Publication 901102
9.4.5 Frequency of monitoring
In the early years of operation, the monitoring of a fixed offshore installation
should be sufficiently frequent to meet the requirements of Section 9.4.4. These
will vary from structure to structure, and they are determined by the nature of the
structure, its cathodic protection system, and the extent of past experience with
similar systems in the same environment.
Subsequently, if the cathodic protection system has performed satisfactorily, only
representative and trend analysis data are required.
Typical frequencies of monitoring might be:
daily for first 12 months
alternate days for 2nd and 3rd years
weekly thereafter
daily current and voltage measurement of impressed current power supplied,
throughout life.
9.4.6 Handling of monitoring data
As a minimum requirement, all fixed monitoring systems should produce hard copy
(paper) records of the collected data (Figure 62). These should be routed to the
corrosion engineer or equivalent responsible person charged with interpreting the
data, detecting trends from it, and issuing regular reports on the performance of the
cathodic protection system.
The volume of such data dictates that best use of the data can be made if they are
stored on computer, and if a specific database with spread sheet and graphics is
available for rapid production of out-of-limit reports, trend analysis and graphical
presentations.
Computer entry may be manual from the hard copy, but it may be more
economically achieved by direct link to the data logging facilities available in most
modern facilities or by a dedicated data store dumped to a portable computer
compatible with the onshore database system.
The records of the fixed monitoring system should be supplemented by the
necessary inspection of critical areas for fatigue or impact damage. It is emphasised
that data from fixed monitoring systems are only of value if assessed by the
corrosion engineer, or responsible person, and if decisions for action accrue from
the assessment. Appropriate action may range from a reduction in future survey
requirements to investigative surveys and possible repair of cathodic protection
systerns.
Cathodic protection of offshore structures
201
Figure 62
Micro-processor controlled
monitoring system topside display
unit - produces hard copy (courtesy
Global Cathodic Protection LfdJ
9.5
Y
PERIODIC SURVEYS
9.5.1 Frequency of survey
Periodic cathodic protection surveys are, in part, integrated into the mandatory
periodic sume s of the certification authorities. The Lloyd’s Register of Shipping
Draft RuIes(ld call for cathodic protection surveys only at biennial surveys and, for
impressed current systems with mandator fixed monitoring, at alternate biennial
surveys. Det norske Veritas recommend$) annual potential surveys on fixed
structures and subsea installations, the extent of the surveys to be dependent upon
the results of previous surveys.
202
MTD Ltd Publication 901102
However, without a monitoring system on a fixed offshore facility, the oniy data
regarding the efficacy of cathodic protection come from potential surveys and
visual inspections for corrosion damage. Considering the past performance of
cathodic protection systems, the corrosion rate of under-protected steel in sea
water, and the time and high cost of remedial action, an interval of 2 years hetween
such data collections is not considered to be appropriate. Similarly, the proposed
interval of 4 years between potential surveys for structures with impressed current
systems and mandatory monitoring systems appears unduly long.
O n the other hand, it seems excessive to require annual potential surveys of
structures fitted with detailed cathodic protection monitoring systems and having
an established record of good performance and no corrosion. The following
recommendations are made:
The incorporation into all diving or ROV activities of a topside metallic contact
and a system for local measurement of the steeYsea potential (See Figure 56).
The potential data should be overlaid on the video record, and any values outside
a range set by the corrosion engineer, typically -0.85 to -1.15 V AdAgCl, should
be reported to the corrosion engineer for assessment.
Structures with operational fixed monitoring systems complying with Section 9.4.4
to be surveyed:
annually for the first 3 years
- thereafter biennially if no areas of under protection are detected.
Revert to annual surveys if any areas of under protection or corrosion damage are
detected by survey or monitoring.
Structures without operational fmed monitoring systems complying with Section
9.4.4 to be surveyed annually.
-
9.5.2 Extent of survey
It is suggested that periodic surveys should be in such detail that the data from
them, from previous surveys, and from any fixed monitoring system, will together
detect any areas of under protection. Each survey should include:
a representative steeYsea water potential survey of each vertical and each
horizontal frame of the main structure
a detailed potential survey of representative nodes of the main structure selected
from the above, or previous data as being the least well protected nodes on the
structure
a representative steeYsea water potential survey of known areas of possible or
actual under protection, namely:
- conductor bays
- conductors, at mid points between frames
pile guides
- pile sleeves
- any partly closed compartments or shielded areas
- inside uncapped piles
- any areas of high stress and any areas where steels of higb strength (yield stress
> 700 MPa) are used
.
-
Cathodic protection of offshore structures
203
- any areas of known mechanical damage, by impact or fatigue.
a representative steelkea water potential survey, specifically using local point
contact for the measurement (Figure 56 on page 194) of areas of possible
electrical discontinuity from the main structure for example:
piles
- riser, caisson and J-tube clamps.
a 50% visual inspection and potential calibration of any fixed monitoring system,
such that any two consecutive periodic surveys Will survey 100% of the
monitoring system
a 100% visual and full operational check of all topside impressed current or
monitoring equipment and all topside isolating or continuity bond provisions.
-
.
9.5.3 Survey procedures and supervision
AIl survey procedures should be reviewed and approved by the corrosion engineer
of the operating company or equivalent person charged with interpreting the data.
The initial stages of each survey should be supervised by the operating company's
corrosion engineer or by a corrosion specialist acting on his behalf.
Typical deficiencies include poor calibration of equipment (including poor
calibration procedures provided by manufacturers), incorrect selection of the
locations for potential measurements (predominance of measurements at or near
anodes and incorrect selection of contact location),poorly-made contact point
topside, and surveys on risers, piles or clamps undertaken with structure
connections instead of local point contacts. Such errors in survey procedures may
result in undue confidence in the ability of the cathodic protection system to
prevent corrosion of certain areas of the structure, or they may result in retrofits
being executed unnecessarily.
Calibration of surve equipment should be in accordance with Det norske Veritas
recommendations('"). In particular, before and after each dive, the entire
measurement circuit should be confirmed for accuracy by check measurements
using portable voltmeters and standard reference electrodes.
Topside connections should be used only for surveying main structural members
and should be checked daily, confirming that the resistance between the connection
and an identical adjacent connection is less than 0.2 ohm. The connections should
be made to bright metal, and they should be protected from moisture.
All ancillaries which might be electricaliy isolated from the structure should be
surveyed with local point contact devices, or confirmed as continuous with the
structure prior to using topside connections. Confirmation of continuity can be
properly made only by comparison of topside contact measurement with local
contact measurement at the same location. The steel sealwater potentials measured
should agree within 5 mV if the same reference electrode is used (Figures 56 and
63).
All such setting up and calibration procedures should be fully documented.
204
MTD Ltd Publication 90/102
Surface meter
.- ... .--
contains only reference
Note:This technique isonly suitable for surveys of
the main structure.Surveys of piles,clamps
and other possibly discontinuous items must
be by local point contact ( Figure 56 1 or must
have specific metaliic contact provisions made
Figure 63 Potential survey - topside metallic coniact
9.5.4 Particular requirements
4.5.4.1 Interaction with adjacent cathodicprotection systems
The risk of interaction exists where sufficient electrical continuity is not achieved.
This risk should be considered when knowing the extent and frequency of
monitoring and surveying, particularly of risers but also of clamps and piles.
Interaction of a catbodic protection system with an adjacent structure, pipeline,
riser or component can occur if the various items are not sufficiently electrically
continuous. Examples are piles which are not sufficiently well bonded to pile
sleeves, and risers with pipelines in close proximity to seabed impressed current
anodes intended to protect the structure.
Interaction causes accelerated corrosion, localised at the point of current discharge.
Failure may occur in months, for the discharge of only 1A for 1 year consumes
approximately 10 kg of steel. This discharge may be localised at a singIe coating
defect on a riser.
Cathodic protection of offshore structures
205
Survey specifications and scope of work should particularly address possible areas
of interaction effects, including detailed potential surveys, using local point contact
where appropriate. Where the possihle sources of interaction currents are
impressed current anodes of high current output, these areas should be surveyed in
each survey. The surveys should concentrate on areas of first or close proximity
between members or structures which are, or may he, electrically discontinuous.
9.5.4.2 Intemption of impressed current system for diving operalions
One problem with impressed current systems, mentioned in Section 12, is the
practice of switching off structure-mounted anodes for safety reasons while divers
are working in their vicinity. Switching off the anodes not only removes the
protection potential from the steel, but it may also lead to accelerated deterioration
of the anode itself, as well as interfering with monitoring of the CP system.
Research on the phenomena concerned has been described by M~ulton(''~)and as
a result of these studies the Diving Inspectorate of Department of Energy issued a
Safety Memorandum in 1985('06). This contains the following statement:
"1. A reassessment of underwater electrical safety criteria suggests that impressed
current anodes do not constitute a hazard to divers providing that the voltage at
the anode does not exceed a nominaI 24 volts DC.
2.This implies that if the power is derived from a rectified AC source, adequate
protection must be provided to trip the supply if
(a) the higher primary voltage breaks through to the secondary circuit; and
(h) the ripple on the rectified DC exceeds 5%, for example due to phase failure.
3. Provided that these precautions are taken impressed current anodes need not
be switcbed off when divers are working in their vicinity. However, operators
may consider reducing the voltage to 6 volts."
This statement is backed up by the Code ol Practice for the Safe Use of Electricity
Under Water(1m),which also recommends the use of physical barriers to prevent
divers entering areas where hazards may still exist.
Where there is any doubt as to the possihility of electrical hazards from systems
(e.g. those with higher voltages or a significant ac component), it is recommended
that steehea potential surveys should be undertaken with ROVs. This should
enable the characteristics of the cathodic protection system to be determined under
operating conditions, before divers are used.
The overall aim should be, whiIe maintaining diver safety, to keep switch-off
periods to a minimum and wherever possible to take the option of reducing the
voltage, but to obtain cathodic protection survey data with the cathodic protection
system operating normally, if necessary by using ROVs.
9.5.5 Handling and recording of survey data
The quality of data presentation, by diving, ROV and other survey contractors, and
by the operators themselves, is now of a high order. However, it is not always clear
206
MTD Ltd Publication 90/102
that a sufficiently critical review of cathodic protection survey data is undertaken
b y either the contractors or some operators. The integration of fixed monitoring
and periodic survey data into one database, and the regular production of out-oflimit reports, trend analysis and illustrative graphical presentations is strongly
recommended.
The operator's corrosion engineer or equivalent responsible person, assisted if
necessary by an external specialist, shouid have a detailed record of the developing
performance of the cathodic protection system for every offshore facility under his
control. This record should be used in conjunction with the 0 erations Manual Maintenance specified in the Department of Energy Guidance . It is evident
from the work undertaken to update Section 12 that, although most of the
descriptive data and records stipulated in the guidance notes are available from
various sources within operating companies, the stipulated single document
Operations Manual - Maintenance relating to cathodic protection and monitoring
systems is not being prepared by the design contractors or the project teams, as part
of the document package generated at time of construction.
Ke,
It is recommended that design contractors responsible for the design of cathodic
protection systems for offshore faciIities produce the Operations Manual Maintenance as stipulated in the Department of Energy Guidance, for it forms a
record of the installed system which is valuable when interpreting survey data.
The extent of periodic surveys may be reduced in accordance with the judgment of
the corrosion engineer if data from previous surveys prove that the levels of
protection are adequate and stable (see Section 9.5.1).
9.6
ASSESSMENT OF REQUIREMENTS FOR MODIFICATION OR RETROFIT
Areas of under protection because of incorrect design, failed components, structure
modification, or extended life may be determined by reviewing the cathodic
monitoring or survey data andlor from the results of other survey activities. Data
quaIity should be assessed and, if necessary, additional confirming surveys should
be instigated.
These surveys may, in addition to the steeYsea potentials collected under normal
circumstances, also collect data on anode current outputs and current density data
from both anodes and cathodes.
Anode current output can be surveyed by using magnetic flux measurement clamps
around the anode stand-off tubulars. Cathode and anode current density can be
determined by surveying field gradients around the structure, using multiple
electrode arrays('08). All these data may be valuable in determining the cause of
the deficiency and the extent of any necessary modification or retrofit. Once the
requirement is determined, it is worth devoting significant engineering and
planning effort to minimising the number and installation time of any retrofits,
because this allows large cost savings in the offshore installation programme.
Mathematical modelling of survey data andlor novel retrofit anode shapes or
distributions may be valuable at this stage (see Section 5).
Cathodic protection of offshore structures
207
9.7
CONCLUSIONS
The integration of fixed monitoring with periodic surveys, both properly specified,
supervised and interpreted, can result in significant savings in offshore inspection
costs, significant savings in future cathodic protection systems, and the early
detection of under protection.
208
MTD Ltd Publication 90/102
Section 10 Structural aspects of cathodic protection systems
Cathodic protection of offshore structures
209
10- Structural aspects of cathodic protection systems
10.1
INTRODUCTION
Certain aspects of cathodic protection play a role in determining structural details
adopted for offshore platforms. Extensive usage of cathodic protection systems
has occurred in the area of fixed steel platforms (jackets) and this Section
discusses this aspect.
The geometry and framing arrangements adopted for b e d steel platforms are
governed by such major considerations as:
the water depth
.
.
the function of the platform and consequently the weight and disposition of the
topside load to be supported
the seabed soil conditions
the wave heights and other environmental data.
After selection of an arrangement and design of a structure which initially satisfies
these criteria, the need arises to examine the impact of the cathodic protection
requirements on the proposed design. At this stage, the cathodic protection
requirements are not usually a governing feature of the main structural selection
process, but they increase in importance as details are resolved prior to the
structure being committed to the construction stage.
The main features of the cathodic protection system which affect the structural
engineering are:
the location of sacrificial, impressed current and monitor anodes
.
.
.
.
10.2
the implication of the additional weight of the cathodic protection system
the increase in drag forces from the presence of the cathodic protection system,
and the demand that this places on the structure to resist them
the details adopted for attaching the cathodic protection systems to the
structure.
LOCATION OF ANODES ON JfIXED STEEL PLATFORMS
10.2.1 Anode location criteria
A number of structural considerations require the engineer to seek a compromise
between an even spread of anodes providing uniform cover to the structure and
the need to avoid placing anodes in unsuitable structural locations.
Cathodic protection of offshore structures
211
The initial approach to anode location considers the following points:
Protection to conductors, conductor guides, cones and supporting steelwork is
best provided by stand-off anodes located on the conductor bracing levels.
Anodes for caissons, j-tubes and their guides are mounted on adjacent
smctural members.
Anodes for buried areas of piles and conductors are located on the bottom
horizontal frame of the structure, often on the underside. The clearance
between the underside of this framing and the mudline should be determined,
allowing for settlement, in order not to bury the anode.
Anodes for internal surfaces of piles within the pile sleeve are mounted on the
pile cluster and the jacket leg. Anode numbers derived from surface area
calculations may have to be increased to ensure adequate current distribution
among the pile clusters.
.
.
.
103.2 External faces of structure
Attempts should be made to avoid placing anodes on the externally facing surface
zones of legs and braces. These locations would cause the anode to project
beyond the envelope of the structure bounded by the imaginary plane joining the
outer points of the legs. Underwater surveys of existing platforms bear witness to
the comparatively large number of wire rope lengths which have been found on
the seabed close to the jacket or still wrapped around jacket members. These
have ofcen snagged on projecting anodes, and they result in damage either to the
anode or its attachment to the structural member.
Underwater repairs, unassociated with cathodic protection but involving the
lowering of repair clamps, have been executed in the past, but the presence of
projecting anodes on the jacket faces has caused restrictions on the positioning of
lowering ropes and tugger Lines. Most important is the need to consider the safety
hazard involved in line snagging during diver operations. It is thus preferable to
locate the anodes on the underside and inward facing zones of tubular bracing
members and on the inward facing surfaces of main legs.
An additional hazard occurs for anodes Located near the splash zone. Anodes in
this region should be positioned on the inside face or underside of legs and
bracing members. In the event of broadside bumping of a ship, there is less
chance of either the vessel or the jacket tubular bracing member being holed.
Particular care should be paid to the region below the platform cranes.
10.2.3 Clearances to pilng
For a fixed steel pIatform held to the seabed with steel piles, the possible
interference between the piles during their installation and the cathodic protection
arrangements has to be investigated. For the smaller four-leg platforms in
shallower water, where steel piles are to be inserted down through the legs, clearly
no encroachment on the inside of the leg by anodes or by conduits for monitor
cabling can be tolerated. Steel spacers welded to the inside of the leg perform the
function of centralising the pile. These cannot be considered to offer protection
212
MTD Ltd Publication 90/102
to cable funs attached to the inner wall of the leg, even though these fall within
the shadow of the spacers or centralisers.
For steel jackets with several piles located in pife sleeves around the base of each
leg, the alignment of the piles may follow the batter of the leg, or it may be
vertical. Battered piles have been used on most of the existing North Sea
structures, utilising steam hammers for their installation. More recently, the
advent of underwater hammers has resulted in more vertical pile clusters being
used with less risk of the pile striking the anodes while it is being lowered into the
pile sleeves at the foot of the jacket.
For battered piles where the pile is lowered through guides attached to the top of
the jacket and attached to each of the plan bracings, the spacing of the pile
around the leg (and hence the clearance between pile face and leg face) is usually
determined by soil mechanics considerations at the mudline. It is normal practice
to seek a minimum clearance of 500 mm between the face ofthe pile and the
closest brace member. It is essential that a check is made to avoid encroachment
by anodes or cabling upon these clearances (Figure 64).
Minimum
clearance
500rnm
Ver t ic a I
( a 1 true view down jacket leg -inclined piles
parallel t o inclined jacket leg
Figure 64
[ b ) plan view a t seabed bracing level-vertical
piles with inclined leg
Configuration of piles round steel jacket leg
Cathodic protection of offshore structures
213
Where vertical piles are used, the upper pile guides are dispensed with and
reliance placed on the vertical pile sleeves attacbed to tbe base of the jacket leg.
Vertical piles, unlike battered piles, cannot easily be placed within tbe jacket
envelope. Piles 1 and 2 (Figure 64(a)) are relocated on the outer side of the
jacket as shown in Figure 64 (b).
This removes the problem of the pile fouling the anodes on the inside face of the
bracing, and it leaves only the problem of adequate clearance to the outside face
at the two lower plan levels. It is structurally desirable to have the centroid ofthe
semicircle of piles close to the point where the battered leg strikes the mudline.
This forces the pile close to the brace and aggravates the clearance problem.
10.2.4 Clearance for fabrication
The location of anodes on nodes whicb form support points for jackets at roll-up
during construction should also be considered carefully. Figure 65 shows the
envelope swept out by the support cups or saddles during frame roll-up. The rollup cups are usually positioned on the thick-walled cans forming the barrel of the
node. If anodes can be permanently omitted h-om these locations, it avoids having
to weld them into position after the roll-up cnps are removed. Cable runs for
monitor anodes are particularly vulnerable at these points, and they should be
routed on the inward facing surface of the node. These nodal areas, nsed to
support frames on roll-up cups, occur on all four frames of an eight-leg jacket.
Similarly, the outer leg of the frame during roll-up starts at ground 1eveI and
finishes in the air.
n
Crane
supports
Zone occupied
' by cups during
Self weight,anodes,
frame roll-up
affects anode
//M/N/H/N/rnZiA
\
Cup or
saddle supports
( a ) Prior to roll-up
Figure 65
214
( b ) After roll-up
Sections through jacket frame
MTD Ltd Publication 90/102
Crane attachments are made to this leg so the same principle of avoiding anodes
or cables at nodes opposite roll-up cups applies. The method the fabricator
proposes for supporting the frames during construction is his contractual
responsibility, and it is usually not known at the time the structural engineer
prepares the detail drawings. The engineer has to recognise the need to be
flexible when reviewing proposed anode relocations to suit fabrication
requirements. Although many of the clashes between anode locations and the
fabricator’s temporary works are removed, welding high in the air instead of near
the yard surface triples the welding cost, because of the access problems.
Further dficulties arise with the positioning of sea fastenings for a Iaunched or
lifted jacket. These frequently take the form of tubular members, some 600 to
1000 mm in diameter, which attach the structure to the transportation barge.
Substantial forces are generated within the sea fastenings, and it is usually essential
to weld them to the thickened wall of the nodes forming part of the launch or
support legs of the jacket. There can be as many as four seafastening members at
each node, occupying a considerable proportion of the surface area and clashing
with proposed anode positions.
Re-locating the ends of the sea fastenings onto adjacent thin walled members can
incur the penalty and cost of substantial internal ring stiffening. It is usually
preferable to move the anodes onto adjacent braces. Unlike temporary works
attachments at the fabrication yard, there is insufficient time for anodes to be
attached in the period between removal of the seafastenings and the launch or lift
of the jacket.
10.2.5 Template for early production
A steel template structure, located on the sea bed, is often used to shorten the
otherwise lengthy drilling period folIowing platform installation. The template is
usually installed in the season prior the main jacket placement. This permits a
number of wells to be drilled, using a drill ship or jack-up, and then temporarily
capped. The main structure, its well-slot pattern matching that of the template, is
installed by docking above the template, and the pre-drilled capped wells are
connected by conductor pipe to the platform deck. When a sacrificial anode
system is planned for the main structure, the template is initially protected by its
own anodes. These are frequently smaIler in size than those adopted for the main
platform, and they are distributed within the body of the compartmentalised
framework forming the template. Although anodes within the envelope of the
template are not usually considered a diver hazard, care should be taken if
positioning anodes on the upper surfaces. They should not interfere with the tieback operation of connecting the conductor to the pre-drilled wells.
Additional anodes are provided on the main structure to allow for the extra
current load should the template become connected to the structure via the
conductors. The area of steel in the ternpiate is usually small in relation to the
jacket, corresponding to between 0.5 and 2% of the main steel mass.
Cathodic protection of offshore structures
215
10.3
WEIGHT ASPECTS OF THE CATHODIC PROTECTION SYSTEM
The weight of the cathodic protection system influences some of the structural
arrangements. In general, the more weight the structure carries the greater the
increase in overall cost. In this respect, a sacrificial anode system is heavier than
an impressed current or a combined coatindanode system, and it thus imposes a
heavier penalty.
Experience indicates that for North Sea jacket structures uncoated below tbe
splash zone, the weight of the sacrificial anode system contributes between 4 and
6% of the weight of the structure. These percentages relate to the weight of a
structure ready for launch.
A rolling margin allowance of typically 2% is added when calculating the lifc or
launch weight to cater for the effects of steel plate and other steel components
being supplied to a plus tolerance. This same allowance needs to be retained by
the engineer when assessing the weight of the sacrificial anode system, because
most specifications call for the supplied weight to be within -0 to +2% of the
specified weight.
The proportion of the strength of the steel structure which can be considered as
resisting the vertical load from the modules and deck, as we11 as its own self
weight, depends on the junction of the particular platform and whether the
presence of a large number of conductors or other appurtenances is attracting
additional wave loading. However this proportion often lies in the range of 30 to
40% of the weight of the jacket structure, and hence the presence of a cathodic
protection system with several hundred tonnes of anodes requires the appropriate
amount of jacket steel to support it. Reductions in the weight of the cathodic
protection system thus give an indirect benefit, albeit smal1, by reducing the
quantity of steel used in fiaming the structure and forming the piles.
The effect of cathodic protection weight on the overall weight of a platform also
has to be considered when the installation method is being examined. For the
large central and northern North Sea structures, the installation weight usually
dictates the provision of a barge launcbed jacket structure or, less frequently, a
self-floating tower structure. A graving dock is used to construct a self floater
which relies on the provision of large diameter legs to provide buoyancy for the
tow to its final destination. The buoyant legs are proportioned to carry the self
weight of the structure including the weight of the anodes, and the legs remain an
integral part of the tower throughout its life.
For a barge-launched jacket structure, the combined weight of the structure with
its cathodic protection system is dealt with by fitting temporary buoyancy tanks to
assist with floatation prior to jacket upending. The weight of such tanks
represents some 9 to 14% of the jacket weight, so a small proportion of this
additional tank weight can be considered as supporting the cathodic protection.
Unlike the buoyant legs of the tower structure, these tanks are removed
216
MTD Ltd Publication 901102
immediately the jacket is installed, and they do not spend their whole life
amacting additional wave loading. The secondary effect which the weight of the
cathodic protection system has on the overall structural weight is thus marginally
smaller for the barge launch jacket.
Smaller platforms in the shallow water depth of the southern North Sea, or
slender platforms in the central and northern North Sea, can be installed by lifting
from their transportation barge and placing on the sea bed. The advent of a new
generation of heavy lift vessels in recent years means that larger steel jackets can
also be installed by lifting. This has the attraction of dispensing with the launch
trusses and runners and some of the temporary buoyancy. In this case, the weight
of the cathodic protection system presents a different problem.
For the given size of jacket and water depth, the operator is obliged to set a
maximum limit on the weight to be lifted. This lifting limit is governed by the
avaitabitity and capacity of the heavy lift vessel and represents a not-to-exceed
weight for the design engineer. Exceeding the weight means that there is no vessel
to undertake the lift, with the schedule and cost consequences associated with
reverting to a launched jacket, or it may mean that a larger class of lift vesseI is
required and hence a large step increase in the installation cost.
There is a strong incentive to minimise the structural weight to ensure liftability,
and the cathodic protection system is one area which should be closely examined.
By dispensing with the launch trusses, the steel area is reduced with proportional
reductions in the cathodic protection weight. Further small reductions can be
achieved by optimising &heweight of the specified anode material. Alternatively,
by considering a combined coating and sacrificial anode system for the submerged
part of the jacket, a worthwhile weight reduction may be achieved. In cases such
as this, although the cathodic protection cost may increase, the overall cost benefit
needs to be established. For a lifted jacket, it is usually necessary to undertake a
study at the conceptual design stage to establish the optimum approach to
cathodic protection weight.
For lifted jackets, consideration should also be given to the increased weight
associated with the 12 mm of wall thickness added to structural members passing
through the splash zone and the weight of risers, caissons, J-tubes and other
appurtenances in the same region. A coating system for appurtenances may be
shown to be beneficial from a weight reduction viewpoint. While most of the
risers and J-tubes may be in position at lift time, the caissons can often be
engineered for later installation, and a saving in lift weight can be achieved.
10.4
WAVE ACTION AND CATHODIC PROTECTION
A large proportion of the strength of a fixed platform is devoted to resisting the
horizontal forces imposed by wave and current action and the resultant
overturning effect this exerts on the structure.
Cathodic protection of offshore structures
217
Hydrodynamic forces on an element of a k e d structure may be split into two
components: a drag force and an inertia force
F=
where
4e
Fd
=
CipVu" + C,pAk2
(10.1)
Ci = inertia coefficient
c d = drag coefficient
V = volume of element
A = presented area ofelement
u = water particle velocity
u" = water particle acceleration
p = density of sea water
The presence of marine growth and protrusions from the structure such as the
cathodic protection system does not significantly increase volumes and inertia
forces, but it does increase areas and drag forces. Water particle velocity decreases
from water surface to sea bed, and fortunately it is here that the cathodic system
(particularly sacrificial anodes) tends to have its greatest distribution density.
It is normal practice when assessing the drag on the structural elements to make an
allowance for the increase resulting from the presence of the anodes and conduits,
whether these serve a sacrificial, monitoring, or impressed current function. A
review of the effect on representative plan bracing levels and jacket members is
undertaken, and an adjustment is made to the drag coefficient for the individual
members. The effect is more severe for areas with denser cathodic protection. As
an indicated value for the case of sacrificial anodes, the effect might increase the
drag coefficient for the associated tubular member by 7 to 10%.
When drag and inertia effects are taken into account, the net result on the wave
and current loadings on the jacket is to increase these by about 5% for a sacrificial
system. This increase in wave loading is passed on through the jacket to the
foundations. Diagond bracing members and horizontal frames help to distribute
the wave loading between the jacket legs, and these need to be capable ofcoping
with the anode-induced wave loading. The horizontal loading on the structure
presents itself as a shear force at the sea bed and as axial forces in the legs and
piling.
All these members have to be proportioned to cater for the effects of the wave
loading induced by cathodic protection, although their basic design is governed by
other considerations,
The mudmats, attached to the corners of the structure at the mudline, are also
indirectly affected by changes in the wave loading. Their function is to provide
bearing and sliding resistance to overturning, and to prevent undue settlement after
the jacket has been set on the sea bed prior to piling.
218
MTD Ltd Publication 90/102
10.5 ATTACHMENT DETAILS FOR ANODES
Consideration of the detailed attachment of the cathodic protection system to the
structure is completed during the stage prior to the issue of drawings at the start of
construction.
As indicated earlier, the location of conduits carrying cables from monitor and
anodes should be chosen carefully. Where monitor anodes or impressed current
anodes are used, their cabling systems are normally Tun in conduit, and it is
attractive to route these from their position on the plan bracing across to the legs of
the structure at a low level before routeing them up the inward facing surfaces of
the legs, This avoids unnecessary drag on smaller frame members in the higher
velocity areas near the wave crests. Conduit is run on the underside of tubular
members to give a degree of protection from minor dropped objects. The choice of
legs on which to run the conduits is partly governed by the location of topside
entry points, but where choice is available the legs remote from the sea surface
during the sea tow should be chosen. This avoids wave slam on the conduits,
should the overhanging outer legs on a barge launched jacket dip into the wave
surface during transportation. Similar considerations apply to a self-floating tower
structure, where damage during a long tow is known to occur to conduits fixed to
members too close to the wave surface. For the upper plan brace which carries the
walkway, the conduits can be partially protected by walkway supports.
Although conduits can be run internally within structural members, it has usually
proved preferable in the past to retain them on the surface of the member. This
avoids sealing problems and water ingress into otherwise watertight tubular
members.
The advantages of running cables within members, and the precautions to be taken
when doing so, are also discussed in Section 6. When the penetration of members
is unavoidable, the structural engineer has to ensure that water ingress is prevented,
and that adequate reinforcement is provided around the opening.
Sacrificial anodes need to be grouped more closely at nodes, and these can
represent difficult areas. Welding direct to a thick-walled node which has
previously been stress relieved is unsatisfactory, and doubler plates to receive the
anode insert should be welded to the node prior to its entry into the oven.
Doubler plates for anode attachment are also required when analysis shows tbat
highly stressed regions exist in frame members and in fatigue-sensitive areas.
Doubler plates should also be detailed For the unusual situation of attaching an
anode offshore. Doubler plates should have rounded corners to avoid stress
concentrations.
The position of girth and seam welds in tubular members is generally not known
prior to the start of fabrication, but it should be made clear on design drawings or
specifications that welding the end of the anode insert on top of girth or sea welds
is prohibited.
Cathodic protection of offshore structures
219
For an area of congested small framing members such as occurs in the conductor
guide areas at the plan bracing levels, spanning anodes between adjacent members
should be avoided, The small differential flexing between these members may
impose unnecessary stresses at the attachment weld. A similar consideration
applies when spanning anodes across a substantial change of tubular wall thickness
on the same member, although this may be unavoidable at nodes where a thickwalled stub changes to the thinner wall of the brace itself.
Anode congestion at the comer legs of the jacket in the pipe sleeve area frequently
Provision needs to be made for sufficient anodes to protect the legs, sleeves
and connecting shear plates, as well as tbe associated vertical and ring stiffening. In
addition, an allowance needs to be made for pile stick up above the top of the
sleeve and the internal pile surface within the sleeve. This zone is subject to severe
vibration during pile driving, and the anode fixing details should be checked for a
50 g acceleration. Smaller anodes than used elsewhere on the structure may be
necessary to give proper protection to the compartmentalised surface created by the
rings and stiffeners in this area.
OCCUTS.
Although all tubular members are normally fully sealed and left unflooded, this is
not the case with the jacket legs. For barge-launched and lifted jackets, provision
is made, by means of water ballast entry valves and air vent valves, to flood the legs
partially during the installation operation, and to flood them completely to improve
on-bottom stability prior to the pile being installed. Valve closure normally
ensures that the leg does not subsequently behave as a free-flooding compartment,
but the advisability of attaching anodes to the internal surface of the leg should be
considered or provision should be made for prior placement of corrosion inhibitors
together with biocides.
10.6 STRUCTURAL DESIGN OF ANODES
10.6.1Loads on anode cores
Anode cores need to have sufficient structural strength For:
transportation, launch or lift, and upending
.
.
pile driving vibrations
fatigue from wave loading.
10.6.2Transportation and emplacement
Motions and accelerations of a structure on a barge can be predicted from linear
motion theory for ships. Maximum resolved accelerations are unlikely to exceed
about 2 g. However, if parts of tbe structure enter the water during transportation:
.
.
220
linear theory over predicts accelerations. Model test data give a more realistic
prediction
anodes may be subjected to wave slam.
MTD Ltd Publication 901102
Launch of a jacket of a barge c m cause slam of the nodes of 5 to 1.0 m/s as the top
of the jacket enters the water. The slam impact veIocity is predicted by naval
architecture motions analysis for launch. Upending of a self-Boating structure may
include a 45' roll which also causes slam loads on anodes.
DnV rules(94)for structural design of offshore structures give guidance on slam
loads.
10.6.3 Pile driving vibrations
Anodes on pile sleeves, and some anodes on tbe structure, will be subjected to
severe vibrations during pile driving. Measurements by Mobil on the Beryl B
jacket('w) indicate that pile sleeves experience accelerations oftbe order of 50 to
100 g when battered piles are driven by a slimline underwater hammer such as the
Menck MHU 1700. Steam hammers generally have hardwood to cushion the
impact of the ram onto the anvil, and so they cause lower accelerations than the
MHU 1700, which has a steel to steel impact. The use of vertical piles should also
cause less energy to be lost, and hence less vibration in pile sleeves.
The following steps can be taken to assess the probability of fatigue failure of
anodes on pile sleeves during pile driving:
Assess maximum acceleration of pile sleeve for each hammer blow.
Assess effective mass (mass of metal plus added mass) of anode.
Compute maximum bending stress in anode core at supports.
.
.
.
Compute natural frequency of anode.
Assess stress response of anode by assuming half-sine wave input and suitable
level of damping.
Assess fatigue class (S-NCurve).
Compute fatigue damage for the sequence of stress ranges experienced from to
one blow,
Multiply by the number of blows to drive a pile, which can be predicted from
pile driveability studies.
Shock and vibration tests should be performed on cathodic protection monitoring
systems (if fitted to the structure).
10.6.4 In-pIace storm and fatigue
Structural checks should be performed on anodes located close to the water line.
Water particle velocities for the design winter storm (wave plus curreat) can be
extracted from wave grid runs for the structure. For anodes, drag forces should
predominate. Allowance should be made for the increased water particle velocity
resulting from the proximity of the member to which the anode is attached.
Typically, local velocities may be twice the free stream velocity. For laminar flow
around a tubular, the potential flow model for the doublet (point source and sink)
may be used.
Cathodic protection of offshore structures
221
Deterministic fatigue checks should also be performed, With waves grouped into a
limited number of height classes.
10.6.5 Doubler plates
Doubler plates serve to protect the structure against damage:
if the anode is torn off the structure or needs to be replaced in service.
Doubler plate to shell welds should be stronger than attachment to doubler
plate welds. To avoid small fillet welds onto thick shells, doubler plate to shell
welds should generally have a leg length 2 m m less than the thickness of the
doubler plate.
because of relatively poor quality of anode to shell welds under adverse
conditions (e.g. site, as opposed to shop, welding), or welds to sensitive areas of
heat-treated items after post-weld heat treatment. Doubler plates on such items
enable anodes, etc to be attached after post weld heat treatment.
.
It follows that doubler plates are not needed for anodes attached to low stress areas
which are not at risk from fatigue or brittle fracture, areas which are welded in
shops, and areas which are not prone to mechanical damage in service. Excessive
use of doubler plates may cause fit-up and organisational (non-conformance)
problems during fabrication. Doubler plate locations should be checked when
shop drawings are prepared.
222
MTD Ltd Publication 90/102
Section 11
Current legislation, standards and guidance
documents
Cathodic protection of offshore structures
223
11,
Current Legislation, standards and guidance
documents
As this Section makes many references to documents, these are cited hy numher.
A key at the end of the Section shows these numhers and the organisations
responsible for the documents, listed by country of origin, as well as the Reference
number under which (at the end of the Guide) full details are given.
To distinguish guidance issued by government departments and agencies, these
documents are asterisked. Though they are not formal documents, NVO papers
(N6,N8) give much information on the requirements set by the Norwegian
Petroleum Directorate and Det norske Veritas, along with comments on these
standards.
The documents listed are those current at the time of preparation of the Guide.
As both legislation and technical perspectives change with time, readers are
advised to verify that they are using the most recent versions of the documents
they consult.
It has to be emphasised that the aim of this Section is to summarise what the
documents themselves say. No attempt is made to reconcile divergent views or
apparent disagreements with information presented elsewhere in the Guide.
Numerical values and units of measurement (e.g. potential scales) are reported as
they appear in each document.
11.1
LEGSLATION
Altbough this Section is devoted primarily to the technical content of the
standards and guidance documents issued in various countries, it is useful to begin
witb a brief summary of the legislative provisions from which, in the two countries
principally concerned in North Sea activities, the technical guidance derives. The
provisions made in other countries are also briefly mentioned.
In both the United Kingdom and Norway, the ultimate motive for legislation is
safety, and this, of course, covers very many aspects of the construction, operation,
and maintenance of offshore petroleum installations, Requirements for corrosion
protection, and in particular for cathodic protection, form only a part of this body
of legislation and regulations. For a fuller account of the legislation and its
implications the reader is referred to the recent book by Barrett, Howells &
Hindley (GB1).
11.1.1
United Kingdom
The first significant enactment is the Mineral Workings (Offshore Installations)
Act 1971, extended by the Petroleum and Submarine Pipelines Act 1975, and
amended by the OiI and Gas (Enterprise) Act 1982. These Acts apply to the
territorial sea adjacent to the UR and outwards to the limits designated in the
Continental Shelf Act 1964. Under the first Act, the Offshore Installations
(Construction and Survey) ReguIations (SI 1974/28a) were made in 1974. These
Cathodic protection of offshore structures
225
require all relevant offshore structures to b e “certified” as fit for their specified
purposes. The Secretary of State for Energy has authorised each of the following
independent bodies (termed Certifying Authorities) to issue Certificates of Fitness:
-
American Bureau of Shipping
0
Bureau Veritas
a
Det norske Veritas
a
Germanischer Lloyd
Lloyd’s Register of shipping
Offshore Certification Bureau,
A Certificate of Fitness is issued when the Certifying Authority is satisfied that an
installation is fit for its intended purpose, having regard to its design, specification
and methods of construction, and that it will remain so during the time that the
Certificate remains valid (which may be up to 5 years).
The United Kingdom Department of Energy issues Guidance Notes (GBZ) which
set out the procedures and criteria to be adopted in assessing an installation for
the issue of a Certificate of Fitness. Where appropriate, the Notes refer to
relevant Codes and standards, but their main purpose is to provide guidance when
suitable codes and Standards are not available. Some sections of the Guidance
Notes cite background documents, which support the recomrnenda tions of the
guidance itself with supplementary technical information. For the assistance of
designers and operators, Certifying Authorities have also produced guidance
documents.
11.1.2
Norway
Several sets of Regulations were made by Royal Decree from about 1977 onwards,
and these are in the process of updating and revision. The most recent legislation
is the Act pertaining to Petroleum Activity March 1985,which is supplemented by
Regulations of July 1985. A summary of the 1985 Act and its implications is given
by Braathen (N8).
Under this Act, the Norwegian Petroleum Directorate has delegated authority,
from the Ministries of Petroleum & Energy and of Government & Labour, to
(inter alia): verify that petroleum activities are carried out in a safe and sound
manner, control all phases of petroleum activity, and maintain contact with
scientific and educational institutions. In addition to its legislative functions, the
NPD prepares guidance documents, summarised below. Some of these supersede
earlier documents, and the following technical discussion is to some extent based
on documents still in draft form.
11.1.3
Other countries
In the Federal Republic of Germany, the body with legislative authority is the
Oberbergamt Clausthal-Zellerfeld, which is responsible for mining activities in
Northern Germany, including territorial waters and the Continental Shelf. The
226
MTD Ltd Publication 90/102
Oberbergamt approves offshore structures on the basis of reports from
Germanischer Lloyd, an independent expert organisation. In Denmark,
permission for construction is given by the Danish Energy Agency, which requires
adherence to guidance documents produced by the Dansk Ingeniorforening (DIF).
These are prepared by working groups of specialists and have no legal status per
se, but become binding if their use is prescribed by law. They include the new
document listed here (DK1). In Holland, a guidance document is available
(ND l), and the rules of the American Petroleum Institute, The American Welding
Society, the British Standards Institution, and Det norske Veritas are Followed.
11.2
GUIDANCE DOCUMENTS
11.2.1
Requirement for cathodic protection
A11 the cited documents allow, and some require, the application of cathodic
protection to external submerged steel surfaces. Combination with coatings is
generally allowed. GB6 forbids the use of coatings without cathodic protection.
N3 advises the use of coatings in areas where uniform current distribution is hard
to achieve. Similar stipulations are made for pipelines and risers. Internal
submerged regions open to the flow of sea water are also required to have cathodic
protection with or without a coating. N7 forbids the use of impressed current
cathodic protection in closed compartments with limited water circulation. N8
discusses two methods by which thermally insulated pipelines may be cathodically
protected, sacrificial anodes being placed either inside or outside the insulation.
GB2 stipulates that cathodic protection of internal flooded spaces should be
adequate for all conditions encountered, and also warns of the danger of microbial
action in closed submerged spaces. Cathodic protection, combined if desired with
coatings, is also called for on structures and pipelines below the sea bed. Here, a
lower protective current density may be allowed, for example by DKl,N3,N7 (see
also Section 11.2.3). D1 points out that sacrificial anodes on pipes below the sea
bed should be assumed to give a lower current: the value in each case is to be
agreed with Germanischer Lloyd,
11.2.2
Design principles
The documents vary widely in the degree of detail with which they set out the
principles to be used in designing a cathodic protection system. Some (eg GB2)
merely state that the system has to have a life similar to that of the structure and
be capable of polarising all parts of it to a sufficient level to minimise corrosion
(see Section 11.2.3). Where a coating is also applied, allowance is to be made for
damage it may suffer (GB3).
Other documents, particularly US1,N3 and NDI, set out in detail the data needed
for a design, as well as the procedures for carrying it out. US2 gives similar details
for the cathodic protection of pipelines. ND1 points out that a preIiminary
investigation at the site may be needed to obtain the necessary information.
Warnings are given against the dangers of overprotection (hydrogen entry and
embrittlement), eg GB2 (see Section 11.2.3). Several documents (US 1,NDl)
summarise the factors governing the choice of a sacrificial against that of an
impressed current system. These include the availability and reliability of power
Cathodic protection of offshore structures
227
supplies, the total current required, the space and geometry available for the
cathodic protection installation, and the costs of initiai installation and subsequent
maintenance. The superiority of sacrificial cathodic protection in terms of current
distribution is mentioned (US1, D1, NDL).
Many of the documents give detailed instructions for calculating the total number
of sacrificial anodes required, and they include formulae for the anode resistance,
from which the current output of each anode may be derived
(N3,GBS,USl,N7,GB6,DKl).
The thrust of all these recommendations is that the
current distribution should be such as to bring a11 parts of the structure to a
protective potential (see Section 11.2.3), both initially and later in life. The role of
coatings favouring a more uniform distribution is often emphasised (eg N3,N7),
and several documents (N7,N3,ND 1) recommend increasing the number of anodes
above that calculated from the current demand of the structure, as a further means
of improving distribution.
N 3 recommends that with uncoated structures of complex shape the design value
of the driving voltage (see Section 11.2.8) should be reduced by 30 to 80 mV to
allow for IR drops in the cathodic areas. The improved distribution of impressed
current resulting from careful placing of anodes and the use of dielectric shields is
discussed (N3,USl,ND1). GB6 requires anodes protecting buried steelwork to be
placed immediately above the mud line.
For calculating the weight of sacrificial anodes, GB6 details the reduction in their
size which is to be assumed at the end of life. N7 points out that initial cathodic
protection schemes are often prepared early in a project, and that estimates ought
to be revised at the end of structural design to ensure the iaclusion of all relevant
steel areas.
N 7 stipulates that an impressed current system shall be able to deliver 30 to 40%
additioaal current density, to allow for a higher demand during the initial period
of exposure. USl,NDl,N7 and N 3 call for a 25 to 50% excess capacity to allow for
non-uniform current distribution, this being accompanied by the recommendation
that the uniformity ofimpressed current distribution may be improved by using
dielectric shields and placing the anodes at large distances from the structure. For
the same reason, D1 recommends increasing the installed current output by a
factor between 1.2 and 1.5, and stipulates, as does N3, that, unless dielectric
shields are fitted, anodes should be at least 1.5 m from the structure.
GB2 indicates that an impressed current installation may need to be provided with
a sacrificial backup system for use when the current is turned off (eg during diving
operations). ND1 suggests that the system should he capable of an increased
output, in case of future modifications to the structure. For pipelines, N1 gives
similar recommendations to those of N3. Here, cathodic protection is normally to
be by sacrificial anodes. GB6 requires it to be "shown by appropriate
calculations" that the system can polarise the structure adequately.
228
M T D Ltd Publication 901102
11.2.3
Criteria of protection
All the documents regard the potential as the primary factor in the control of
corrosion. There is virtual unanimity that the threshold of satisfactory protection
is at -0.80 V Ag/AgCYsea water. Many of the documents alIow the use of other
reference electrodes (eg cu/CuSO,, Zinc, Saturated Calomel), and they give
equivalent potentials on these scales. Reference 12 provides a discussion of the
alternative electrodes, and also a nomogram for converting Ag/AgCl values to
Saturated Calomel in media other than sea water.
The only other potential criterion, suggested by US1, is that of the “potential
shift”. Here, protection is to be judged adequate if the steeI potential shifts
negatively by at least 0.300 V when the (impressed) current is imposed.
All the documents point to the need for enhanced cathodic protection when ‘
conditions are anaerobic and SRB are present. N3 specifies potentials in the range
-0.80 to -1.05 V Ag/AgCl under aerobic conditions, and -0.90 to -1.05 V under
anaerobic conditions or when the bacteria are present.
There are frequent warnings against the danger of causing hydrogen embrittlement
by over protection, and for this reason a limit of -1.05 V Ag/AgCl (DK1 gives
-1.15 V c u / C u S 0 4 ) is set.
It is recognised that these metallurgical limitations are more serious with stronger
steels, and they are stated to hecome significant at tensile strengths in the range
700 to 800 MPa. D1, however, limits the yield strength to 800 MPa: above this
value, the permitted potential is to be agreed with Germanischer Lioyd. N3
requires prior testing of steels with a yield stress above 700 MPa if they are to be
exposed at potentials more negative than 0.90 V Ag/AgCl.
With impressed current systems, potentials are to be measured with the current
on, and with a11 systems care is to be taken to minimise the disturbing effect of IR
drops between the steel surface and the measuring elecbode. To that end, the
latter is to be placed as close to the steel as possible, and marine growth is to be
removed before measurement (US 1). However, DK1 recommends that marine
growth should not be removed (see Section 11.2.11).
The documents present their current density criteria as guidance based on
existing experience as to the current densities needed, in various locations and
circumstances, to achieve the necessary protective potential. The dominant factor
here is the availability of dissolved oxygen at the steel surface, and most
documents point to low temperatures, high flowrates, and sand scouring as
important factors in increasing the current requirement. N7 and N3 call for the
current requirement to be increased by 10 to 20% for steel areas within 20 rn of
the sea surface. ND1 requires the inclusion, for purposes of area calcuiation, of
all adjacent structures not specifically isolated from the structure being protected.
Many of the documents give tables of suggested values for different locations,
sometimes showing initial, mean and final values. The value to be used in design
calculations, for example of the number and weight of sacrificial anodes, is usually
Cathodic protection of offshore structures
229
indicated (N3). The final value has to be achieved at the end of life by anodes
consumed to the extent of the utilisation factor (see Section 11.2.8). It is
recognised that considerably higher current densities are needed early in the life of
a structure while a protective calcareous scale is forming. US1 and GB6 warn that
storm damage may partially remove this scale, and the latter requires the system to
be able to provide enough current to bring the exposed steel quickly to a
protective potential.
It is recognised by GB6 that particular installations may require different current
levels, and these are allowed, provided they are appropriately justified. D1 calls
for field tests to determine the required current density if all necessary parameters
at the site are not known. Recommendations similar to those of N3, but for
pipelines, are given by N1.
Documents giving tables of recommended current densities include
Dl,N7,NDl,DKl GB6,GB5,N3 and US1. These cannot all be reproduced here,
but may be exemplified:
Area
Southern N. Sea
Northern N. Sea
Cook Inlet
Initial current
density (mAlm2)
130 to 150
160 to 180
250 to 2000
Average current
density (mAlm2)
85 to 100
85 to 120
400
The values of Cook Inlet are the highest specified, and they serve to show the
upper limit of normal requirements. N3 indicates that initial current densities of
250 to 300 mNm2 may be needed in the North Sea at latitudes greater than 65w.
N7 requires polarisation to be achieved within 3 months of the installation of the
structure. O n the other hand, GB2 and GB5 require, in the case of impressed
current systems, that critical regions should be protected by supplementary
sacrificial anodes, to ensure adequate polarisation from the moment of immersion.
The latter reference requires magnesium anodes used for this purpose to be placed
at least 3 m from the steel surface.
The requirement for steel below the sea bed is generally in the range 20 to
40 mNm2 (Dl,GB4,N7,DKl). N3 recommends 50 mNm2 initially for buried
pipelines, with mean and final values of 40 mNm2. For pipelines carrying hot
product, DK1 calls for the addition of 1 mNm2 for every OC by which the
temperature difference between product and sea water exceeds 25OC, while N3
requires an additional 2 mNm2 for every OC the "operating temperature" is above
25OC. Several documents specify the current to be provided for wells below the sea
bed (GB6,N3,N7: 5 Nwell, ND1: 3 Nwell).
11.2.4
Coatings
Most of the documents recognise the benefits which come from combining cathodic
protection with coatings, though they vary considerably in the attention which they
give to these. GB2 merely points out that coatings can reduce the total current
demand on a cathodic protection system and also improve current distribution. A
230
MTD Ltd Publication 90/102
warning is given against the danger of disbondment under over protection. For
coated steel, GB4 sets a potential limit of -0.9OV AdAgCI.
ND1 states that coatings are rarely used in the underwater zone for economic
reasons, and stipulates that cathodic protection designs have to allow for the
deterioration of coatings with time. For coated submarine ipelines, the current
demand is stated to he anywhere between 5 and 200 m N m ,depending on coating
quality, the latter to he established by tests or on the basis of experience.
O n the other hand, N7, in addition to the advantages mentioned above, specifically
suggests consideration ofcoatings on submerged areas within 20 m of the sea
surface. Emphasising that the current demand increases with time as the coating
deteriorates, the document tabulates the initial, mean and final current densities to
be assumed for thin coatings on structures with lifetimes ranging from 5 to 40 years.
In all cases, an initial current density of 4 mNm2 is set, rising to final values of
12 mNm2 for a 10-year lifetime and 90mNm2 for a 40-year lifetime.
P
N3 gives a table of thin film (i-e. less than 1 mm) breakdown for lifetimes ranging
from 10 to 40 years. N1 tabulates the 25-year breakdown of four types of pipeline
coating. D K l tabulates the initial, mean and final percentage degradation to be
assumed for four types of coating over 25 years. The "final" values here range
from 10% for 2 to 4 mm polyethylene up to 50% for 0.3 mm coaltar/epoxy.
N8 and N3 tabulate minimum design current densities for a 30-year life with
pipeline coatings of various types. Initial current densities are only a few mNm2,
particularly for buried coated structures, while the final values range from 5 mNm2
for buried steel with both coating and a concrete weight coat, up to 50 mNm2 for
steel with a thin film coating exposed directly to sea water.
D1 stipulates that coatings have to resist blistering down to a potential of -1.10 V
Cu/CuSO, and also not be susceptible to hydrolysis and saponification. For a 30year life, the current density requirement is set at about 50% of the value for bare
steel, though initially the demand is estimated at 10% ofthis value. For pipelines,
buried pipes, and risers, D1 specifies the details of a coating system and its
application procedure which have to be provided to Germanischer Lloyd, alluding
to relevant DIN specifications.
GB6 allows consideration of reduced current densities if "suitable" high duty
coatings are used, and GB3 requires pipeline coating materials to be proved by
extensive testing or experience, listing several coating properties (including
resistance to cathodic disbonding) to be assessed. For mobile units having coatings
combined with impressed current cathodic protection, GB5 calls for prior removal
of rust and mill scale to Sa 2.5 ofSwedish Standard SIS 05-59-00.
11.2.5
Reinforced concrete
GB2 requires the exposed steel parts of reinforced concrete structures to be treated
as stipulated for bare steel. The interaction of cathodic protection installations with
adjacent reinforcing and pre-stressing steel needs to be considered, also the risk of
enhanced corrosion of steel structures adjacent to reinforced concrete, as a result of
electrochemical cells between steel in concrete and steel in sea water. Reinforcing
Cathodic protection of offshore structures
231
steel should therefore either be eIectrically isolated from cathodically-protected
steel or, alternatively, incorporated in the cathodic protection design with
allowance for current flowing to the steel within the concrete. Consideration has to
be given to avoiding over protection of pre-stressing steel, with its risk of hydrogeninduced stress corrosion (see Section 3).
N7 stipulates that when exposed steel is in contact with steel embedded in concrete,
a current drain on the latter between 0.2 and 4.0 m N m Zshould be allowed. As an
average, 1.0 mNm2 should be assumed in the tidal and submerged zones.
Embedded steel should be connected to external steel, to extend cathodic
protection to any reinforcing steel exposed during the life of the structure. D1 calls
for steel outside concrete to be connected to the reinforcement. The current
allowance for the "external" reinforcement (the outermost layer of reinforcing
steel) should be 3 to 5 mNm2, its area being approximated to that of the concrete
surface. N 4 recommends 0.5 to 1.0 mNm2 for the outer reinforcement layer.
GB7 recommends that there should be no connection between a concrete-coated
pipeline and the reinforcement, to prevent the pipe from being shielded from the
protective current. It is further suggested that connection between the
reinforcement in successive pipe joints should also be avoided, so that if pipe and
reinforcement are accidentally connected, the consequences are confined to that
locality.
11.2.6 Interactions
In discussing both pipelines and structures, most of the documents warn of the
possibilities for interference between a cathodic protection system and adjacent
metal components, either unprotected or under another cathodic protection system
(e.g. GB2,GB3,GB7,US1,DK 1). According to the circumstances, the advice is
either to insulate from the "foreign" components or to bond to them. In the latter
case, their area has to be incorporated in the cathodic protection design. On
account of their relative areas, a platform is more likely to affect an adjacent
pipeline than vice versa (US1). GB4 advises the provision of additional current
capacity, and a flexible connection, for the use of vessels moored alongside
structures.
N D l gives a criterion for judging interference by an adjacent (impressed current)
system: if, when turning the cathodic protection system on, the positive potential
shift of the structure in question (expressed in mV) is more than twice the
resistivity of the environment (in ohm-m), the interference is significant. GB6
requires all parts of a protected structure to be bonded electrically. If bonding
straps are not installed, potential measurements need to be made on the relevant
components to ensure that significant corrosion is not occurring.
DK1 discusses insulation materials and procedures in detail. D1 requires risers to
be insulated from platforms, even if both are the same cathodic protection system,
and ND1 stipulates that insulating devices for risers should be above the splash
zone and readily accessible. US1 states that insulating Ranges in pipes should be in
vertical sections to avoid short circuits by internal deposits. The dangers of
sparking at such insulation points should be recognised. NDI points out: that
232
MTD Ltd Publication 901102
insulating shields and coatings can help to minimise interference.
US2 discusses all aspects of interference currents in pipelines, including
implications for cathodic protection. Interference problems are considered less
serious with sacrificial than impressed-current systems.
11.2.7
Impressed current anodes
Possible anode materials are: silver-lead alloy, platinum-lead alloy, graphite,
silicon-iron, platinum on various substrates (tantalum, titanium, niobium), and
scrap steel. Most of the documents discuss the use, and limitations, of some or all
of these materials. The use of dielectric shields and insulating coatings to prevent
excessive current flow to nearby parts of the structure is also generally described.
ND1, N3,US1 indicate the advantage of a minimum separation of anodes from the
structure: these documents all recommend a minimum of 1.5 m.
D1 allows silver-lead anodes only at depths less than 30 m, and also sets limits on
the potential allowable at the surface of platinised anodes: Pt-Ti 8 V, Pt-Nb 60 V,
Pt-Ta 150 V. Several documents tabulate the maximum current densities allowed
for anodes of different types (USl,NDl,DKl,GB4).
These range from about 10 A/m2 for graphite to around 1000 Nm2 for platinised
niobium (3000N m 2 in US1). In some cases (NDl,USl), the rate of consumption
of these materials is also given as a function of the total charge passed. In most
cases, the loss is small, a few grams or tens of grams per A-year, but, for scrap steeI,
ND1 gives 8.8 kg/A-year. GB4 advises that an ac ripple at frequencies below 100 Hz
can reduce the life of platinised anodes.
D1 specifies the details of anode composition and manufacture to be provided
when seeking Germanischer Lloyd's approval.
11.2.8
Sacrificial anodes
The sections devoted to sacrificial anodes are among the most detailed in these
documents, and they are inevitably much compressed here. Three metals serve as
the base for anode alloys: magnesium, zinc and aluminium. Magnesium alloys are
very active electrochemically, and they are usually recommended only for short
exposures and in positions where replacement is easy (US1,Dl). N3 forbids their
use in closed compartments.
Zinc is the only material for which an official specification exists, the American
MIL-A-1800lJ specification(61)being frequently cited. DKI suggests reduced
ahminiurn and iron contents for anodes to be used at higher temperatures. For
aluminium anodes, many different proprietary alloys are available, and the
documents therefore set out in detail the allowed compositions and manufacturing
procedures. For the same reason, laboratory and field tests are described (e.g. N7),
sometimes in great detail (N3,NDl). N7 and N3 warn that the potentials found in
short laboratory tests are usually more negative than those achieved on actual
offshore structures. The general approach is that, unless an anode is of a known
type and manufacture, it is not accepted without testing to the satisfaction of the
Cathodic protection of offshore structures
233
authority concerned. The identification and documentation of individual anodes is
also specified in detail (GB5Y7,Dl).
The usual alloying additions in aluminium, to achieve suitable electrochemical
properties, are mercury, indium, zinc, and tin. NDl remarks that aIIoys containing
mercury may be deprecated on environmental grounds. Metallurgical variables
(e.g. cooling rate after casting), may influence electrochemical properties (US 1).
The principal electrochemical parameters of a sacrificial anode are the potentid
achieved on closed circuit (i.e. when giving current) and the total charge delivered,
in Ah. Both these quantities are functions of the current drawn from the anode,
and the documents tabulate in greater or less detail the performance of different
alloys under typical current loadings.
The closed circuit anode potential is used derive the "drivin voltage" by
f voltage is then
subtracting from it the protective potential of the s t r u ~ t u r e ( ~This
used in conjunction with the various anode resistance formulae (see Section 11.2.2)
to calculate t h e current supplied by each anode (Nl,N3,DKl,GB6,USl,NDI ,GB5).
In addition, an effective anode has to have a low tendency to passivation and to
intergranular corrosion (N7,Nl,N3). The risk of passivation is greatest when
protecting coated structures, with their low current demand (N3).
Temperature has a significant effect on the performance of anodes, as discussed by
DKl,N3. With rising temperature, the capacity falls and the potential becomes less
negative. These references forbid the use of zinc anodes on surfaces above 5OoC.
The former states that for aluminiudindium alloys there is a linear fa11 of capacity
with temperature, from 2000 Ah/kg at 2OoC to 1000 Ah/kg at 60'C.
Many documents describe the calculation of anode life. This requires an estimate
of the "utilisation factor" (i.e. the fraction of the anode consumed when t h e
remaining material cannot provide the required current). Values between 0.75 and
0.95 are quoted, the highest (0.90 to 0.95) being attributed to long slender anodes
(N3,N4,N7). AIternatively, a "safety factor" is used in the calculation (Dl). GB2
warns of the reduction in output towards the end of anode life, and GB6 gives the
reduction in physical size to be expected at this time.
US3 specifies the non-electrochemical requirements of anodes, such as size,
manufacture, quality, and documentation. Detailed requirements for the
composition, form, and surface preparation of steel anode cores are given in GB6
and N3.
11.2.9
Mechanical aspects
The documents all indicate, in greater or less detail, that anodes, cables, and other
components of a cathodic protection system are to be attached to structures or
pipelines in such a way as to resist damage from the regular conditions of exposure
such as wave forces and impact from vessels, as well as from storms, piling activity,
and anchor damage. Cables should be protected from impact and stressing. US1
recommends that the structural members themselves should be designed to take
234
MTD Ltd Publication 90/102
advantage of cathodic protection and facilitate the latter's installation. ND1 and
US1 suggest that the initial design should make provision for the later addition of
further anodes, if these should prove necessary. D1 states that impressed-current
anodes should be easy to replace under water. USl,GB6,US2,N3,GB5 detail
allowed methods of anode attachment, particularly welding. GB2 and GB5 allow
bolting, but not studs "fired" into a structure. For sacrificial anodes on pipelines,
GB7 prefers welding to bolting, and it also suggests that anodes incorporated in
flange structures offer a means of mitigating crevice corrosion within the latter.
11.2.10 Electrical aspects
Under this heading, the documents aim to ensure good engineering practice in the
specification and installation of all electrical components, such as transformers,
rectifiers, switchgear, insulators, etc. To this end, US1 gives recommendations in
some detail, and D1 calls up another portion (Chapter 3, Section 5 : "Machinery
and Equipment :Electrical Installations") of Volume 1 of the Germanischer Lloyd
Rules.
US2 requires thermoelectric generators to have a reverse-current device, in case of
flame failure. There are also some specific corrosion-related stipulations. US1
indicates that insulating joints should always be above the water line, and that those
in pipelines should be in vertical runs, to reduce the risk of shortcircuiting by
internal deposits. All insulating devices should be accessible for inspection, and if
not they should be provided with leads to allow electrical testing, The risk of
sparking at insulators should be appreciated (US 1). All bonding connections
should be accessible for testing (US1). ND1 requires the ripple voltage on systems
supplied by an ac source to be less than 10% of the dc voltage. GB6 calls for a trip
to operate if the ripple rises above 5% of the dc voltage, also if a fault deveiops
between the high voltage ac supply and the dc output. GB5 requires a warning
light to indicate the state of the cathodic protection installation to any diver control
point.
11.2.11 Assessment and repair
There are two broad aspects of assessment: the continuous measurement of the
performance of a system, and occasional special inspections. The latter includes
verification soon after start-up that the installation is operating as required.
The fundamental measure of effectiveness is the potential achieved on the
structure. Potentials are to be measured with an approved reference electrode,
usualIy AglAgCl or zinc (see Section 11.23). US1 and ND1 recommend a
complete survey after a system is commissioned. N7 calls for the potentials to be
monitored within 3 months of the launching of an uncoated structure and within 12
months of launching a coated one. Others (D1 and ND1) suggest yearly potential
surveys.
DK1 recommends a first inspection of a structure within 3 months ofinstallation
(within 1 year for a buried pipeline), followed by annual inspections until operation
is stable, after which the intervals may possibly be increased. GB2 and US1 advise
particular attention to areas which may be shielded from the protective current,
Cathodic protection of offshore structures
235
also to those at risk from over protection.
DK1 requires sacrificial anodes to be examined physically and hy potential
measurements, the latter being taken hoth before and after the removal of white
corrosion product. On impressed current systems, the potentials and current
output of individual anodes is to be recorded weekly. Reference electrodes used to
control the systems are to he checked frequently.
Reference 12 describes reference electrodes, including standard calomel and
CdCuSO, electrodes, the latter being considered unsuitable for continuous
immersion. Two different grades of zinc may be used for electrodes: pure zinc to
ASTM B418-80 Type II(60)and zinc alloy to MIL-A-18001J(6'). Acceptable
potentials relative to these electrodes are given. US1 gives a full description of
possible types of electrode, including those guided to the surface by a (insulated)
suspension wire. For hrackish waters, US1 and D1 require the Ag/AgCl potential
to be corrected for chloride content. In all cases, measurements are to be made
with the impressed current on.
Precautions to be taken in making potential measurements are set out in greater or
less detail in most of the documents, The chief concern is to minimise the ZR drop
hetween the measuring electrode and the steel surface, caused by the flow of
protective current. For this reason, the electrode is to be placed as close as
possible to the surface, and marine growth is to he removed (US1). DKX points out
that, under typical conditions, a voltage drop of the order of 30 mVlm is to be
expected in the sea water adjacent to a plane uncoated steel surface. DK1 also
suggests that potential measurements should be taken without removal of marine
growth, to avoid temporary disturbance of the potentials. US1 comments that
measurements made immediately after switching off ("instant off' readings) may be
useful in eliminating the IR drop when the current density andlor water resistivity
are high.
It is envisaged that measuring electrodes will he positioned hy divers or ROVs, and
D1 requircs such periodical surveys to be combined with visual inspection of
anodes for signs of passivation or intergranular corrosion. US 1 suggests that visual
inspection of the steel, and suitably-placed corrosion coupons, are useful
supplements to a potential survey.
N5 and N4 (Appendix I) include a potential survey and corrosion observations in
the requirements for initial and annual inspections.
ND1 and N3 set out details of electrical instruments, measuring procedures, and
inspection intervals, and US2 gives similar information for pipelines. N3
recommends that potentials should be measured at a minimum of 2% of sacrificial
anodes and at one anode on each riser.
Permanently-installed reference electrodes are also useful, particularly for points
difficult to access (N3,US1,N7), and such electrodes are specifically required hy
N3,GB5, GB6 and N7 for impressed current systems. GB5 requires impressed
current systems on mobile units to include at least four permanent reference
electrodes, not within 10 m of an anode. Fixed monitoring electrodes should be
236
MTD Ltd Publication 90/102
checked during the periodic surveys (N3).
N2 is devoted entirely to the monitoring of cathodic protection systems.
Electxodes, instruments, calibration procedures and measuring procedures are
described in great detail. Zinc is not considered an accurate measuring eleceode,
though it may be used as a permanent electxode. In sea water, it should read -1.00
to -1.05 V AglAgC1. Zinc electrodes should carry a small anodic current to keep
them active. N2 recommends that steel potentials less negative than -0.80 V
AglAgCl should be taken as showing under protection, while steel potentials
beyond 3 m from an anode should not be more negative than -1.02 V AglAgC1.
Anodes showing potentials less negative than -0.95 V AglAgC1 may be passivated or
polarised by a high current demand, and the potential of adjacent steel areas
should be checked. Readings more negative than -1.10 V AglAgCI for aluminium
anodes, or more negative than -1.05 V AglC1 for zinc, should be considered
erroneous, and the equipment should be checked.
Only a few documents consider the repair measures to be taken if inspection
reveals an unacceptable degree of corrosion. US1 and N3 briefly suggest repairing
or adjusting the components of the system, including insulating devices and bonds,
as well as the provision of supplementary cathodic protection. ND1 lists the same
measures, in addition to the application of coatings and the breaking of elec&rical
connections with other structures. DK1 emphasises the need to consider, while it is
still operational, the possible future re-establishment or upgrading of a cathodic
protection system. Installations should be so designed that subsequent repairs are
easy to carry out. Retrofitting of sacrificial anodes is considered in some detail.
Attention is drawn to the newer methods of underwater welding for providing
reliable connections.
11.2.12 Documentation
Two types of documentation are called for: design details prepared before an
installation is implemented, and the records of experience, inspections, and
modifications and repairs during service. GB2 stipulates, very briefly, that details
and records of both types should be prepared, along with particulars of
maintenance procedures. US1 gives a full and systematic list of design parameters,
with the intention that individual operators should select those which they wish to
specify, and US2 provides similar suggestions for pipelines.
DK1 sets out the design features and particulars of components, including the
composition and properties of anodes, which need to he documented in preparing
a design.
GB5 and GB6 give detailed listings of the particulars on which Lloyd's require to
be satisfied before approving a scheme. Among its requirements, the latter asks for
"details of any computer modelling" - the only reference to computer methods in
all the documents reviewed here. N3 lists the design and operational information
which should be recorded. The documentation of sacrificial anodes is specified in
detail. US3 details the particulars of non-electrochemica1 properties which a
manufacturer of anodes should record.
Cathodic protection of offshore structures
237
With regard to inspections, operational experience and modifications, GB2 states
hriefly that these should be recorded, while US1 gives a full list of suggested
records. GB6 stipulates that potential measurements, taken at agreed intervals,
should be made available to Lloyd’s, DK1 discusses the control measurements and
observations to be recorded, as a means of assisting failure detection and the
assessment of systern performance.
11.3
KEY TO THE DOCUMENTS
Number
Reference No.
Organisation
United Kingdom
Barrett, Howells & Hindley (Book)
GBl
GB2*
Department of Energy
GB3*
Department of Energy
GB4
British Standards Institution
GB5
Lloyds Register
GB6
Lloyds Register
GB7
Institute of Petroleum
Norway
N1
N2
N3
N4
N5*
N6
N7*
N8
112
18
115
72
116
102
118
Det norske Veritas
Det norske Veritas
Det norske Veritas
Det norske Veritas
Norwegian Petroleum Directorate
Det norske Veritas (Paper)
Norwegian Petroleum Directorate
Norwegian Petroleum Directorate
(Paper)
10
103
9
94
117
110
97
111
United States of America
National Ass. of Corrosion Engineers
us 1
National Ass. of Corrosion Engineers
us2
National Ass. of Corrosion Engineers
us3
12
11
71
Federal Republic of Germany
Dl
Gerrnanischer Lloyd
95
Denmark
DKl
Dansk Ingenigrforening
113
The Netherlands
NDl
Stichting Materiaalonderzoek in de Zee
114
* Documents issued by government departments and agencies.
238
MTD Ltd Publication 90/102
Section 12 Review of recent experience in the North Sea
Cathodic protection of offshore structures
239
12. Review of recent experience in the North Sea
12.1
INTRODUCTION
12.1.1 First Survey (1980-1)
In November 1979, tbe Department of Energy established a Study Group to survey
current British practice in offshore cathodic protection and to advise on any action
required. One of the Study Group’s activities was the circulation of a
questionnaire to the 15 companies then operating some 75 fixed platforms in UK
waters.
Responses were obtained from nine operators and were representative of 33
platforms. The activities of these operators typified the whole range of steel and
concrete, oil and gas structures in both the northern and southern areas ofthe
North Sea.
A summary of the data formed part of Department of Energy Report ofthe
Cathodic Protection Study Group(’).
The Questionnaire contained 10 major sections, and sought information under the
following headings:
general details of the structure
.
details ofthe steel jacket
.
.
.
.
.
details of the concrete platforms
sacrificial anode systems
impressed current systems
hybrid systems
corrosion monitoring
electrical continuity of the structure
performance of the system
effectiveness of the system.
In addition, a final section asked for general comments.
Preliminary comments were drawn from an initial analysis of the responses from
eight operators. These were as follows:
“i)
Although the three types of system (sacrificiaI, impressed current, and
hybrid) seemed to achieve a reasonable level of protection, the sacrificial
method - which is hy far the most common - has tended to give the most
satisfactory results, However, there was some evidence of underdesign in
sacrificial systems and also of inadequate anode attachment in some cases.
Cathodic protection of offshore structures
24 1
ii)
The record of impressed current systems was less satisfactory, generally due
to the mechanical and electrical problems with anodes and cables rather
than to inadequate current distribution and protection. There was a
general feeling that these systems are less reliable and more costly to
maintain than sacrificial installations.
iii)
Analogous findings applied to hybrid systems : in these it was the
impressed current component that was less satisfactory.
iV)
Permanently-installed monitoring systems all seemed to have behaved
badly, particularly on account of cable problems, The newer method of
acoustic data transmission was therefore being watched with interest.
v)
Overall, the enquiry confirmed the anticipated need for improved methods
of system design."
These comments, and the tabulated responses from which tbey were derived, were
reviewed at a meeting attended by the Cathodic Protection Study Group, and some
of the operators who had responded. The foliowing additional conclusions were
reached:
"i)
It was generally agreed that current demand was bigher in the northern
part of the sea area considered, altbough some apparently geographical
differences may also reflect different installation dates and therefore
changed design procedures.
ii)
Further enquiries should be made to complete the information on hyhrid
systems.
iii)
Further information on the interaction of cathodic protection with marine
growths should be sought.
iv>
The fundamental aspects of cathodic protection should be further studied,
witb the aim of basing improved practice on a better understanding of the
processes.
242
v)
Better methods of modelling cathodic protection systems were very
desirable.
vi)
There was a need to reduce the costs of cathodic protection systems, while
maintaining their effectiveness.
vii)
Retrofitting is very expensive, so that initial expenditure on the design and
installation of a reliable system is often well justified.
viii)
It was felt that any further use of the results would require additional
clarification of the raw data. Unsatisfactory methods of monitoring
corrosion were thougbt responsible for much of the uncertainty in the
responses to tbe Questionnaire.
MTD Ltd Publication 90/102
k)
12.1.2
Notwithstanding the lack of information from some operators, the replies
were thought to give a fair and ty-pical view of the current behaviour of
cathodic protection systems in the North Sea. However, practice is
continuously changing, and improved behaviour may be expected in the
future."
Second survey (1988)
As a contribution to this Guide, the earlier survey was updated and extended by
personal visits to operators between February and July 1988. The second survey
was undertaken using copies of the original questionnaire, in an effort to collect
data similar in nature to that of the earlier survey.
12.2
OBJECTIVES
Within the available time and budget, it was not possible to attempt to obtain data
on all the platforms in the UK sector. The foIlowing priorities were determined:
to update the data from the 1980181 questionnaire for representative structures
and operators
to add to this sample of structures and operators additional data representative of
the main stream of developments in the UK sector
to add selected samples of structures that were either:
- novei but likely to be trend setting for particular fields.
- trend setting in respect of the structure design or the design of the cathodic
protection system or its monitoring provisions,
.
.
.
12.3
PROCEDURE
A total of 11 operators was approached to take part in the up-dating questionnaire,
and all agreed. Three had not participated in 1980/81 activities, although one had
subsequently taken over as operator of platforms which were in the original data
set. Only one of the nine operators in the original questionnaire was not
approached.
The original survey collected data on 33 platforms. The present survey updated the
information on 23 of these, and it added a further 25 new platforms or other
facilities. The 1988 survey sample represents 43% of operational oil and gas
facilities in the UK sector compared with some 44% in the 1980/81 survey.
Both the 1988 survey and the 1980181 data are referenced here only by individual
platform numbers.
In order to enable comparison ofdata from the 19801'81 and 1988 surveys, the
original tabulated format is followed with only minor changes, for clarification, in
the wording of the headings. All of the operators involved in the 1988 survey
agreed to the data being published in this format.
Cathodic protection of offshore structures
243
In addition to the factual tabular data, many of the operators’ representatives were
willing to give their personal opinions on the adequacy of their cathodic protection
systems and their preferences for future systems. Although these views do not
represent the considered view of the operating companies, they may be indicative
of future trends.
It is possible that the identities of one pair of platforms have been transposed
between the two sumeys, and that the updating of one platform is recorded under a
number different from that given to it in 1981. Footnotes in Table 23 indicate
these possible anomalies, but the conclusions now drawn are not affected by them.
12.4
RESULTS
Tables 13 to 22 present the factual data obtained in the sumey, and Table 23
presents the comments from the operator’s representatives
Some of the data represent the operator’s best estimate of information not available
or traceable in his records. This is particularly true of information regarding
surface areas of the structures concerned, details of the original cathodic protection
design, and the extent of consumption of sacrificial anodes.
12.5
DISCUSSION
The following genera1 conclusions can be drawn from the information collected:
Sacrificial anode systems continue to provide the most consistent and troubIefree corrosion protection.
Only two of the hybrid systems, one installed in 1979 and one in 1984,are
perceived by their operator to be satisfactory, and not to have required extensive
enhancement or retrofits. These two systems show chat, with the most rigorous
design and construction standards, hybrid systems are feasible, but they continue
to require a higher level of specialist supervision and maintenance than sacrificial
anode systems.
A significant proportion of even recently designed sacrificial anode systems was
deficient in the distribution of anodes in regions of high complexity or high
surface area density, such as conductor bays or pile sleeve area. These
deficiencies do not appear to indicate a requirement for mathematical modelling
or improved design parameters, but merely higher standards of detail design and
a more rigorous quality audit of the designs prior to installation.
One trend in sacrificial anode design, arising from an increased awareness of the
requirement for a high initial current density for polarisation, followed by lower
maintenance current density than stated in earlier design codes, is t h e move
towards longer, thinner anodes. This trend may extend the demands upon the
available manufacturing and metallurgical skills: anodes with growing longitudinal
cracks were virtually unknown in 1980/81,but they were increasingly a problem
in 1988.
.
.
244
MTD Ltd Publication 90/102
b
The 1980181 survey indicated poor performance with hard-wired monitoring
systems and revealed considerable interest in the then emerging acoustic systems.
From the 1988 survey emerges a remarkably improved performance from hardwired monitoring systems, presumably reflecting improved design and
construction standards.
Some OP the early acoustic monitoring systems performed badly, but later systems
appear to be adequate, although not reaching the high level of reliability of the
most recent hard-wired systems. The difficukies with the acoustic systems appear
to relate to communication, particularly in structurally complex areas and in
storm conditions,
The comparison of 1980181 and 1988 diver or ROV potential survey data points
shows clearly that significantly more data are being collected on the performance
of the cathodic protection system. The variety of techniques used and, in a few
cases, the incorrect use of techniques (e.g. the use of topside structure
connections in the measurement of pile potentials in order to determine the level
of pildstructure continuity) indicates tbat there is a requirement for higher levels
of technical quality audit in such survey procedures,
.
With very few exceptions, none of the operators was able to produce a single
unified document or file containing all the data required to complete the
questionnaire. Most had good access to annua1 survey data, and, for those
structures fitted with permanent monitoring systems, to monitoring data. Some
with very competently designed monitoring systems did not know why they had
these monitoring systems, at what frequency data should be collected by them,
nor the value of the data.
All of the operators, representatives interviewed during the survey in 1988 would
be assisted if the designers of the cathodic protection systems and any monitoring
system associated with them were to prepare a single "Operations Manual Maintenance" document as described in Clause 6.1.2.6.of Offshore Installations:
Guidance on Design and Construction(").
Cathodic protection of offshore structures
245
TABLE 13
GENERAL INFORMATION
FROM 1988 SURVEY OF PLATFORMS IN UK WATERS
Cunstruclion
Date
Design Connectionto
Water Design Date
installed system
life CP adjucent
depth life
(m)
(years) (19-)
commissioned ( p a r s ) structure
Gas drill
G a s drill
9ecl
Gas
Stccl
Gas
StceI
Oil drill
Oil prud
Oil drill/prad
Oil drill/prad
Steel
25
25
2s
25
20
20
S;u .wcHhcad
Gus drill
Gas conip
Oil drill/prod
Oil tlrill!prad
Stcel
Stet1
30
38
35
38
50
50
158
110
34
26
26
25
25
158
30
-
18
011
Srcel
Srcel
Concrrrr:
Concrctc
Coricrclc
143
15
I6
17
Oil tirill/prod
Oil
Oil
Oil
19
G89 cump
35
20
21
21
Gas conip
Stccl
Steel
Sled
Steel
Concrete
S1cel
Reference Type
number
I
2
3
4
5
6
7
8
9
10
11
I2
I?
14
GIs
Oil
Oi I
Oi t
Oi t
0
1t
23
14
25
20
27
Gas
Gas
2s
SICC~
Sttd
Stwl
Srwl
Steel
Steel
Steel
Stcel
Steel
S1ecl
Stcrl
Stcel
Stccl
144
I40
t 40
I40
I40
35
30
I38
I33
141
86
I20
2s
Stccl
Steel
Steel
25
25
110
120
130
120
Stccl
XI
31
32
33
34
G:ts
Oit
Oil
Oil
Oil
Oil
3s
Oil suhstxi
Stecl menifold I17
26
37
Oil drill
Steel
3s
Oil
39
40
Oil Ilu:rlin~
29
3 1
Oil prod
130
Steel
130
Stccl
113
Sted tethcrcd 49U
Cuncrctc
Iso
Steel
161
Stcel nianifold I53
20
25
-
20
25
25
25
25
20
20
20
30
15
73
74
67
72
79
80
76
76
73
74
80
77
77
20 +
74
71
74
20
71
75
20 t
25
30
7s
79
75
77
77
75
78
77
75
75
67
72
19
w
77
77
NONE
78
77
75
75
15
15
20 4-
20
2.5
Bridge
Bridge
74
75
74
75
86
15
20
7s
74
75
85
86
85
20
86
20
82
20
25
30
30
30
25
68
68
W6
86
87
a4
84
30
30
30
22
22
22
22
20
LS
20
20
20
20
25
20
20
41
41
oiI
43
<>:is prod
Cias wdl hc:Id
G a s drill
G a s prod
Eccl
38
25
1(4
78
81
82
75
Sled
Steel
37
30
N4
75
84
37
30
83
8:
Stccl
37
30
83
Oil
Ci:rs prod
GBS
G;ts drill
(.;as drill
Cias drill
Oil
Oil
Oil
Oil
Oil flaitiiq
Oil
Steel
I00
20
83
87
Sleel
85
84
Stccl
Stccl
26
26
40
40
40
40
40
84
Steel
29
29
29
Stwl
69
69
69
I I0
85
86
85
85
85
85
85
85
85
87
80
80
10-35
82
83
20
Oil
Oil suhsea
41
45
46
47
48
40
so
Sl
52
53
54
5s
56
57
58
Sleel
StlVl
Steel
Stccl
Stecl tethered
Steel
118
186
20
2u
11-16
11-16
11-16
15
15
20
85
85
85
85
xs
78
83
83/84
-
NONE
20
20
74
-
68
30m bridge
NONE
NONE NONE
NONE
25
25
NONE
25
30
30
30
30
NONE
NONE
NONE
30ni bridge
NOKE
80
25
25
S0m bridge
NONE
80
ti2
68
68
68
78
7w
50n1bridge
20
25
80/81*
30
30
78/79*
7x*
40m bridsc
30m bridge
40m bridge
40111bridge
2s
311
31)
30
77
-
20'
10
30
30
30
20
40
40
40
40
40
12-14
12-14
12-14
15
NONE
NONE
NONE
NONE
NONE
30m h r i d g
30m bridge
NONE
KONE
NONE
NOtiE
NONE
YONE
70ni bridgc
70m bridge
NONE
NONE
NONE
NONE
NONE
Notes
1980 data
'1988 update
1980 dam
1980 data
1988 update
I988 update
Temp CP 7/76 1988 update
1988 update
1980 data
1980 data
1980 data
* 1988 updaie
Temp C P 5/76 1988 update
I ONX updalc
1988 updatc
1988 undalc
1988 update
1988 updaic
1980 data
1980 da la
1980 data
* 1988 update
*1988 update
*l988 update
I988 update
1980 data
1988 upkite
1988 update
1988 update
1988 update
1988 update
1988 update
1988 update
1988 data
19x8 d m
I988 data
I988 data
19811 data
1988 dala
1988 data
1988 data
1988 data
3On1 bridge
30m bridge
30m bridge
30m bridge
1988 dat;i
1988 data
1988 data
1988 da ta
I46m bridge
1988 dara
Ix2Sm IxSUm, bridge 1988 dala
1988 dam
25rn bridge
1988 data
50rn bridge
1988 daia
NONE
NONE
1988 dam
NONE
1988 data
NONE
1988 data
NONE
1988 data
NONE
1988 dala
1988 data
NONE
t988 data
NONE
Notes
* d i m alkrcd by 1988 updale from that presented in 1980.
-
no rrsponsc 10 qoestiunn:iirr
over 20 years. a typicid rrsponsc
ID+
Thc data (or pl;itk)rm 16 niay relate LO Same platform prcsen[ed in 1988 ;is platrorni 38.
246
MTD Ltd Publication 90/102
TABLE 14 STEEL JACKETS
Reference Complexity Uia. of
number
factor
legs
no.1eg.s x
(m)
no. horiz
irames
1
32
3
40
40
Surface
area
Surface area Distance
submerged
in
stnl
sealmi
(m' )
ma)
0.84
0.84
0.84
3251
3251
325)
IS80
8770
56
0.99
1.75
1.75
z.4/9. I
1.2!2. I
1 .o
1 .0
1.0
2.0/4.8/6.0
58
1.?/2.4
14
28
1.515.7
1s
I9
20
22
24
25
26
27
30
7.3
4
16
24
24
24
24
5
-
G
I
a
12
12
12
9
10
11
12
13
28
24
14
-
?U
56
4.4i9.2
I .I8
40
40
T04.3
TO 6.0
4w
56
1 .(I
12
24
28
28
28
29
30
31
32
33
34
35
0.9
0.9
1.714.3
I .714.3
1.7143
1.714.3
6400
71800
46450
1956
2798
2798
80000
59620
52380
521 16
38000
38000
66000
49000
42150
60000
3250
2320
930
37000
37000
37000
37000
of 5 t n l
465
465
465
525
5750
1900
18915
26000
981
1377
1377
-
8205
51 13
2230
13000
13000
6950
5200
7750
I80QO
1580
560
560
3700
3700
costcd
Mow
waler
Zn siliatelvinyl
Cod Lar epoxy
C o d tar epoxy
-
-
5
5.8
10
5
5
-
120
506
117.
8366
S
5
37
38
48
2.5
73900
6275
17615
10
39
24
1.8
40000.
-*
32*
-
I
83406
841 1
-
8*
43
44
4s
24
12
24
14
1
1.5
3946
5440
1050
1.5
10960
2586
1.5
20hY
49
40
16
28
2.3/4.u
1.611.8
1.7
7228
6rlooo
50
8
51
8
8
20
20
20
52
53
54
5s
56
57
58
'5
24
-
-
-
Glass flake polyester
-
jArnercoai 99
Coal tar epoxy ( 5 0 0 ~ )
-
L
\Epoxy primer (25)
Coal tar epoiy (ISO)
Coal iar epoxy
Coal tar epoxy
Coal tar epoxy (475)
lCoal tar epoxy (625)
Coal tar epoxy to -10
/High build cpoxy to -4
Glass Rake epoxy to -5
-
-
Subsea tcmplaie/manirold*
Coal tar epoxy
Coal tar epoxy
High build epoxy + 4 to -4 m
Floating hull only 32m draft
2n Phosnharc to -32
42
48
Tideguard I71
Tideguard I71
Coal tar epoxy 1300)
Coal tar cpovy (300)
Coal tar epoxy (300)
-
46
47
Tideguard I71 LO -5.8
Amercoat 99 10 -3.0
Coal Lar epoxy {500)
25
2s
1.07
2.0/3.0/5.0
2.0/3.S/S.U
-
Glass flake polyester
-
Coal tar epoxy (500)
25
120
-
-
5
-
-
41
-
10
4
3.6
-
6740
Glass flake epoxy - Splash zone
Glass flake epoxy Splash zone
Coal tar epoxy
Splash zone
Coal iar epoxy
Splash zone
-
32
12'
56
48
36
(m)
-
3700
-
Above water
splash zone
coatings
lir
(m)
120
I20
3700
T y p l of coating
(Licknrrr microns)
depths
11175
1.6/2.0
142.0
1.6i2.0
-
prime (700) Subsea ttrnpkdte/rnanifold
(450)
.o
I .45
I .45
1.45
3
153*
13551
5177
9806
-
9090
9WO
I8027
8.5
2.6j9.6/30.5 125216
I620
20000
12252
344%
1915
1915
191s
-
I
-
-*
2 I375
5
5
5
5
10
2
2
2
2
2
4
4
4
3
12*
186
Epoxy
Coal tar epoxy
Coal tar epoxy
Coal par epoxy
Vinyl tar
Cu/Ni Cu/Ni
Cu/Ni Cu/Ni
Cu/Ni Cu/Ni
Cu/Ni Cu/Ni
Cu/Ni Cu/Ni
Coal tar epoxy
Coal tar epoxy
Coal tar epoxy
Coal iar epoxy
Cod iar epoxy
Cod tar epoxy
EPOXY
Coal tar epoxy
Coal tar epoxy
Coal tar epoxy
High build epoxy -3 to
+ 5 rn
Coal iar epoxy
Coal iar epoxy
Coal tar epoxy
Coal tar epoxy
Floating hull only 22 rn dran
Coal tar epoxy
Kotes:
Data iiltcrcd hy 1988 updatc fronl thnc presented in 1980
- no response to qucstiorrn:iire
Cathodic protection of offshore structures
247
Reference lksign
iypc
numbor
16
Kslcnl
Columm Surface
No. DIA armsteel ofcoating
(m) skirts,etc steel
(m’ 1
Condeep 3
-
-
Costing
uwd
Skim
Coal lilt
uncoated.
All other
cpo~y
Rcinfornment
Ihskirts
Are post
Risers. conductors
E:leelrirally Calhadically hsrc CP
tension tcndons
caiswns etc
bonded?
protected?
Present? Anchor Ucctricslly Separntcly
puinls bonded?
csthodicrlly
NO
NO
YES
NO
-
NO
YES
NO
NO
-
-
-
NO
YES
NO
WV
YES
NO
-
NO
YES
YES:
YES’
YES
YES’
YES’
NO‘
NO
-
-
NO
YES
sitachrneots
uncoated
17
Seatank
4
-
-
-
IS
Condeep 3
-
3716
Skins
23
Gravity
12650
Cortcd
-
-
2#
I4
41.8
-
Cnal t i r
uncoated
cpoxy
All other
atiachrnenis
uncoated
Zincspray YES’:
and
coal Iar
epoxy
40
Seatank
4
-
-
NO
I
Not-:
data altered by 1918 updated from that presented in 1980
# concentric
interconnected - “equipotrntial nelwork“
- no response to questionnaire
:
248
MTD Ltd Publication 90/102
TABLE 16 SACRIFICIAL ANODE SYSTEMS
Relerenn
nmber
I
2
3
4
5
6
9
10.
I4
I5
17
18
23
25
26
27
Anode
material
Amde
number
250
289
Al
Al
Al
Al
Al-Zn-In
Al-Zn-In
Al-Zn-In
Al-Zn-In
Al-tn-1ig
hl-Zn-In
hl-Zn-In
Al-Zn-In
Al-Zn
166
54
747
427
48
61
I356
160
Individual
amdr WT
(ex core)
Anode originnl
dimensions
(years)
(ks)
(m)
1980
15
15
15
I48
148
I48
I48
0.2xo.2x I.5
0.2x0.2x I.5
0.2x0.2x I.5
O.lx0.2x I .5
I48
148
300
-
10-20
10-20
10-20
10-20
5-10
I380
15
20
20
20
20
20
25
25
25
30
I233
2500
2s
20
-
Al-Zn-In
Al-Zn-Hg
Design
life
+
+
-
1%
362
240
28
29
Zn
30
Zn
31
Zn
32
Zn
33
Zn
Al-Zn-In
Al-Zn-In
34
35
98
780
800
820
800
-
30
22
22
22
21
64
-
36u
-
I60
IS
69
36
20
37
20
38
40
41
hl-Zn-Hg
hl-Zn:ln
At-zn-ln
2508
-
/;::#
160
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
58
Al-Zn-In
ALZn-Hg
Al-Zn-In
Al-Zn-In
Al-Zn-In
Al-Zn-In
AI-Zn-Hg
Al-Zn-Hg
Al-Zn-Hg
Al-Zn-Hg
Al-Zfl-Hg
Al-Zn-In
Al-Zn-In
Al-Zn-In
Al-Zn-In
Zn
60
:1
304
205
306
205
1876
1030
336
534
495
495
476
540
468
20
25
20
(Now est'd)
33
20
360
360
360
20-200
288
226
216
950-1 I50#
12.4
120
20
30
30
30
20
40
40
40
40
40
o.133xo.I3x).ss
0.27~0.3~2.44
0.23~0.17~2.44
0.2IxQ.2 lx2.77
0.JxO.ZQx0.24
0.9x0.18x0.08 .
0.3xo.24xo.24
0.46~0.IxO.l
0.5XxO. 13~0.13
I5x0. i 8~0.19
I.5xO.l8xO. 19
1.5~0.18~0.19
I.SxO.18xO. 19
Unknown
Unknown
-
0. I91x0.2 1 6x3. I60
0. I 9 1 x0.2 I6x2,s
0.2 lxO.225x2.0
0.76Q1.376 din
a10. IH7-0.665 length
0.635~0.175~1.25
0.064~0.14~0.61
0.159~0.184~
I. 55
I
-
10-95
10-15
10-15
10-15
10-15
-
I
-
-
43
434
434
290
354
354
354
-
200
0.26~0.265~2.75
0.26~0.265~2.75
0.26xO.26Sx2.75
0.227~0.227~2.6
0.25~0.25~2.6
0.25~0.25~2.6
0.?5x0.25x2.6
0.25x0.25~?.6
0.25~0.25~2.6
0.193xo.193x2.75
0.193~0.193~2.75
0.193xo.193x2.75
0.2 I6x0.2 16%1. I
-
12-14
12-14
200
-
15
-
ZZUS
20
284
-
+
40
15
-
-
-
-
-
2.5-50
-
10-1s
60
95.
0.I7xO.17~
I .2
200
25% Max
-
126.5
12-14
0.1
15
2.5-50
2.5-50
0.207~0.207~2.75
354
-
-
10
2.8x0.26 dia
354
0.1
Unknown
-
300
0.1
0.2x0.2x3.0
0.23~0.27~2.44
-
{;:
Unknown
Segmented Bracelet
0.13x0.13xl.S25
2.8x0.26 din
;1;
(4
25% Max
-
Anode
sfand4
1988
Unknown
10
.= 5*
0.22~0.22~2.4
328
326
330
2&200
20-200
Frrirnate%
consumptionandm
-
30-40
30-40
30-40
30-40
Nane
None
None
None
None
None
None
None
None
10
2.5-50
20%
20%
20%
0.261
IOV"
None
None
Nonc
None
0.1
5
5
5
5
5
€5
€5
€5
Minimal
0-10
Unknown
Back & sides
None
None
Unknown
Unknown
None
None
None
None
Back
Back
Back
0.4
0.6
0.025
-
IO?A
None
0.5
None
None
0.4
0.135
Unknown
Unknown
Unknown
Unknown
Unknown
None
None
0.025
0.3
<5
30%
10%
None
None
None
0.1
0.2
0.2
0.1
Unknown
Unknown
Unknown
Unknown
Unknown
Extent
of coatin(
on anoda
back
Back
Rack & sides
Back & sides
Rack & sides
Back & sides
None
None
None
Nonc
None
None
None
Unknown
None
None
None
0.4
-
0.4
-
None
0.3
0.3
0.3
+
0.3
Unknown
Unknown
Unknown
Unknown
None
None
0.3
0.3
None
None
0.24
(1.2
0.2
0.2
0.2
0.2
0.254
0.254
0.254
None
None
None
None
None
None
None
Nonc
Nonc
None
Back
-
0.025
Notes:
Retrofit anode system installed in 1980.
Retrofit anode system installed in 1984/5. Original accelerated consumption attributed to impressedcurrent interaction horn adjamnl
pipeline CP system.
# flush mounted hricelcts on different tubular sizes.
- No rcspnnse to questionnaire
+
Cathodic protection of offshore structures
249
CJ
m
0
Reference Anode
fixing
number
8+
Retract
Uistance
Are dielectric
from nearest shidds fitted?
mernher
lype
tm)
4.1
through
tubc
Nor 00 structure
GRP on tubc cnd
Anvdc
material
Pt wirejNb
rcplaced
Anode
number
Total
installcd
Current current
(A)
(A)
+
Kurnbcr of
transformer
rectifiers
Output
rating
(A)
6
Manual
Signal source
for automatic
automatic runtrul
or
(V) conlral
1400
40
Auto*
Ag/AgCI
reference
electrodes
Unknown 3
250
12
Mannel
-
40
3 SO
I
350
36
Manual
-
I50
1800
6
300
13
down
rated
to 150
55
100
8400+
( 5 500)
9
-
Polyethylene heat- Pt Wire/Nb 9
shrink slccvcs
2mm thick
198l/2
PtlNh
lo**
I1
Rigid on 3.0
structure
Retract
Glass reinforced
cpoxy
Pt/Pb
3.6
through
tube
19
Rigid on 4.5
structure
20
Rigid on I
structure
Epoxy mastic
2 x 13m
Coal tar epoxy
4 x 15111
Pb/2% Ag
12
down
rated
to 7s
Notes:
z
*
**
+
++
-
K
7z
iP,;'
c.
0
P
Operated as manual systeiii
Impressed current system hilcd. Kcplaccd with sacrificial system see Table 16
Impressed current systrm partially Failed. Supplemented with sacrificial systems - see Table I 8
1988 correction
N o response to qucstionnaire
-
Origintilly Was dual
auto, now ng/AgCl
manual
- Zn
Uef
nmnkr
Anode
fi%iny
Distrancr
IMcrtric
hum nexresr shields?
rnnnstr
Anode
mrccriil
T p
,\nude
nurnkr
(m)
7
U
Kctractable
rhrough tube5
to
3m long
GRP on
61
r u b erds
4.6
k;Nb
Src'fimblc 17
for I9UO dakb
Now hybrid
0
a
1.1
u1
P
e
GRP on
;mode
mpporr
Cbd
Pt/Nh
48
Tnral
Number of
i m p r d tnrafwmer
Currcw current
mtitien
(A)
(A)
ins
48
100
216
6
22
5n
1200
(rcducud
to 2100)
1 nxm
Pr!Ti
coated
Uulpl
Manual or
ntiw
automatic
rontrol
(A)
(\'I
Sacrificial
Signal source '4 of
l
anudr
for autnmalic ~ o t aCP
control
pruridcd mrtrrial
by I C
Sumber ur
rscrikial
anndrs
Ihtributiun d
nrritirid
533+
Upper JOni
309
Splesh zone/
conductor guider
479
:inti main node!.
smdrh
1J
reduced
6
10
50
6
1400 40
Manually
87
17
9
I
I
50
Aucom~tiu Zn
with manual electrodes
over-ride
15
4
14UO J3
Auiomaiic
opcralcd
miinually
75
Auiorn;itic
operared
m;inually
90
AI-Zn-Hg
rcduccd
Al-Zn.lim
10 60
50
40
32
12
R
I5
10
CD
GRP on
rube ends
22
sU.i bed
rlctlr
65
NO
Pi wire/Nb 42
rcplxcd
Ircduced
r r m 1979 10 2s)
Pr!Kh
4
Prpb
pkad
and
135
200
700
2800
4
1000 36
I?
600
(reduced
to 6 plus
3 Slandhy)
7 ?W
tl
I200 50
Al.2n.h
Retrolit lor upper
43111 and pilu
IlLYKI
24
hhcd
sleds
3Y
liull
rnounlcd
PiINh
cl'id
75
and
1 so
NO
-
O.ObKl.2~47111 P1,l-I
GRP t coaling plated
I?
(rCdUCCd
to 5 plus
600
7200
6
1200 51)
h)
Y!
x
+
-
80
Al-2n-lit
650
40
Al-Zn-Ill
91 lonnc
Aulornatic
90.100
Al-Zn-In
240
reduced
10
I Standby)
(ro
70
4200
70
Noln
**
Au~om~tic 2n
opcritcd
e l ~ wrodcc
manuully
Rctrnlir syrlenr inwllcd to rrpl;iw cnmc f d e d impressed current :indcr/tubs IY(IO/I
Additional rcrrofil system iiiiralled 10 rcplaoc more failzd/d;lm3gcd impressedcurrent nnodcrjtubcs 198315
Rcrrulit racrificial sptmi to tnmprovc currcnl distribution i n IYUIjS
Wcrrnfit s;icriticial systcnis to improw current distribution in 1981186
Was imprcrscd current system. Now hybrid. Retrolit drcplacement ianprerud crtrrcnl m d sacolicial systcmr in IY7X/79
NOresponx lo yueslionn.iirc
I2
Zn
elcct rodcr
+
Splash zone/
conducror guidcr
;ind n i m n d c r .
Rclrnlih a1
selected locations
F~cdominanfly
at
nodes
TABLE 19 MONITORING AND SURVEYS
(a) permanent monitoring systems
Reference
number
Reference C(0rlrodcs
fitted originally
Kutxr
norking
transmission Sacrificial
1,'P
a t &It
method
Ihta
A r r anode C u r r C n B
anodts
of suvey
I
None
NOW
3
4
None
5
6
7
8
9
10
II
12
13
14
IS
16
NOW
NOW
Dual Zn & Ap/AgCI
AdAgCl
Ag/AgCl
None
AdAgCl
Ag/AgCI
-
-
-
-
- -
-
-
Yes
NO
None
2
2
Cable
Cable
Cahlc
Cahlc
Cablc
4
20
Cable
Cahle
Cable
Cable
?I
-
-
22
36
18
25
Now
25
21
NOW
25
16'
10'
Nonc
NOW
-
-
30
8
28
10
28
35
Nanc
34
Zn
Zn & Ag/AgCI
Zn
Zn & Ag/AgCI
Zn 8; Ag/AgCl
3s
Nonc
31
32
33
36
Zn & Ag/AgQ
37
Zn B AgzAgCI
Zn & Ag/AgO
38
39
40
41
42
43
44
4s
46
Zn
47
48
49
50
5l
52
53
54
55
56
57
58
-
NOnP
Nonc
Nonc
25
L
20
34
32
Unknown
28
28
40
36
Cablc
Ca blc
Cablc
-
Yes
No
Yes
YCS
Yes
No*
NO
No
Na
-
-
-
-
NO
-
NO
Yes
Yes
Yes
NO
NO
No
KO
No
No
-
-
-
Yes
-
NO
-
-
No
No
Daily'
Weckly
-
Evcry 3h
Wcckly
I
Monthly
Wcckly
Wtckly
Weekly
-
No
No
Monthly
-
-
-
-
-
-
-
YCS
Wcckly/Now dcniriid
-
-
-
-
-
-
-
Daily
Daily
Daily
Yes
YCS
NO
NO
NO
Cahlc
Acoustic
-
-
-
-
-
Acoustic
No
Yes
Monthly
Acoustic
No
YCS
Monthly
Acoustic (Interrogation by dipping:.Acoustic and sortwdre problcms never rully
commissroned)
Gable
NO
YCS
Yes
Ddrly
-
Z n & Ag/Ab<l
Zn & A g / A K I
Z n & Ag/AgCI
15
14
14
14
14
Acoustic
Acoustic
Acoustic
None
-
I4
Z n & Ag/AgCI
Z n 8; Ag/AyCI
Zn & AgJAgCI
Zn 8; Ag/AgCI
ZI1& AglAgCl
22
22
II
17
KOIX
WOW
-
-
-
-
-
-
-
-
-
-
-
Yes (2)
Ycs
Daily
NO
NO
NO
NO
-
Daily/Now monthly
Yes (6)
Yes
11
Cable
Cable
Yes (4)
YCS
17
C'ahlr
17
1
4
Yes
YCS
1f$
Cahlr
Cable
YCS (4)
Yes (4)
Yes (4)
YCS
Daily
Daily
Daily
Daily
Daily
-
-
-
-
-
-
-
-
YCS
Evcry 12h
Yes
Wcckly/Now monthly
17
-
-
-
-
-
-
-
-
5
2
5
2
Cahlc
Cable
4
1
36
34-
Acoustic
Ca blc
soles
* Rtviscd data in 1988
' Mosf h i k d by cut cahlcs during hook up
# Ag/AgCI failed. possibly by contamination with drilling mud
252
NO
Yes
Yes ( I 2)
No
-
-
Zn
f n & Ag/AgCI
Zn B AglAgCl
NO
-
Now
h'omc
Z n C AgJAgCI
N O
Uahlc
-
NOW
No
No
Ciihlc
Cable
None
None
None
-
No
Cahlc (cahlus cut uff during hook-up)
Acous~ic
Nn
Ycs
I
2w
29
-
No
YCS
2
42
26
No
-
NO
4.
28
b
6
Zn
Zn 8; Ag!AgCCI
Yes
-
No
No
-
-
23
24
Frqucnry d record
NO
-
No*
Cable
12-1s
NO*
Cable
- -
28 zinc#
G
6
Yes
Acoustic
19
13
12
-
IY
recorded?
No
No
No
-
IS
eurrmt
YCS
5U
41
AgjAgCl
6
None
Zn
19
Ncinc*+
None (Systcm dcstroycd during platform iiistallation)
17
autornstKslly
-
Now
None
None
Zn
Is data
Impred
modes
(KO-of munilorcd snoda)
No. (1980or lpilnl
2
rnonilored?
-
-
-
Yes (6)
5
+
.
YCS
-
-
Ddil)'/NOW
monthly
-
-
-
flcutrodcs dclibcr;itcly rcmovcd for platform modific;ition
Conduits IOSIin splziah zonc
One darnaEed 31 flo:a out Second assumed cablc Cailurc
MTD Ltd Publication 90/102
TABLE 19 MONITORING AND SURVEYS
(b) surveys
I
ANNUAL
1
ANNUAL
3
ANNUAL
4
5
ANNUAL
ANNUAL
6
ANNUAL
7
ANNUAL
LI
ANNUAL
9
MONTIILY/
ANNUAL
10
.HOKTIILY/
ANNUAL
I1
MONTHLY/
ANNUAL
I?
ANNUAL'
1'1
A N N U A L MIN
14
A N N U A L PART.
15
APWUAl. PART'
Diver hcld elccirode.
Conwcl & read topside.
Diver hrld clccirode
ramp1riser then local
mnracr/rrad*.
Diver held clecirodr.
Cnnmci& read topside.
Diver hr!d eln.trodc.
niver held electrode.
Contact by spike or
topside. Read topside.
Contact by rpikc or
topside. Read topside.
Divcr dr ROY. Contact
topside or spike i i
iroletd. Rcad topside.
Diver & ROV. Contact
topsick. Rcad topside.
Dipped electrode/
topside contact d; read.
Diver hrld & m d
I prr5m
Ip 5 m
-
I
-
Occasionally
-
-
-
--
5-10
5-10
5-10
5.10
on20c12
-
-I2
12
rnewr.
I6
17
Dip+ rlmimdcl
topside contact& read.
Diver hrld & wad
meter.
Dipped rlcrtrockl
topridc CO~IBCI
B rrad
Diver held & mad
Spike c1.1rnp.i'
ROV carried clrclrode.
Topside conncciion.
D m video.
ROV clcctrode. Sreb
connection &top. Diver
mcter. Video.
NO CATHODIC
PROTECTION.
NO SURVEY.
Dip rrll (no access)
Dip cell (no access)
Divrr held meter.
Seab connection.
Diver hcld meter.
Stab conncrrion.
ANNUAL'
ANNUAL'
ANNUAL
20
ANNUAL
?I
??
NOT REPORTED
Mainly ROV. Topside
ANNUAL'
connection. Video.
Same arb.
ANNUAL.
Somc d i w hcld.
Topside connection.
Visual on concmr.
Mainly ROV as 22.
Some d i w hcld
ANNUAL'
Topside connection.
Visual on concrete.
.Mainly ROV as 22.
A N N U A L PART' ROV clcc~ralc.
Connection sub & rop.
Diver meter. Video.
NOT REPORTED
Diver hetd meter.
ANNUAL
Slab connccrion.
Diver held mcttr.
ANNUAL
Stab connection.
Divcr hcld mctcr
ANNUAL
Slab conntclion.
Mainly ROV. Top
ANNUAL.
ccrnncrlioii Video
ANNUAL.
24
25
26
27
28
29
30
31
12
9
9
Sckcrcd
f
mclcr.
ROV cilrrred elcclrcdc.
Conlacr lop. Rend lop
ROV rarried etrclrode.
IS
19
23
6
3
3
m
3
Occnsionally hiinimzl
I
I
Per 2.5m
Minimal
Occasionally Minimal
Minimal
Per 2 Jm
Minimal
Occasionally Minimal
Minimal
5-10
Mimrnal
Dccarionellv
-
k r 5tn'
Minimal
NO
-
NO
Per h'
Minimal
NO
-
NO
Per Sm'
Minimal
NO
-
NO
I
I
-
Limited
Limited
Per IOm
Per IOm
Limilcd
Limrtcd
Some sub.
Cathodic protection of offshore structures
253
TABLE 19
-
MONITORING AND SURVEYS
(b) surveys continued
Rtkrewc R q u e n c y
number
olsurrty
Mleihrd of measurement Typiral nu. of
.I2
33
ANNUAL..
A M NUA I..*
I)ivrr hcld 31 selcctcd
areas. risers, clamps.
diirnagc. Mainly ROV
24
ANNUAL
35
?b
ANNUAL.
A N N U A L I’AKT
potential
Mnin
measurments tubular
p r surwy
83
-
3
3
Critiral
~mds
I
I
Typical m. of plcntiil mesuremenn on each:
Riscrs
Coahrtors Piles
J-Tuk
Per !Om
Per 10m
Limited
Limited
I
I
--
Cahns
Limited
Limited
30.
3a
ASNllAI.
39
ANh’UAt.
40
AfWliAl.
41
EVERY ?.4
ROV or diver. Mainly
top. Some stab. Mainly
top rerd.
KOV. Stab colinection.
ROV ropride. Video.
Some d i n r held/a~b.
ROV topside. Video.
Some Jivcr hcldlaoh.
KOV k diver. All
topside coiinectionsl
repd. Vidco.
ROV. Topside for hull.
Stuh nn tcthcrs.
Dip cell (was
(I-nionthly)
ROV. Subconnection.
J?
YEARS
EVERY ?-4
ROV. Sub conncchn.
20-30
-
-
-
-
43
YEAKS
A N N UAI.
I so Plus
a
52
9
I
44
ANNUAL
Diver hcld instrucfinns.
Sfab cxcept risers.
I)ivcr hcld instruments.
8
NI.4
9
I
8
-
9
I
8
Y
N/A
I
All
Conrinuous Conlinuous Each (fop)
Continuous Continuous
to-20
I F r 2m
kr?m
lnlout
Per ?m
Per ?tn
10.20
-
-
Each (top)
In/out
Per 2m
Per 2m
-
10-20
-
Per 2rn
Each (top)
In{0ut
R r Zm
Per 2m
-
10.20
-
Rr 2m
Each (top)
In/out
Rr 2m
Per Zm
-
10-20
-
Per 2111
Each(top1
In/out
Per 2111
Per 2m
-
All
Per I Sm
AnOmakS
Each(stab)
-
5m
Per 5m
37
AKNUAl. PART
2000 on commissioning20 + thcrcafici
All
All
Continuous Continuous 50% Stab
60-80
+
60-80 +
Anomdlirr
All
Continuous Continuour 50% Slab
> 2oOo
All
Continuous Continuous Each (top)
> 500
-
-
-
-
All
Anomalies
Iso
hnonielics
Diver held instruments. I50
Sub cxwpt risers.
hriomelics
niver hcld instrurncnu. I50
Sia b except riscrs.
z ?ooD
Diver & KOV. Top
c(mnectiims/rcad.
Viden.
Diver electrode/top
conncciion & read.
Isohted stab.
Diver clccirodciiop
L;lb except risers.
45
ANNUAL
41
ANNUAL
47
ANNUAL
48
ANNUAL PART
49
connection B read.
1sol;rted stab.
so
AWNUAL M R T
Sl
AWNUAL PART
51
A N N U A L t’AR‘l
53
ANN UA1.
54
ANNUAL
5.5
ANNUAL
56
ANNUAL
57
ANNUAL
5s
AMNUAI.
13ivcr elec~rode/top
conncctim & read.
lsolirletl rceh.
Divcr elcctrndc/tup
cunnectioe k rc:td.
Isolrted stub.
Diver cleci rode/top
conncaion & mad.
Isuhad s t h .
M;iinlg ROV. Top
connccfioo,kead. Video.
Some divcr hcld nieter/
sieh. Video.
Samc diver held tncterJ
si:lh. Video.
Mainly ROV.Top
connection. Video.
Diver held meccrlsriib.
ll/C sysrenr switchcd olfl
Mainly ROV. Top
Conlinuous Continuous
and slab
Continuous Continuous
and stab
Continuous Continuous
-
I
-
All
Rr
15m
Anomalies
Each(stab1 -
-
All
Per ISm
Anomnlies
Each [stub)
-
Per Sm
-
I
Per
Limited
I
-
Limited
lorn
Only to chcck calibrate pcrmaneni reference elcctrodes
> to00
All
All
All
Each (stab)
All
All
conneaionlrciid.Video.
Nnm
-
254
diiia alicrcd by 1988 updore from that presented in IYSO
no response to quenionnaire
MTD Ltd Publication 901102
Kcferenm
number
1
2
3
4
5
6
7
8
9
10
II
I2
13
14
IS
16
17
18
19
20
21
22
23
24
2s
26
27
28
29
30
31
32
33
34
35
36
37
3a
39
40
41
42
43
44
45
46
41
48
49
50
51
52
53
54
55
56
57
58
Sacrificial, i m p r d
or hybrid syaem
Rkm
Conductors
Sacrificial
Sacrificial
Sacrificial
Sacrificial
Sacrificial
Sacrificial
Hybrid
Imprcssed/hybrid*
Sacrificial
Sacrificial
Imprcsscd
Hybrid
Hybrid
Sacrificial
Sacrificia1
Nonc
Sacrificial no data 1980
Hybrid
Impressed
Impressed
No data rcportcd 1Y80
Hybrid
Sacrificial
Hybrid
Sacrificial
Sacrificial
Sacrificial
Sacrificial
Sucriiicial
Sacrificial
Sacrificial
Swriliciill
Sacrificial
Sacrilicial
Sacrilicial
Insulated
N/A
Insulated
Insulated
NIA
Insulated
Bondcd
Bonded
PC
NIA
PC
PC
PC
PC
Bonded
Sacrificial
Sacrificia I
S;arificia I
Hybrid
Sncrificial
S;icri ficia I
SncrificioI
Sncrificial
Sacrifici;!I
Sacrificial
Sacrificial
Sacrificial
Sacrificiiil
Sacrificial
Sacrificial
Sacrificid
Sacrificiai
Sacrificial
Sacriticial
Sacrificial
Sacrilicial
Imprcssc
Sacrificial
N/A
Bonded*
Bonded
Bonded
Bonded
Insulated
-
Bonded
Bonded
PC
PC
N/A
Bonded*
Bonded
J-Tubes
Caissons
Pila
PC
PC
PC
PC
Bonded
PC
Bonded
Bonded
PC
PC
PC
PC
Bonded
Bonded
PC?
-
-
NIA
Pc*
Bonded
Bonded
Bonded
Bonded
Bonded
NiA
NIA
PC
-
-
Bonded
NIA
NIA
N/A
N/AL
Bonded
NIA
PC
-
-
Bonded*
-
Insulated
Bonded
Bondcd
PC
lnsulaled
Insulatcd
PC
PC
Insulated
PC
PC
Insulatcd
PC
PC
Insulaicd
PC
PC
Bonded
Bonded
Bonded
PC
Insulatcd
PC+
PC
Bonded
PC
PC
PC
PC
PC
Bonded
Bonded
Bonded
PC
Insulated
Bonded
Bonded
Bonded
PC
-
-
-
-
Insulated
PC
PC
PC
PC
Bondcd
Sea bed
manifold
Bonded
Bonded
Insulated
PC
lnsulaied
Pc
Sea bed
manifold
Insulated
NlA
N/A
Insulatcd
Insulated
Insulatcd
N/A
Insuliikd
Insulatcd
Insulated
Insulated
-
PC
PC:
PC
PC
PC
Insulated
-
-
-
-
-
PC
PC
PC
PC
PC
-
-
Bonded
Bonded
PC
NIA
lnsulaied
PC
Flow lines and
vcll tube
Manifold
Bonded
8onded
Bonded
Insulated
NIA
Insulated
PC
-
-
PC
PC
N/A
NIA
NIA
Insulated
NIA
Bonded
NIA
Bonded
Bondcd
Bonded
PC
N/A
N/A
Bonded
Bonded
Bonded
-
-
PC
-
PC
PC
PC
PC
-
-
PC
-
-
PC
PC
PC
PC
PC
-
-
PC
PC
-
PC
-
PC
PC
PC
PC
PC
PC
Bonded#
Bondcd#
Bonded#
Bonded#
PC
Bonded
Bonded
Bonded
PC
Bonded
Insulated
PC
PC
PC
PC
PC
PC
PC
PC
PC
PC
PC
Bonded
Bonded
Bonded
Bonded
Bonded
PC
PC
PC
PC
PC
PC
PC
Bonded
Bonded
Bonded
PC
Bonded#
Bonded#
Bonded#
Bonded#
Bonded#
PC
PC
PC/Bondcd#
PC
PC
-
PC
PC
Not-
*
daia al[cred by 1988 updatc from that presented in 1980
#
one insulated wiih rcsist;me conrrolled bond
piles houdcd after surwys shows some discontinuities
no rcsponse t o questiariniiirc
presumcd contact
+
-
PC:
Cathodic protection of offshore structures
255
Reference
number
S~~tcm
type
Performance
satisfaciory.
KOrepairs
or mods
Haw wcre
problems
What problems
wcre detected?
'"yp of
failure
Prernt levels of protcdion
Date
Potentials
negative millivolb
Least
Most Range 90%
All with mpcct to Ag/AgCI
Action
taken
SUrnmary
Replacc
Good
sjrto
970
1003
970-IOOO
Mechanicnl
Mcch/
consumption
-
None
Good
Good
87
79
313
930
1000
ions
425-980
930-1005
79
Maintain &
retrofit sac
ItetrofiI sac
Y30
ROO
800
674
100s
1050
MLxhmicnl
Good
Good
Good
Border
87
80
80
930
900
1180
to00
950
I000
1000- 1 050
950- 1000
950-1000
detected?
~~
sac
Yes
Ycs
SURVEY
3
SlC
N O
4
5
6
Sec
Sac
Yes
Survcy
in 1977
-
soc
Hybrid
YCS
-
No
Moniror/survcy
Mechan ic;lIjconductors
Monitorjsurvcy
cablcs,/junaion boxes
Mcchnnical!anode riser
I
2
sitc
7
Localised under
proteciion
-
Hybrid
No
ssc
Yes
-
SX
Nil
Moniror/survey
I1
lmprcsscd
No
Routine maint.
12
Hybrid
Ycs,/No
Monitoring
Original i/c syslcm
cables
Anode supportsjbroken
cclls
8% anode rods bccarnc
loose
Anode &cable failure
13
Hybrid
No
Monitoring
14
sac
No
Survey
IS
ShS
16
17
18
None
Sac
I9
20
lniprcsscd
Hybrid
Yes
Yes
Yes,!No
-
21
-
Yes
Yes
-
-
22
Hybrid
No
Moni tor,inrrvey
24
25
soc
Hyhrid
sac
Yes
No
NO
26
27
sic
-
Sac
N0
28
29
30
SX
Yes
SX
Sac
sac
Yes
Yes
Ycs
YCS
Yes
31
32
33
sac
SilC
Undcrprotcctionconductor hay
-
-
lmprcsscd
23
0
t
4
2 Anodes inissing
9
10
8
Y
Ycs
Anode fell
I
Moniror/survey
Survcy
Survey
Minor dcficicncy
in anodcs
-
Off
-
Mechanical
-
Replace 100%
-
-
Line
Good
Good
87
87
87
1050
1270
950- IOOO
950- IOOO
800-I 100
Mechanical
Full sac
Good
rctrofit
Mcchiinical
Rci m a t e
Good
RO
950
I000
Y 50- !000
Mechanicid
Ti ghtcned
Good
87
970
t 16s
990- I 146
Mechanical
Hybrid
rctrofit
Good
87
900
1200
YOO- I050
13csign
Retrofit
Good
87
860
I020
Y 40. I 000
Good
87
903
935
-
Good
Good
87
87
Good
Good
Design
-
93s
903-935
YO3-935
903
935
903-935
79
79
800
800
I000
-
1000
900-950
900-950
Good
87
8 50
119n
I 000- 1050
Good
Good
87
us0
860
950
87
I I37
850-950
950-1050
-
-
903
Distribution]
early T-R problctns
Design/
Relocate/
tncchanicnl
rcpeir/
-
-
Distribution!
early T-R problems
Cn hle dniiiage
Design:
mechanical
retrofit sac
Rclocaie/
Underprotection
Dcsign
Blanked
Good
87
836
1012
900- t 000
Early
Good
87
-
Depleted modes
guidcs
Retrofit sac
948
1066
950- 1050
I/C interaction
consumption
-
-
-
pile Quides
-
-
-
-
-
-
-
-
repair/
rctrofit sac
Good
Good
Good
Good
Good
Good
87
R7
87
87
87
87
910
962
93s
908
903
925
974
976
995
911
993
914
I
920-960
920-930
Av. 950
Av. 914
Av. 939
Av. 947
Refercncc
number
System
Performance
How wen
type
sntislactory.
No repairs
problems
What problems
were dctecmd?
failure"
Type
Action
taken
Summary
detectd?
P a n t levels 01protcction
Date
Potentials
negative rnillivolb
Mosr
Range90Y0
All with rpspert to Ag/AgCl
Least
or mods
34
Sac
No
35
Sac
YcslNo
36
37
38
39
Sac
Sac
Sac
Hybrid
Sac
Sac
Yes
40
41
42
Sac
Sac
Sac
43
44
47
Sac
53
SllC
Sac
No data
Yes
Yes
Yes
YCS
Yes
Ycs
54
55
SllC
Sac
Yes
Yes/No
48
sac
Sac
sac
Sac
51
S?
sac
I/C
56
51
Sac
58
-
=
=
J-R =
Ycs,"o
-
850-950
-
Good
86
750
930
750-900
-
Good
Good
Good
Good
Investigation
860
840
a90
1036
1000
940
938
916
928
Y80
1020
1066
I I50
1030
960
980
1002
1022
890-940
940-980
Good
Good
Good
Good
81
87
87
87
87
85
86
87
87
925-1000
980-1010
-
Good
Good
87
87
910
880
976
1002
923-960
900-990
-
-
-
Good
Good
Good
Good
Good
Good
87/88
87/88
87/88
87/88
950
1050
87
Good
897
87
1008
898
Good
Good
87
87
870
750On
600 OK
995
950
Av.939
87
943
I013
Av.990
-
5 Anodcs wiih langiSurvcy/monitor
tudinol cracks
2 Anodes with longiiudioal cracks
-
-
-
-
survey
Yes
SurvcyJmoniior
Survey/monitor
-
Longitudinal cracks
in anodes detected
before launch
Anodes
changed
-
Good
Pile bonded
Discontinuous/
unprotcctcd pilc
I
Poor control &
Dislribution
Some anodcs lost
-
-
Design
Improved
monitor
control,
adjust
retrofitted
Mechanical
Nota
Sac
1030
-
on piling
ric
810
I
Nu
YesiNo
87
-
YCS
Yes
Ycs
Yes
Ycs
YeslNo
YCS
YcsJNo
Good
Coat subsequent conductors
I
Yes
s;s
Sac
49
50
Design
-
Yes
46
45
Survey
Conductor mid spans
under-protected
(-607rnV)
Still polatisingl
stainless stecl
impresxd current
srcrifici;il anode
transforiner - rectifier
iii perfwnxmcc col i i i d i a i p satisfactory performance, but somc remedial work undertaken
no rcsponse to questionnaire
Good
950
950
950
Av.1050
Av.950
1050
1050
1050
1000-1040
1000-1040
1000-1040
1000-1040
I050
1000-1OdO
1004
When
Av.1000
1044-1130
lOlU
-
Av.880
On
Good
I
h,
cn
do
Gcncral evrrvsivn
Rclcrcnw
nurnhfr
-3I
4
5
6
7
x
Y
I0
II
I2
13
14
15
16
I7
in
19
20
?I
2
23
24
2.5
26
17
18
29
30
31
32
33
34
35
36
37
38
Main
Critical
luhrs
jsinls
No
So
No
So
Hiscrs Conductors l’ilcr
Pitting corrosion
J ‘lirk Caikwns Welds Ilni! Main
tubes
No
Xu
So
So
No
Slight No
No
No corrosion detected
Stighl No other corrosion tlctccrcd
Slislil No other corrosion dctcctcd
Ycs
Yes
Yes
YCS
NO
so
Nil
Ycs
So d t i ~ apwuidcd
No corrosioi, dciccted
No corvosion dctcctcd
No d u ! ~provided
Some general corrosion
No corrusivn dctcc~cd
KOcorrosion dctcctcd
No corrosion dc~cclcd
No t1;~t;i a w i l ; h l c
N o data ilrail;rhlc
No d m k~v:iihbIt
N o d m ;iv;iihbIe(1980)
No data ~~v~iilahlc
(1980)
No data availablc t 1980)
Some general corrosion hisroriciil
No eorrosioir dclcctcd
-
Somc general corrosion - historical
So cnrrosioir detcctcti
So data
No ctbrri)sion dctcctcd
No corrosion dclcctcd
No corrosion dctccicd
No eorrosioii dctcctcd
N o corrosion detected
No corrosion dcteclcd
No corrosion dctectcd
No corrosion dclectcd
No corrnsion detected - cicepr nn holts
No corrosion delectcd
N o corrosion drlectcd
No corrosion detected
-
-
No
No
No
So
So
Wo
No
No
No
80
No
Critical Rixrs Conductors Piles J Iubcs Caissons Welds Haz
joints
No
No
No
No
No
Nu
No pirring detected
Yes
Yes
Yes
90 pitting detected
S o pirring dercctcd
Yes
Yes
Ycs
So
so
-
Ycs
Yes
So
So
Su data providcd
S n pitting detccted
Sn pitling drtecrcd
Sonie
S o significunt picring
S o pitting dctccrcd
So pilling dercctcd
Sowe pitting detected
No d a r ~i i w l i b l c
No data availahle
Nu tlar;~avuilablc
No tlata aviiilable (1Y80)
Sn dara availahle (1980)
XUJIII iveilahle (IYXW)
Srimc pitting corrosion - historical
Sv pitting detecred
Somc pjtting corrosion - historical
No piltriig detected
-
“1
so
So
No
Werecracks
detected?
No
No
No
So
SYgtit No
No
Ycs
No
-
Yes
Ycs
Ycr - not cvrrosion
YCS nut corrosion
Ycr with & wiihout
-
-
No
No
Ycs
-
corrosion
Yes
-
Yes
- without corrosion
Ycs
No
- without corrosion
Yes
0
r
YCS
NO
Yes
None with corrosion
drtr
Yes
YCS
KOptiting detected
h’o pilling detccred
So piltinp derectrd
Yes
No pitting dctcctcd
Xn pitting dctcctcd
So pitring detected
So pining dctcctcd
No pitting detccted
so
No
KO
No
xo
No pitting detcct~d
h‘o
Xo
XU
No
NO
No
Sn
No
Yes
No
No
Yes - nftdatictitr only.
No corrosion
so datu
So pirringtlctccrcd
xo
No dam
No pitting detected
42
No corrosion dctcctcd
NO
43
44
45
46
No significant corIosion detsred
No pitting detected
No pitting detcctcd
Nu pitring dctcctcd
No
No
NO
39
40
No corrosion detected
41
So corrosion dctccrcd
47
48
49
50
51
52
53
54
55
56
57
SU
No sigiiilicaiit corrosion detected
No sigiiliciaiit corrosion dercctcd
No significanr corrosion dcrected
Nu &to aviilrhlc
No corrosion dctcctcd
$0 corrosion detected
No corrosion detecrcd
N o corrosion dctccrcd
Xo cnrrosion dctectcd
No corrosion deterred
No corrosion dtrectcd
So corrosion dclccrcd
Xtr corrosion detccrcd
Nu pitting dctcctcd
So piltingdctcctcd
No da!a available
S o pining detected
So pitting detected
So pitting tlctectcd
No Filling dctccted
S o pJlllngdetecred
SOpuang detected
No PillIIlg dctccted
No piiling detected
Fu piiring detected
No pitting dcrcctcd
No piiiing dctccted
- rcpaircd 1979
Snnx N o
Yes
Yes
No pitting detected
Xo
- no1 corrosioir
NO
NO
NO
NO
No
NO
NO
YO
No
No
No
Yes
None with corrosion
TABLE 23 GENERAL COMMENTS
Sacrificial system users
High anode weight provided. Some improvements possible in anode
distribution and shape. Would consider coated or part coated structure in
future. If all is good after 1-2 years, fixed monitoring not essential.
Performance good except in conductor bay area where anode distribution
insufficient. Probably use similar system in future. Fixed monitoring system
failed - not replaced because its purpose achieved in early years.
Satisfied with performance and will continue with uncoated structures and
Al-Zn-Inanodes, with emphasis OQ provisions of adequate peak anode output.
System designs could be further improved if adequate fixed monitoring by
acoustic systems were provided, but difficulties because of benefits not accruing
to project installing the monitoring systems. Allow for current drain for pile
"stick up" and inside piles.
Satisfied with performance. Would coat conductors in future. Monitoring
system may reduce surveys in future.
Satisfied with performance. Would make no changes for future. Require better
information on requirements for monitoring the cathodic protection system.
No problems. No changes for future. System could give extended life if
required. CP surveys incorporated into all ROV work at negligible cost,
detailed surveys only to investigate anomalies.
Good performance attributed to high initial current output capability and good
distribution of anodes.
Trouble free, easy to maintain. Requires fixed monitoring for: variations
through year, re-certification with reduced survey work, to improve future
designs. Excessive anode consumption suspected because of debris at base of
structure, short design life of CP system may dictate future retrofit for extended
iife field.
Excellent performance. Very conservative design, No changes for future.
Monitoring system performance good, but direct entry to computer data base
advantageous.
Good conservative design. Partial coating and aluminium anodes could give
optimum weightkost system. Extensive fixed monitoring in first 2 years gives
valuable data for future designs. Anode distribution beginning to be
determined by computer model.
.
0
.
.
.
.
.
.
Cathodic protection of offshore structures
259
-
TABLE 23 GENERAL COMMENTS continued
Impressed currenuhybrid system users
Early generation system. Design not suitable for environment. Inadequate
current control. Unsatisfactory anode distribution. Too few anodes of too high
current. Maintenance costs too high. Some sacrificial anodes poorly located.
For a new structure, would use sacrificial, but a reliable hybrid system
considered feasible with proper input using better current density data and
more complete sacrificial back up properly distributed.
Second generation system. Has worked and is working. Too complex,
particularly in control setting options. Corrosion historic because of periods
without power. A revised design could avoid switch-off during diver activity.
Good monitoring and survey data being achieved with intention of reducing
survey inspection requirements.
Early generation system heavily modified. Would be better with a fixed
monitoring system. Satisfied with present performance. If impressed current
anodes fail - retrofit sacrificial. Could balance system well with good
monitoring system and mathematical modelling. An advantage of impressed
current is that extended platform life can be accommodated.
Early/mid generation systems. Would not have same system again. Prefer
sacrificial systems. Would consider sacrificial plus low cost coating to tubulars
only - not nodes. Tf impressed use dictated, would prefer remote anodes to
structure mounted. Fixed monitoring essential with impressed current. Prefer
hard wired but with improved cabling system. Suggest modular system, making
extensiodrepair possible. Detects 50 mV potential variation winter/summer
with fixed monitoring - would not detect this by diver surveys.
Late second generation system. Good performance, but could have simpler
control system. Was powered immediately, therefore no corrosion. Have to
switch off during radio silence for well perforation. For future systems, would
consider combined sacrificial and coating for lower cost and greater simplicity.
Greater data from impressed current system is considered an advantage.
System switched off in 1 year for 5 months during survey work,
260
MTD Ltd Publication 90/102
The Marine Technology Directorate Limited (MTD Ltd) aims to
promote, develop and advance, in the national interest, research,
training and information dissemination in marine technology,
including all aspects of engineering, science and technology relating
to the exploration and exploitation of the sea.
MTD Ltd is an association of members having interests and
capabilities in marine-related technology. They include industry,
government, research establishments, academic and other learned
institutions, and the Science and Engineering Research Council
(SERC).
MTD Ltd advances marine research and development, primarily by
means of its research activities in Higher Education Institutes and
partly funded by SERC. MTD Ltd also provides an interface between
such research and the requirements and expertise of its members. In
1989, MTD Ltd absorbed UEG, the research and information group for
the offshore and underwater engineering industries, thereby
expanding its interests to include multi-sponsor projects.
For further details, contact:
The Secretary
The Marine Technology Directorate Limited
19 Buckingham Street, London WC2N 6EF
Telephone 071-321 0674
Facsimile 071-930 4323
List of illustrations
Figure 1
Figure 2
Figure 3
Figure 4.
Figure 5
Figure 6
Figure 7
Figure 8
Figure 9
Figure 10
Figure 11
Figure 12
Figure 13
Figure 14
Figure 15
Figure 16
Figure 17
Figure 18
Figure 19
Figure 20
Figure 21
Figure 22
Figure 23
Figure 24
Figure 25
Figure 26
Figure 27
Figure 28
Figure 29
Figure 30
Figure 31
Figure 32
8
Schematic representation of aqueous corrosion
Polarisation diagram schematically representing the electrochemistry of
aqueous corrosion
Polarisation diagram representing control of corrosion rate by sluggish
cathodic kinetics (in this case controlled by the rate of arrival of oxygen at
the surface) and the effect of increasing oxygen availability
Corrosion profile of steel piling after 5 years exposure
Schematic diagram showing how corrosion can be reduced or scrapped, by
applying cathodic protection
Representation of cathodic protection using zinc alloy sacrificial anodes on
a structure in sea water
Sacrificial anodes installed on space frame structure prior to launch
Representation of impressed current cathodic protection using inert anode
in sea water
Platinised titanium impressed current anode installed on structure member
prior to launch
The corrosion, cathodic protection and over-protection rkgimes expressed
as a function of electrode potential
Schematic S-Ncurve
Basic S-Ndesign curves for protected and unprotected welded tubular
steel joints
Schematic fatigue crack growth rate curve
Effect of sea water on the fatigue crack growth rate of BS 4360 grade 50D
steel
Effect of potential on the corrosion fatigue crack growth of BS 4360 grade
SOD steel in sea water
S-Ndata for planar welded joints of BS 4360 grade SOD steel in air and in
sea water without and with cathodic protection
Comparison of experimental S-N data for tubular welded joints with the
design curve for protected joints
Curves of crack depth against percentage of fatigue life for planar and
tubular welded specimens
Intact and well adherent coating
Well adherent coating with damage extending to the substrate
Damaged coating together with region of poor adhesion
Coating with region of poor adhesion resulting from application on to rusty
surface
As Figure 20, following cathodic disbonding
As Figure 21, following cathodic disbonding
Current flow through a sacrificial anode
Measurement of electrochemical potential
Simulation of one-dimensional cell
Electrode potential plotted against current density, in sea water
Polarisation curves
Ty-pical node on a tubular member
Single node on a tubular member
Resistance network to replace conducting electrolyte
MTD Ltd Publication 90/102
Figure 33
Figure 34
Figure 35
Figure 36
Figure 37
Figure 38
Figure 39
Figure 40
Figure 41
Figure 42
Figure 43
Figure 44
Figure 45
Figure 46
Figure 47
Figure 48
Figure 49
Figure 50
Figure 51
Figure 52
Figure 53
Figure 54
Figure 55
Figure 56
Figure 57
Figure 58
Figure 59
Figure 60
Figure 61
Figure 62
Figure 63
Figure 64
Figure 65
Figure 66
Offshore structure zinc reference anode
Typical stand-off and flush-mounted anodes
Typical pore size distributions in cement paste (volume intruded by
mercury under increasing pressure)
Schematic of pore system in concrete
Effective oxygen concentration profile through concrete cover, showing
relative effects of water-filled and air-filled porosity
Effect of surface on transport of oxygen through cement paste
Influence of solution pH and potential (with and without presence of
chloride ions) on corrosion of steel
Schematic of the effect of chloride on the anodic polarisation of steel in
concrete
Schematic of the effect of limited oxygen availability on the cathodic
polarisation of steel in concrete
Schematic of potentials of steel in concrete (free corrosion can only occur
at a rate corresponding to the current where the curves intersect)
Current flow through concrete in air and in sea water
Schematic of couple between steel in sea water and steel in oxygendepleted concrete
Schematic of couple between steel in sea water and passive steel in
concrete
Typical optimisation of bracelet anode system
Performance of sacrificial anode materials in hot saline mud
Bracelet anodes €or concrete-coated pipelines
Attenuation of pipe potential
Typical attachment of bracelet anode to concrete-coated pipe
Typical clamped, tapered bracelet anode
Proposed pipeline strip anode
Typical monobloc isolation joints for pipelines
Monitoring of pipeline cathodic protection system, using remotelyoperated vehicle
Monitoring of pipeline cathodic protection system, using towed instruments
Potential survey - local point contact
Hard-wired dual reference electrode, zinc and silver/silver chloride
elements
Acoustic-linked reference electrodeltransponder for acoustic-linked
monitoring system
Hard-wired monitored anodes
Approximate comparison of potentials using zinc, copperkopper sulphate,
and silver/silver chloride reference electrodes
Correction for salinity when using silver/silver chloride electrodes in sea
water
Micro-processor controlled monitoring system topside display unit
produces hard copy
Potentias survey - topside metallic contact
Configuration of piles round steel jacket leg
Sections through jacket frame
Variation of sea water resistivity as a function of salinity and temperature
Cathodic protection of offshore structures
-
9
List of tables
Table 1
Table 2
Table 3
Table 4
Table 5
Table 6
Table 7
Table 8
Table 9
Table 10
Table 11
Table 12
Table 13
Table 14
Table 15
Table 16
Table 17
Table 18
Table 19
Table 20
Table 21
Table 22
Table 23
Table 24
Table 25
Table 26
Table 27
Table 28
10
Estimated maximum corrosion rates of clean steel in North Sea water at
7OC
Estimated maximum current density required to protect clean steel in
~ o r t hSea water at 7OC
Formulae used for calculation of anode resistance
Information to be considered in performing catbodic protection design
work
Guidance on minim= design current densities for cathodic protection of
bare steel
Potential Limits for cathodic protection of steel
Principal advantages and disadvantage of sacrificial and impressed current
systems
Guide on coating breakdown criteria for cathodic protection
Typical tabulation of surface areas, current requirements and anode weight
requirements
Typical sacrificial anode alloy characteristics at ambient temperatures
(25OC)
Impressed current anode performance characteristics
North Sea structures and their installation dates
General information from 1988 survey of platforms in UK waters
Steel jackets
Concrete structures
S a d c i a l anode systems
Impressed current systems
Hybrid systems
Monitoring and surveys
Electrical continuity
System performance
Effectiveness of system
General comments
Composition and properties of typical sacrificial anode alloys at ambient
temperatures (up to 25'C)
Potential limits for cathodic protection of steel
Coating breakdown
Compositional specifications for zinc
Anode resistance Formulae
MTD Ltd Publication 901102
fatigue damage summation failure limit
area
linear dimension, distance
h e a r dimension
final coating breakdown factor
mean coating breakdown factor
drag coefficient
final current density for protection of bare steel
inertia coefficient
mean current density (A/m2) for protection of bare steel
outside diameter of pipeline
density of anode material
electrochemical potential
equilibrium anodic potential
equilibrium cathodic potential
corrosion potential
potarised electrochemical potential
Electrical conductance of pipeline coating
electric current
corrosion current
final current to be delivered by the cathodic protection system
limiting current
mean current to be delivered by the cathodic protection system
polarised corrosion current
current density at steel surface
stress intensity
fracture toughness
threshold stress intensity for stress corrosion cracking
length
anode lengeh
distance from drain point to which pipe is protected
consumption rate of anode material
number of fatigue cycles to failure
number of cycles to failure under constant amplitude cyclic loading
periphery of anode
ratio of maximum and minimum steel thicknesses
anode resistance
longitudinal electrical resistance
radius
cyclic stress range
actual anode spacing
anode surface potential
cathode surface potential
anode spacing to meet maximum current requirements
ithstress range
anode spacing to meet mean current requirements
design life (h)
Unconsumed anode thickness
Cathodic protection of offshore structures
11
utilisation factor
water particle vefocity
water particfe acceleration
volume of element
potential
potential at point P
anode material closed circuit potential
pipe potential at drain point
open circuit potential of unprotected steel
positive limit for adequate pipeline protection
tension hill potential
required net mass of anode material
mass per anode
gap between half shells of bracelet anode
arithmetic mean of anode length and width
Kthcyclic stress intensity threshold
r)
overpotential
p
resistivity
6
density of sea water
12
MTD Ltd Publication 90/102
References
1.
ASHWORTH, V.
in Hot risers - managing the corrosion problem
Oyez -IBC (London), 1979
2.
ROWLANDS,J.C.
in Corrosion (Ed L.L. Newnes - Butterworth (London), Second Edition, 1976,256
3.
LaQUE, FL.
The corrosion handbook
Wiley (New York), 1948,384
4.
HUMBLE, H.A.
Corrosion 1949 Vol. 5,292 to 302
5.
ROSS, W.R. and ANDERSON, D.B.
Comptes Rendus du Quatrigme Congres International de la Corrosion Marine et des
Salissures, Juan-les-pins, 1976, 461 to 473
6.
STOTT, J.D.
Private communication (undated) from University of Manchester Institute of Science
and Technology
7.
SHREIR, L.L. and HAYFIELD, P.C.S.
Impressed current anodes
in Cathodic protection - theory and practice (Eds V. Ashworth and C.J. Booker)
Ellis Homood Ltd (Chichester), 1986
8.
DEPARTMENT OF ENERGY
Report of the Cathodic Protection Study Group
HMSO (London), 1983,60
9.
DET NORSKE VENTAS
Cathodic protection design
Veritas Recommended Practice RPB 401, March 1986
10.
DET NORSKE VERITAS
Rules for submarine pipeline systems
Oslo, 1981, reprinted Sept. 1982
11.
NATIONAL ASSOCIATION OF CORROSION ENGINEERS
Control of corrosion of offshore steel pipelines
NACE Publication RP-06-75(Houston, Texas), 1975
12.
NATIONAL ASSOCIATION OF COFUIOSXON ENGINEERS
Corrosion control of steel fixed offshore platform associated with petroleum
production
NACE Publication RP-01-76 (Houston,Texas), 1983
Cathodic protection of offshore structures
26 1
13.
YAGUE-MURILLO, 0.
PhD Thesis
University of Manchester, 1983
14.
Design of tubular joints for offshore structures
UEG Publication UR33, 1985 (available from MTD Ltd)
15.
CROOKER, T.W. and LEIS, B.N. (Editors)
Corrosion fatigue: mechanics, metallurgy, electrochemistry and engineering
STP 801,American Society for Testing and Materials (Philadelphia, Penn.),1981
16.
ORIANI, R.A., HIRTH, J.P. and SMIALOWSKT, M. (Editors)
Hydrogen degradation of ferrous alloys
Noyes Publications (Park Ridge, New Jersey), 1985
17.
MINER, M.A.
Truns. Am. SOC.Mech. Engrs. - 3. Appl. Mech. 1975 Vol. 12 150
18.
DEPARTMENT O F ENERGY
Offshore installations: guidance on design and construction
HMSO (London), 4th Edition, 1990
19.
PARIS, P.C. and ERDOGAN, F
lYans. Am. SOC.Mech. Engrs - J. Basic Engng. 1963 Vol. 85 528
20.
MORGAN, H.G. et aE.
Proc. Int. Conf. on Steel in Marine Structures, Paris, 1981, Paper 5.1
21.
SHARP J.V. and THORPE, J.W.
Implications of new data on the fatigue performance of tubular joints
OTJ '88 Proceedings, Paper 3 (available from MTD Ltd)
22.
BOOTH, G.S
Proc. European Offshore Steels Research Seminar, Cambridge, 1978,227
23.
WYLDE, J.G., BOOTH, E.S. and IWASAKI, T.
Fatigue tests on welded tubular joints in air and sea water
Welding Institute Report 251, Cambridge, 1984
24.
WILSON, TJ. and DOVER, W.D.
Proc. Second Int. Conf. on Fatigue and Fatigue Thresholds,
Birmingham, 1984,1495
25.
KEFUZ, J, HOLMES, R. and BROWN, G.M.
Proc. Conf. on Environment-Assisted Fatigue, Sheffield, 1988, Paper 2
26.
KEFUZ, J. and HUTCHESON, D.
Proc. Second Int. Conf. on Fatigue and Fatigue Thresholds,
Birmingham, 1984,1531
262
MTD Lcd Publication 90/102
27.
DEPARTMENT OF ENERGY
UK Offshore Steels Research Project. Phase 11 - Final Summary Report
Offshore Technology Report OTH 87 265, HMSO (London), 1987
28.
BOCKRIS, J. O’M and REDDY, A.K.N.
Modem electrochemistry
Plenum Press (New York), 1970
29.
GUISEPPI-ELIE, A. el al.
Simulated underfilm corrosion of coated mild steel using an artificia1 blister
Corrosion 1983 Vol. 39 108
30.
LEIDHEISER, H. (Junior), WANG, W. and IGETOFT, L.
The mechanism for cathodic defamination of organic coating from a metal surface
Progress in Organic Coating 1983 Vol. 11 19
31.
SCHWENK, W.
Adhesion loss of organic coating, causes and consequences for corrosion protection
in Corrosion control by organic coating (Ed. H. Leidheiser, Junior)
National Association of Corrosion Engineers (Houston, Texas), 1981
32.
PROCTOR, R.P.M.
Detrimental effects of cathodic protection: embrittlement and cracking phenomena
in Cathodic protection - theory and practice (Eds. V. Ashworth and CJ. Booker)
Ellis Horwood Ltd (Chichester), 1986
33.
AMERICAN SOCIETY FOR TESTING AND MATERIALS
Standard methods for cathodic disbonding of pipefine coating
ASTM publication G8-85, New York, 1985
34.
BRITISH GAS CORPORATION
Evaluation of resistance to cathodic disbonding
PS/CW6: Part 1, Appendix A, 23
35.
BRITISH STANDARDS INSTITUTION
Methods of test for paints
BS 3900
Part F11: 1985
Determination of resistance to cathodic disbonding of coatings for use on land-based
buried structures
36.
BRITISH STANDARDS INSTlTUTION
Methods oftest for paints
BS 3900
Part FlO: 1985
Determination of resistance to cathodic disbonding of coatings for use in marine
environments
Cathodic protection of offshore structures
263
37.
BRITISH STANDARDS INSTITUTION
Specification for performance tests for protective schemes used in the protection of
light-gauge steel and wrought iron against corrosion
BS1391: 1952 (now withdrawn)
38.
JXN, X.H. et al.
The adhesion and disbonding of chlorinated rubber on mild steel
Proc. Symp. on Corrosion Protection by Organic Coatings
EIectrochemical Society Publication 87-2, 1987
39.
HEIM, G, BAECKMANN, W.V. and FUNK, D
Investigating subsurface creeping of covering of steel pipe subject to cathodic
polarisation
3R International 1977 Vol. 6 319
40.
TROTMAN, D.W.
Cathodic protection - overprotection and coating damage on ships and marine
structure
in Cathodic protection - theory and practice (Eds V. Ashworth and CJ. Booker)
Ellis Horwood (Chichester), 1986
41.
WESTERN EUROPEAN COMMITTEE ON CORROSTON AND PROTECTXON
O F CONDUITS
Cathodic disbonding of steel pipeline coatings
CEOCOR Publication CR2 "CD", final draft, December 1987
42.
ROTHWELL, G.P., FRANCIS, P.E. and HALE, K.F.
The effects of heat transfer on the external corrosion of submarine pipelines and risers
Summary Report of Project 340, Society for Underwater Technology (London), 1982
43.
HIGGXNS, G.L, CABLE, J. and PARSONS, L.
Aspects of cathodic disbondment testing at elevated temperature
Industrial Corrosion 1987 Vol. 5 (NoJ), 2
44.
HTGGlNS, G.L., CABLE, R.A. and BATES, C.R.
Investigations into the phenomenon of cathodic disbondment at elevated temperature
Proc. 7th Int. Conf. on Internal and External Protection of Pipes, London, Sept 1987
BHRA (Cranfield), 1987
45.
COULSON, K.E.W. and TEMPLE, D.G.
Impact, cathodic disbondment line-coating test results vary with surface coating
conditions
Oil and Gas Journal 10 March 1986,47
46.
DICKIE, R.A.
Chemical studies of the organic coating-steel interface after exposure to aggressive
environments
in Polymeric materials for corrison control (Eds. Dickie, R.A. and Floyd, F.L)
American Chemical Society (Washington, DC), 1986
264
MTD Ltd Publication 90/102
47.
MORGAN, J.H.
Cathodic protection
National Association of Corrosion Engineers (Houston, Texas), 1989
48.
Surface preparation standards for painting steel structures
Swedish Standard SIS-05-59-00,
1967
49.
NORMAN, D.
Modern coatings for pipe fitting from mill to trench
Industrial Corrosion 1988 VoI. 6 NO.^), 4
50.
SMYRL,W.H.
Current and potential distributions in corrosion systems
Am. Inst. Chem. Engrs. Symposium Series 1981 VoI, 77 152 to 160
51.
NISANCIOGLU, K.
Materials Performance 1984 Voi. 23 36 to 43
52.
GRIGOR’EV, V.P.
Magnitude of the cathodic protection current as a function of the conditions of
carbonate scale formation
J. Appl. Chem. (USSR) 1961 Val. 34 169
53.
GRIGOR’EV, V.P. and POPOV, S.A
Protective properties of carbonate scale
J. Appl. Chem. (USSR) 1962 Vol. 35 1555
54.
STROMMEN, R. and RODLAND, A
Computerised numerical techniques applied in design of offshore cathodic protection
systems
Materials Performance 1981 Val. 20 15 to 20
55.
DeCARLO, E.A.
Materials Performance 1983 Vol. 22 38 to 44
56.
STROMMEN, R. et. ai.
Materials Performance 1987 VoI. 26 23 to 28
57.
MITCHELL, A.R. and GRIFFlTHS, D.F.
The finite difference method in partial differential equations
WiIey (New York), 1980
58.
N O W E , D.R.J. and HINTON, E.
An introduction to finite element analysis
Academic Press (New York), 1980
59.
BREBBIA, C.A. and WALKER, S.
Boundary eIement techniques
Newnes (London), 1980
Cathodic protection of offshore structures
265
60.
TIGHE-FORD, D J. et at.
The use of scale modelling for the evaluation of impressed current cathodic protection
systems for marine structures
Proc. Corrosion '86 Conference
Institute of Corrosion (Birmingham), 1986,163 to 165
61.
US ARMY CORPS OF ENGINEERS
Military specification MIL-A-18001J,1989
62.
AMERICAN SOCIETY FOR TESTING AND MATERIALS
Specification ASTM B418-80,New York, 1980
63.
THOMASON, W.H.
Cathodic protection of submerged steel with thermal-sprayed aluminium coatings
Materials Performance 1985 Vol. 24
64.
LEVINGS, E.A., et al.
The Murchison cathodic protection system
Proc. Offshore Technology Conference (Houston,Texas), 1983,Paper 4565
65.
VARDON, J.O.C., HARVEY, D.W. and COOPER, M.T.
Hutton impressed current cathodic protection system - reliability by design
Proc. Corrosion 85 Conf. (Boston,Mass.), March 1985,Paper 315
66.
KUNJAPUR, M., HART", W.and SMITH, S.S.
Influence of temperature on calcareous deposit deposition upon cathodically protected
steel in sea water
Roc. Corrosion 85 Conf. (Boston,Mass.), March 1985,Paper 316
67.
Corrosion control of offshore pipelines I1
Final Report, NTNF Project No. 1830.5585, March 1982
68.
BRITISH STANDARDS INSTITUTION
Specification for zinc anodes, zinc oxide and zinc salts for electroplating
BS 2656: 1972
69.
BRITISH STANDARDS INSTITUTION
Specification for hot dip galvanised coatings on iron and steel articles
BS 729: 1971
70.
BRITISH STANDARDS INSTITUTION
Specification for electroplated coatings of cadmium and zinc on iron and steel
BS 1706: 1980
71.
NATIONAL, ASSOCIATION OF CORROSION ENGINEERS
Metaliurgical and inspection requirements for cast sacrificial anodes for offshore
applications
NACE Publication RP-03-87 (Houston, Texas), 1987
266
MTD Ltd Publication 90/102
72.
BRITISH STANDARDS INSTITUTION
Code of practice for cathodic protection
CP 1021:1973
73.
WAFWE, M.A.
Application of numerical analysis techniques
Proc. Conf. on Cathodic Protection, Theory and Practice - the Present Status,
Eds. V. Ashworth and C J . Booker, Coventry, 1982, Paper 3
Ellis Horwood (Chichester), 1986
74.
BRITISH STANDARDS INSTITUTION
Quality systems
BS 5750: Various parts published between 1981 and 1987
75.
BRITISH STANDARDS INSTITUTION
Specification for classification of degrees of protection provided by enclosures
BS 5490:1977 (confirmed 1985)
76.
TUUTT'I, K
CBL 477 Swedish cement and Concrete Research Institute, 1977
77.
PAGE, C.L., SHORT, N.R. and TARRAS, A. ER.
Cement and Concrete Research 1981 Vol. 11 395
78.
TAYWOOD ENGINEERING
Effectiveness of concrete to protect steel reinforcement from corrosion in marine
structures
Offshore Technology Report OTH 87 247, HMSO (London) 1987
79.
GJORV, 0.E.
Nordisk Betong 1972 Vol. 1 19
80.
GJORV, O.E., VENNESLAND, 0. and BUSAIDY, A.H.S. El
Proc. Corrosion 76 Conference (Houston,Texas), March 1976, Paper 17
(also in Materials Performance Dec 1986 VoI. 25 39)
81.
FIDJESTOL, P., RONNING, B. and ROLAND, B.T.
Proc. Corrosion 85 Conference (Boston,Mass.). March 1985, Paper 262
82.
POURBAIX, M.
Corrosion Science Jan. 1974 Voi. 14 25
83.
HANSSEN, C.M.
Cement and Concrete Research April 1984 Vol. 14 574
84.
YONEZAWA, T., ASHWORTH, V. and PROCTER, R.P.M.
Proc. ASTM Meeting on Pore Water Expression for Determination of ReinforcingBar
Corrosion Conditions, Dec. 1985
Cathodic protection of offshore structures
267
85.
WHEAT, H.G. and ELIEZER, 2.
Corrosion Feb. 1987 Vol. 43 126
86.
FIDJESTOL, P. and NILSON, N.
Field test of reinforcement corrosion in concrete performance of concrete in marine
environments
American Concrete Institute (Detroit) Publication SP-65,1980
87.
ARUP, H.
Proc. Corrosion 79 Conference (Atlanta Georgia), March 1979
88.
WILKLNS, NJ.M. and LAWRENCE, P.F.
Fundamental mechanisms of corrosion of steel reinforcements in concrete immersed
in sea water
Concrete in the Oceans Report TR7, British Cement Association (Wexham Springs),
Publication ref. 15.616, 1980
89.
WILKINS, N J.M. and LAWRENCE, P.F.
Proc. Conf. on Concrete in the Marine Environment, London, Sept. 1986
90.
KING, R.A., NABIZADEH, H. and ROSS, J.K.
Corrosion Prevention and Control April 1977 Val. 11
91.
HILDEBRAND, H., SCHULZE, M. and SCHWENK, W.
Werhtoffe und Korrosion 1983 Vol. 34 281
92.
GJORV, O.E. and VENNESLAND, 0.
Proc. Corrosion 79 Conference, (Atlanta, Georgia), March 1979, Paper 139
93.
SHAW, J.A.
Civil Engineering (ASCE, New York) June 1965 Vol. 39
94.
DET NORSKE VERITAS
Rules for the design, construction and inspection of offshore structures
DnV 1977, reprinted Oct. 1981
95.
GERMANISCHER LLOYD
Rules for the construction and inspection of offshore structures
Vol. 1, Section 8 Corrosion protection (English translation)
Supplement 1 of 1985 reprint
96.
ESPELID, B., FIDJESTOL, P. and NILSEN, N.
Durability and corrosion behaviour of dynamically-loaded offshore concrete
structu res
Proc. Conf. on Concrete in the Marine Environment, London, 1986
97.
NORWEGtAN PETROLEUM DIRECTORATE
Guidelines for corrosion protection of offshore structures
Draft English translation, NPD (Stavanger), 1987
268
MTD Ltd Publication 90/102
98.
BROWNE, R.D.
Low maintenance concrete - specification versus practice
in Marine and Offshore Structures Maintenance, Thomas Telford Ltd
(London), Second Edition, 1986
99.
COLLARD, MJ.
Inspection and maintenance of North Sea concrete platforms
in Marine and Offshore Structures Maintenance, Thomas Telford Ltd (London), First
Edition, 1977
100.
NORWEGIAN PETROLEUM DlRECTORATE
Regulations for the structural design of fixed structures on the Norwegian
Continental Shelf
NPD (Stavanger), 1977
101. BRITISH STANDARDS INSTITUTION
Code of practice for fixed offshore structures
BS 6235:1982 (now withdrawn)
102. LLOYD'S REGISTER OF SHIPPING
Draft rules and regulations for the classification of fixed offshore installations
June 1987
103. DET NORSKE VERITAS
Monitoring of cathodic protection systems
Veritas Recommended Practice RPB403,March 1987
104. PETERSON, M.H. et a1
Materials Protection and Performance, May 1972,Vol.11 (No. 51, 19 to 22
105. MOULTON, R J .
Hazards to divers from cathodic protection systems
in Proc. Conf. on Cathodic Protection, Theory and Practice - The Present Status,
Eds. V. Ashworth and C J Booker, Coventry, April 1982,Paper 21
Ellis Horwood (Chichester), 1986
106. WARNER, S.A.
Shock hazard from impressed current anodes
Diving Safety Memorandum No. 2, Dept of Energy (London), 1985
107. ASSOCIATION OF OFFSHORE D M N G CONTWCTORS
Code of practice for the safe use of electricity under water
London, 1985
108. WYATT, B.S.
Proc. Conf on Asian Inspection, Repair and Maintenance, Singapore,
February 1985,Paper 19
Cathodic protection of offshore structures
269
109. THOMPSON, G.R.
Beryl B foundation instrumentation
Presented at Meeting of Society for Underwater Technology (London),
12 January 1984
110. MOLLAN, R. and ANDERSEN, T.R.
Design of cathodic protection systems (revised DnV guidance)
Proc. Corrosion 86 Conf, 1986,Paper 286
111. BRAATHEN, J.
NPD’s activities in connection with corrosion protection of offshore installations and
pipelines
Presented at 7th Int. Conf. on Internal and External Protection of Pipes, (London),
Sept. 1987
Published by BHRA (Cranfield), 1987
112. BARRE’IT, B.B., HOWELLS, R. and HINDLEY, B.
Safety in the offshore petroleum industry
Kogan Page (London), 1987
113. DANISH SOCIETY OF ENGINEERS
Korrosions beskyttelse af Stalkonstruktioner i Marine omgivelser
Danish Standards Recommendation DS/R 464,1988
114. WUNGAARD, B.H.
Recommended practice for the cathodic protection of offshore steel structures
(including pipelines)
Stichting Materiaalonderzoek in de Zee (Delft), 1981
115. DEPARTMENT OF ENERGY
Submarine pipeline guidance notes
London, Dec. 1989
116. LLOYD’S REGISTER O F SHIPPING
Rules and regulations for the classification of mobiie offshore units
Part 4, Chapter 2
Draft, January 1986
117. NORWEGIAN PETROLEUM DIRECTORATE
Guidance for the inspection of primary and secondary structures of production and
shipment installations and underwater pipeline systems
Oslo, 1982
118. INSTITUTE OF PETROLEUM
Model code of safe practice
Part 4: Pipeline safety code
London, 4th Edition, 1982,with supplement, August 1986
270
MTD Ltd Publication 90/102
Appendix Essential design information
Cathodic protection of offshore structures
27 1
TABLE 11 lMpRESSED CURRENT ANODE PERFORMANCE CHARACTERISTICS
Typical design
operating voltage
material
0.008
0.800
Pt-Ti
Pt-Ni
Pb-Ag-Sb
Pb-Pt
500
Si-Fe-Cr
500
1
I
Voltage limit governed by statutoryhfety constraints
AlIoy
Environment
Capacity
(fwkg)
N-Zn-Hg
N-Zn-In
N-Zn-In
N-Zn-Sn
Zn (US.
Mil Spec)
Mg (High
Purity)
Mg (High
Potential)
Closed circuit
Potential wrt
AglAgCl
(Negative Volts)
-
Consumption
(kglA-Year)
Sea water
Sea water
Marine
sediments
Sea water
Sea water
2600-2850
2300-2650
1300-2300
760-780
1.0
0.95
Sea water
1230
1.5
7.1
Sea water
1230
1.7
7.1
925-2600
1.05
3.1
3.3
0.95 - 1.05
3.8
-
1.05
3.4
1.03
11.2
1.0
2.0 - 1.10
-
3.4
- 3.8
- 6.7
- 9.5
- 11.5
Note: Values listed are nominal for generic types.
For design purposes, values should be confirmed by long-term testing,
preferably in the field or alternatively in the laboratory.
Protective potential
Environment
.272
Positive limit
Negative limit
Aerated sea water
- 800mV wrt AglAgCl
-1050mV wrt AglAgC1
+ 0-OOmVwrt Zinc
Anaerobic
conditions
- 900mV wrt AglAgC1
-105OmV wrt AglAgCl
+ O.OOmV wrt Zinc
+ 250mV wrt Zinc
+ 150mV wrt Zinc
MTD Ltd Publication 90/102
TABLE 26 COATING BREAKDOWN
(a)
AS a function of exposure time(")
Coating breakdown (% of Area)
Initial
Mean
10
2
7
10
20
2
15
30
30
2
25
60
40
2
40
90
Find
(b) AS a function of coating type Expected lifetime 25 years(10.1 13)
% Breakdown
Type
Initial
Mean
Final
Thick film pipeline coating
1
10
20
Vinyl systems
2
20
50
Epoxy coal-ta 0.3 mm
2
20
50
Epoxy high-build
2
20
50
Epoxy powder 0.5 mm
1
10
20
Rubber
1
5
10
0.5
5
10
1
10
20
Polyethylene 2-3 mm
Reinforced bitumen on tar
Cathodic protection of offshore structures
273
TABLE 27 COMPOSlTION SPECEXATIONS FOR ZINC
(a) American Society for Testing Materialdm)
Zinc Type 2Aluminium
Cadmium
Iron
0.005%
0.003%
0.0014%
max
max
max
(b) United States Military Specification(61)
274
Copper
Aluminium
Iron
Cadmium
Lead
Others
Zinc
0.005%
0.10 - 0.50%
max
0.005%
max
Capacity
780 A h k g min.
-
0.025 0.07%
0.006%
max
0.10%
rnax total
Remainder
MTD Ltd Publication 90/102
TABLE 28 ANODE RESISTANCE FORMULAE
Anode type
Formula
Slender anodes at least 30cm
from platform member
(Modified Dwight)
R, = p/2x4ln(4Ur) - 11
= resistivity
L = length of anode
r = radius of anode ( r = P/2
p
for other shapes where
P = periphery)
Slender anodes at least 30cm
from platform member with
L<4r
R,=
Flat plate anodes close to the
structure (LlQyds)
R,= pJ2Z
Z = Arithmetic mean of anode
length and width
Other shapes and bracelet
anodes
(McCoy)
Cathodic protection of offshore structures
$= 0.31Splfi
A
= exposed surface area of anode
275
Pote ntia 1
The protective potentiats for steel in sea water of 25ohm cm
resistivity a t 2OoC with respect t o the commonly used
reference cells are :
- 0-60 volts Ag lAgC1 Ref.
- 0 - 8 5 v o l t s CulCuS0,Ref.
-0.79voLts SCE Ref.
+0-24volts Zn Ref.
This is shown in the Nomogram, which can be used to express
potentials referred to one standard with respect to either of
t w o other standard half cells. As
Resistivity'ohm
cm) this relationship varies with water
10000
resistivity, the Nornoaram is used bv
constructkg a line originating at (be
water resistivity applicable through
the potential referred t o the half
ceII being used. The equivalent
2000
potentials with respect to the other
1000
half cells are then a t the intersection
of that line and the respective scale.
500
An example is shown for
250hm cm at -0-8voLtsAgIAgCL Ref.
~
200
100
Figure 61 Correction for salinity when using silverlsilver chloride electrodes in sea water
276
MTD Ltd Publication 90/102
IlOf2cm) 10;
~ l l - l l ~ c m9;)
(12-5LkcmI 8:
( 1 4 - 2 ncm) 7-,
I 1 6 - 6 7 f i c m I 6:
(20JLcml
5-.
f33ncm)
3:
167acml
4,
200acml 5 ,
250JLtm) 4 .
SaIinity ( g solids/ kg sea water 1
Note: Values above BO'F
Figure 66
26.7OC I obtained by extrapolation
Variation of sea water resistivity as a function of salinity and temperature
Cathodic protection of offshore structures
277