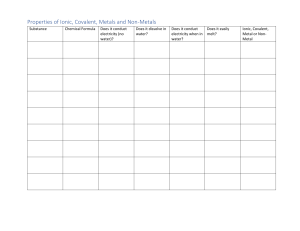
Mrs. Cresci Chemistry A 3.7 Lab: Ionic and Covalent Bonds Note: You do not need to perform the experiments using physical materials. The Virtual Lab link will provide an opportunity for you to engage with the materials virtually. Hypothesis Write 2 hypotheses that answers this lab question. Use the format "if . . . then . . . because . . .” Lab Question: Based on a substance’s properties, how can you determine whether its bonds are ionic or covalent? 1. If a substance contains ionic bonds, then its properties would include… because… 2. If a substance contains covalent bonds, then its properties would include… because… Prelab Complete the table below comparing Ionic and Covalent bonds. Ionic Bond Covalent Bond Definition Draw an Example What kind of elements are bonded? Melting/boiling points Most common phase at room temperature Soluble in… Good Conductor? Data Record your data in the data table below. Oil State of Matter Appearance and Texture Crystalline Structure (yes or no) Solubility in Water Conductivity (yes or no) Ionic or Covalent Bonds? Cornstarch Sodium Chloride Sodium Bicarbonate Reflection Refer to the data you collected in the data section. Complete the table below to summarize and interpret your findings. Ionic or Covalent? Oil Cornstarch Sodium Chloride Sodium Bicarbonate