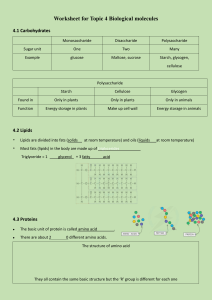
Food Analysis Lab Objectives To distinguish between positive and negative results of chemical tests for the presence of sugar, starch, protein and fat. To use indicators to determine the nutrient content of foods To predict chemical properties of foods by the presence of particular nutrients To analyze nutritional consumption data Introduction Organic molecules are those primarily made up of carbon, hydrogen and oxygen. The common organic compounds of living organisms are carbohydrates, proteins, lipids, and nucleic acids. Each of these macromolecules (polymers) are made of smaller subunits (monomers). The bonds between these subunits are formed by dehydration synthesis. This process requires energy; a molecule of water is removed (dehydration) and a covalent bond is formed between the subunits (Fig.1). Breaking this bond is called hydrolysis; it requires the addition of a water. Each class of these macromolecules has different structures and properties. For example, lipids (composed of fatty acids) have many C-H bonds and relatively little oxygen, while proteins (composed of amino acids) have amino groups (-NH3+) and carboxyl (-COOH) groups. These characteristic subunits and chemical groups impart different properties to the macromolecules. For example, monosaccharides such as glucose are polar and soluble in water, whereas lipids are non-polar and insoluble in water. Figure 1. Dehydration synthesis and hydrolysis of a polymer. Testing for Carbohydrates: Simple Sugars Carbohydrates are molecules made up of carbon (C), hydrogen (H), and oxygen (O) in a ratio of 1:2:1 (for example, the chemical formula for glucose is C6H12O6). Carbohydrates may be monosaccharides, or simple sugars, such as glucose or fructose; they may be disaccharides, paired monosaccharides, such as sucrose (a disaccharide of glucose linked to fructose, Fig. 2); or they may be polysaccharides, where three or more monosaccharides form chains, such as starch, glycogen, or cellulose Many monosaccharides such as glucose and fructose are reducing sugars, meaning that they possess free aldehyde (-CHO) or ketone (-C=0) groups that reduce weak oxidizing agents such as the copper in Benedict's reagent. Benedict's reagent contains cupric (copper) ion complexes with citrate in alkaline solution. Benedict's test identifies reducing sugars based on their ability to reduce the cupric (Cu2+) ions to cuprous oxide at basic (high) pH. Cuprous oxide is green to reddish orange. In this test, a green solution indicates a small amount of reducing sugars, and reddish orange indicates an abundance of reducing sugars. Non-reducing sugars, such as sucrose produce no change in color (the solution remains blue). Starches Staining by iodine (iodine-potassium iodide, I2KI) distinguishes starch from monosaccharides, disaccharides, and other polysaccharides. The basis for this test is that starch is a coiled polymer of glucose; iodine interacts with these coiled molecules and becomes bluish black. Iodine does not react with carbohydrates that are not coiled and remains yellowish brown. Therefore, a bluish-black color is a positive test for starch, and a yellowish-brown color (i.e., no color change) is a negative test for starch. Notably, glycogen, a common polysaccharide in animals, has a slightly different structure than does starch and produces only an intermediate color reaction. Testing for Proteins: Proteins are remarkably versatile structural molecules found in all life-forms. Proteins are composed of amino acids, each of which has an amino group (NH3+) and a carboxyl group (-COOH). The amino group on one amino acid is linked to the carboxyl group on an adjacent amino acid by a peptide bond. The peptide bond forms through dehydration synthesis. This bond is the site of action for the Biuret test for protein. Biuret reagent is a 1% solution of CuS04 (copper sulfate). In this test, Cu2+ complexes with the peptide bond producing a violet color. The violet color is a positive test for the presence of protein; the intensity of color relates to the number of peptide bonds that react. A Cu2+ must complex with at least four to six peptide bonds to produce a color; therefore, free amino acids and very short chains do not react positively. However, free amino acids and very short chains may result in a pinkish color. Long-chain polypeptides (proteins) have many peptide bonds and produce a positive reaction. Testing for Lipids: Lipids include a variety of molecules that dissolve in solvents such as ether and acetone, but not in polar solvents such as water. The primary component of fat or lipid is the fatty acid. When it occurs alone it is called a free fatty acid. This molecule is composed of an even number of carbon atoms terminated in a carboxyl group (-COOH). Fatty acids are most often stored and transported as triglycerides, three fatty acids bonded to a glycerol molecule. This bond is called an ester linkage and resulted from a dehydration synthesis. Tests for lipids are based on a lipid's ability to selectively absorb pigments in fat-soluble dyes such as Sudan IV. Procedure: Part A – Testing for Biological Molecules Watch the following video and complete the data tables using the information in the video: Food Tests - Iodine, Biuret, Benedict's, Ethanol, DCPIP: https://www.youtube.com/watch?v=sLP8dcnWnJg&feature=youtu.be Iodine Test a. b. c. d. e. Using a transfer pipet place 1mL of each test solution into each of the labeled test tube. Add 5 drops of iodine to each test tube. Swirl the solution in the test tube to mix it and note the final color in your data table. Clean out your test tubes for the next use. Record your data in the data table. Table 1. Results of the Iodine Test Material Tested Color at Beginning 1 Fruit Juice 2 Egg White 3 Starch solution 4 Oil 5 Distilled water Color at End (+) or (-) Biuret’s Test a. Using a transfer pipet place 1mL of each test solution into each of the labeled test tube. b. Add ½ mL of Biuret’s solution to each test tube. c. Swirl the solution in the test tube to mix it and note the final color in your data table d. Clean out your test tubes for the next use. e. Record your data in the data table. Table 2. Results of the Biuret’s Test Material Tested Color at Beginning 1 Fruit Juice 2 Egg White 3 Starch solution 4 Oil 5 Distilled water Color at End (+) or (-) Benedict’s Test a. Place 1mL of each test solution into each of the labeled test tube. b. Add 1mL of Benedicts Solution to each test tube and mix well c. Using the Test Tube Tongs, place the tube in the hot water bath for 3-5 minutes. d. Carefully remove the test tubes from the hot water bath and note the final color in your data table. e. Clean the test tubes and use alcohol to remove sharpie labels. f. Record your data in the data table. Table 3. Results of the Benedicts Test Material Tested Color at Beginning 1 Fruit Juice 2 Egg White 3 Starch solution 4 Oil 5 Distilled water Color at End (+) or (-) Ethanol Emulsion Test a. Place 2mL of each test solution into each of the labeled test tube. b. Add 2mL of Ethanol Solution to each test tube and shake vigorously. c. Record your data in the data table. d. Clean out your test tubes for the next use. e. Record your data in the data table. Table 4. Results of the Ethanol Emulsion Test Material Tested Color at Beginning 1 Fruit Juice 2 Egg White 3 Starch solution 4 Oil 5 Distilled water DCPIP Test a. b. c. d. Color at End (+) or (-) Add 1mL of DCPIP Solution to each test tube Place 1mL of each test solution into each of the labeled test tube and mix well. Clean the test tubes and use alcohol to remove sharpie labels. Record your data in the data table. Table 5. Results of the DCPIP Test 1 2 3 4 5 Material Tested Fruit Juice Egg White Starch solution Oil Distilled water Color at Beginning Color at End (+) or (-) Analysis/Discussion Questions for Part A: 1. What is an indicator? 2. What does each of the tests test for? a. Iodine – b. Biuret’s – c. Benedicts – d. Ethanol Emulsion – e. DCPIP – 3. Since both glucose and starch are types of carbohydrate, why could you get different reactions for the Iodine and Benedict’s test? 4. Several food samples were tested with each of the indicators. Based on the results of the tests, which macromolecules are contained in each sample? Food Biuret Benedicts Ethanol DCPIP - - - - - - + - + Nuts + - - + - Salmon - + - + - Milk - + + + - + - + + - - - - + - Potatoes Orange Juice PB&J sandwich Cheese Iodine + Macromolecules? Procedure: Part B – Analyzing Nutritional Data 1. Record everything you eat and drink for 1 day 2. Use the food you consumed to complete the data table below for that one day. The following website can help you determine the amount of grams for each of the food items you consumed. (There are several other websites available as well. In addition, many restaurants and manufacturers have nutritional information online.) Approximate amounts are fine if there are food items you are unsure of. http://www.calorieking.com/foods/ 3. How to calculate calorie breakdown for the day : 1 gram of protein has 4 calories, 1 gram of carbohydrates has 4 calories, and 1 gram of fat has 9 calories. Another quick tip, 1 gram of fiber is equal to 1 gram of carbs, but it has no calories. So foods that are high in fiber will be less in calories than you would expect. One more thing, it's ok if your calorie breakdown percentages aren't perfect, you're just trying to get a sense of your macronutrient balance. Table 5. Food Consumed in one day. Meal Breakfast Lunch Dinner Snacks Total (g) Total Calories: (calculate your total calories for each category) Total Calories for the Day Amount of Water consumed Protein (g) Carbohydrates (g) Fat(g) Analysis/Discussion Questions for Part B: 1. Use the link below to calculate your daily calorie needs to maintain your current weight. List the daily calories you would need to consume to maintain your current weight https://www.acefitness.org/resources/everyone/tools-calculators/daily-caloric-needs-estimatecalculator/?irclickid=1CAwfjUo9xyPTVyVnfyq3zatUkHx5nyl0zL93U0&irgwc=1&utm_source=Affiliate&ut m_campaign=12960&clickid=1CAwfjUo9xyPTVyVnfyq3zatUkHx5nyl0zL93U0&utm_content=Online%20T racking%20Link_984595&utm_medium=Impact&utm_channel=Affiliate_Marketing 2. Were you consuming the correct amount of calories needed to maintain your current weight based on the food you were eating for the day you revorded? Were you surprised by these data/calculations? Explain. 3. You may have heard the advice, "Drink eight 8-ounce glasses of water a day." That's about 1.9 liters. This is known as the 8X8 Rule. This rule remains popular because it's easy to remember, but it is more of a general guide. Calculate how many ounces of water or fluids you should be drinking a day by using the following formula: Your Weight X 2/3, and then adjust for your activity level, add 12 oz. for every 30 minutes you work-out. 4. Were you consuming the correct amount of fluids needed per day? Keep in mind all beverages you consume contain water so those should be used in calculating your fluid/water needs. There is water in many foods we consume as well, but it is difficult to quantify the amount of water in our foods. Were you surprised by these data/calculations? Explain. 5. Explain the importance of drinking water and how it aids in the chemical digestion of food. (Hint: See Figure 1 at the beginning of this assignment)