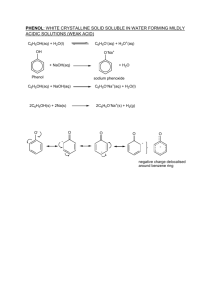
a) ethanoyl chloride b) bromine (1,2) c) nitric acid 1 Phenols Phenol, C6H5OH CH 3 + CH 3 3[O] + Br2 33 1 50 1 6 93.5 Br OH Phenol is an group aromatic is alcohol. It contains an attached −OH groupdirectly bonded directly to a A phenol an —OH group to a benzene ring. + HNO carbon atom in a benzene ring (Figure 10.33). OH NO 2 3 24.3 Phenol (1,3 Thelone C—O bond in phenol is veryonstrong, asatom a result of with the the delocalisation of the oxygen interacts The pair of electrons in the pz -orbital delocalised π-electrons in the ring (Figure 10.34). This increases the electron density + HNO NO the lone pair of electrons on oxygen over the arene ring. There are no inside the ring, making it easier for phenol to be attacked by an electrophile. This 6 5 CO H CO H explains the relative ease with which phenol reactsthe in situation electrophilic with substitution reactions in which it breaks, unlike the alcohols. reactions compared with benzene. It also decreases the δ− charge on the oxygen + HNO NO atom, making it less reactive as an alcohol. NO 2 NO2 3 2 Phenol, C H OH, is a crystalline solid wh 43 °C. It is used to manufacture a wide ran However–OH group in phenol undergoes a variety of reactions. (Figure 24.5). It’s structure is: 2 2 3 19 80 2 The data in Table 25.5 can be interpreted as follow ● H O Figure 10.34 Interaction of the oxygen electrons with H O Figure 25.12 Delocalisation of the lone pair the inaromatic ring 2,4-directing substituents ● The orientation of the incoming group (NO2 or B already in the ring, and not on the electrophile. Some substituents favour both 2- and 4-substituti favour 3-substitution, at the expense of both 2- a OH If we look closely at the types of substituents tha either they are capable of donating electrons to t they have a lone pair of electrons on the atom jo can be incorporated into the π system by sideway Figure 25.12). On the other hand, all those substituents that fav 2 formed, Phenol With Sodium The salt sodium phenoxide, is soluble in water. Phenol alsowith reacts sodium metal, Phenol reacts sodiumvigorously metal, formingwith a phenoxide and giving off giving hydrogen gas. off hydrogen gas and again forming sodium phenoxide: 2 OH + 2Na ⎯→ 2 – + O Na + H2 Substitution into the benzene ring of phenol xide. nol 3 The derivatives of phenol are named in a similar fashion to those of benzene, Nomenclature by numbering the carbon atoms in the benzene ring starting from the −OH group (Figure 18.1.33). OH OH O2N OH 1 6 2 5 4 NO2 3 CH3 OH 1 2 6 3 4 5 NO2 NO2 phenol 2,4,6-trinitrophenol Test yourself Br 2-methyl-5-nitrophenol (not 6-methyl-3-nitrophenol) 4-bromophenol 4 Reaction With Sodium Phenol reacts with sodium in a similar way to ethanol. Bubbles of hydrogen form when a small cube of sodium is added to molten phenol or to a solution of phenol in an inert solvent. The main product of the reaction is sodium phenoxide. 5 Ring Reactions With Phenols At first sight we might expect the –OH group to be electron-withdrawing due to the high electronegativity of O. ring more negative, i.e. mor the reaction occurs more quic However, the –OH group also possesses a lone pair of electrons and overlap of this lone pair into the ring activates the benzene ring. At first sight we might expect withdrawing due to the high e group also possesses a lone pai pair into the ring activates This π donation into the rin effect (due to the electronegat donation of electron densit attract electrophiles more s NO 2 6 Ring Reactions With Phenols NO2 + HNO 3 CO H This π donation into the ring is a bigger effect than the electron withdrawing effect (due to the electronegativity of O). 2 CO 2 H + HNO 3 Therefore there is net donation of electron density into the ring and the ring will attract electrophiles more strongly. The data in Table 25.5 can be ● H O ● The orientation of the incom already in the ring, and not Some substituents favour bo favour 3-substitution, at the If we look closely at the type either they are capable of do they have a lone pair of elect can be incorporated into the 7 Phenols With Nitric Acid Phenols are more susceptible to electrophilic attack than benzene, owing to the delocalisation of the lone pair of electrons on oxygen. This allows phenol to react with reagents that are more dilute, and also to undergo multiple substitution with ease. of concentrated nitric and sulfuric acids to reflux with at about °C forAcid nitration to take place (see 8 benzene Phenols With55Nitric page 385). However, the activated ring in phenol When treated with dilute aqueous nitric acid (no sulfuric acid is needed) readily undergoes nitration with dilute nitric acid at phenol gives a mixture of 2- and 4-nitrophenols. room temperature: OH OH dil. HNO3 OH NO2 + NO2 If we use concentrated nitric acid we get 2,4,6-trinitrophenol formed, shown below: of concentrated nitric and sulfuric acids to reflux with benzene atWith about 55 °CAcid for nitration to take place (see 9 Phenols Nitric page 385). However, the activated ring in phenol If we use conc. nitric acid with phenol, we get 2,4,6-trinitrophenol. readily undergoes nitration with dilute nitric acid at If we use concentrated nitric acid we get room temperature: 2,4,6-trinitrophenol formed, shown below: OH OH conc. dil.nitric HNO3acid O 2N OH OH NO2 NO2 + NO2 If we use concentrated nitric acid we get 2,4,6-trinitrophenol formed, shown below: NO2 NO2 26.11 ring in phenol 10 Bromination of Phenols Chapter 25: Benzene and its en aqueous is added to abromine solutionat ofroom phenol, the bromine is a white Phenolbromine decolorises aqueous temperature, giving mediately decolorised and a white precipitate is formed. This is a substitution precipitate of 2,4,6-tribromophenol. tion and the white precipitate is 2, 4, 6-tribromophenol. BROMINATION OF PHENOL OH Br + 3HBr However, bromine water reacts readily with phenol, decolorising the orange solution and forming a white precipitate of 2,4,6-tribromophenol (see Figure 25.6): − + 3Br2 − Br − − Phenol undergoes similar reactions to benzene, but phenol does so under milder conditions. For example, bromine water will not react with benzene at room temperature. To produce bromobenzene we need pure bromine (not a solution) and an iron(III) bromide catalyst. − OH Br OH OH 2,4,6-tribromophenol Br Br white precipitate + 3HBr + 3Br2 → In th • Des phe rea mo ben • Tes com • Sum phe zene does not react with bromine except in the presence of a halogen Figure 25.6 Bromine water is added to aqueo Br ier catalyst (Section 26.6). In general, phenol is more readily attacked KEY P n benzene by electrophiles. This is because the lone pairs of electrons Br Br 11 Chlorination3Br of2Phenols (aq) 3HBr A similar product is formed by the action of chlorine water on phenol. Br PhenolAreacts with aqueous chlorine in water at room temperature, giving a similar product, formed by the action of chlorine water on phenol, is used in white precipitate solution as of the2,4,6-trichlorophenol. antiseptic TCP (see page 436). OH OH Cl Cl 3Cl2(aq) 3HCl Cl white precipitate 2,4,6-trichlorophenol (TCP) phenol as the ligand. Figure 26.14 summarises some of the important reactions of phenol. 12 Phenol Reactions Summary OH NO2 NO Br − CH 6 C 5 O C Br2(aq) OH − − NO2 NO2 Na O a or N − Br Br l OOCC6H5 − − − OH O3 N H . c con H 2SO 4 . c n co OH − 3 ) q a H( − − dilu te H NO2 O2N − O–Na+ − + − − − OH Which is the stronger acid, ethanol or phenol? 13 Acid Nature Phenol behaves as a weak acid. The aqueous solution has pH of around 5 or —10 6. (K of the acid is 1.3 x 10 ). onsider athe equilibrium: − phenol − H2O + OH – O + H3O + phenoxide ion see The whyposition phenol is a stronger acid than aliphatic alcohols such as ethanol of this equilibrium lies well over to the left-hand side. However, phenoxide ion, the negative charge on the O atom can be partly delocalise phenol is still a stronger acid than water or an alcohol. + the ring. This reduces the tendency of the phenoxide ion to attract H . r words it reduces its strength as a base. This increases the strength of 14 Acid Nature Phenol is more acidic than water, with ethanol being the least acidic of the three compounds. (phenol > water > ethanol in terms of acidity). Compound Dissociation in water pKa phenol C6H5OH(aq) ⇌ C6H5O−(aq) + H+(aq) 10.0 water H2O(l) ⇌ H+(aq) + OH−(aq) 14.0 ethanol C2H5OH(aq) ⇌ C2H5O− (aq) + H+(aq) 16.0 act that it or, showing structures: by the fact that it om sodium 15 Acid Nature from sodium CO ions,2i.e. it ate solutions, i.e. it bonicThe acid. dissociation of phenol may be shown as: than carbonic acid. n sodium oluble inItsodium in water. ydroxide on than in water. It The dissociation of ethanol is: − + H5O Na ) odium hydroxide The dissociation of ethanol is: − + tronger The dissociation of ethanol is: H O Na ) salt (C 6 5 molecules. ere are stronger water molecules. Phenol is more acidic than water, with ethanol being the the three compounds. We this can by explain this by least acidicleast of theacidic threeofcompounds. We can explain –10 –3 Phenol is more acidic than water, with ethanol being the –10 –3 looking at the conjugate bases formed on the right-hand K = 1.3 × 10 mol dm –10 –3 a O H H O looking at the conjugate bases formed on the right-hand K = 1.3 × 10 mol dm a 1.3 × 10 Ka = mol dm Nature enol is16 moreAcid acidic than water, OO HH with ethanol OObeing HH the side of theinequations in Table 24.3. Ththis eion, phenoxide ion, least acidic side of the three compounds. We cane phenoxide explain by of the equations Table 24.3. Th − by st acidic of the three compounds. We can explain this − C H O (aq), has its negative charge spread over the whole C H O (aq), has its negative charge spread over the whole 6 5 looking at the bases formed the right-hand The negative charge of the anion can6−beconjugate the benzene ring.on Figure 25.17 5 delocalised over The ion, C O (aq), has its negative charge spread over 6H5on king at thephenoxide conjugate bases formed the right-hand shows various ways in which this ion can be represented. ion as one of the lone pairs on the oxygen atom overlaps as one of the lone pairs on the oxygen atom overlaps side of the equations in Table 24.3. The phenoxide ion, the whole ion as one of the lone pairs on the oxygen atom overlaps eldite of the equations in Table 24.3. Th e phenoxide ion, with the delocalised π bonding system in the benzene ring. with the delocalised π bonding system in the benzene ring. − adhesive, compact discs and TCP are all ve, compact discs and TCP areantiseptic all –antiseptic O O C6H5O (aq), has its negativeOcharge spread over the whole ing phenol asmaterial. ahas starting material. H (aq), negative charge spread oversystem the whole as5aO starting with theitsdelocalised π bonding in the benzene ring. – – – O O oxygen atom ion as one of– the lone pairs on the overlaps as one of the lone pairs on the oxygen atom overlaps with the delocalised π bonding system in the benzene ring. – This makes the ionsystem moreinstable than ring. an ethoxide ion. TCP antiseptic are all π bonding – h the delocalised the benzene O CH CH ons of the − phenol O 3 – 2 CH3CH2 O – O– – (–) O O ol was the fi rst antiseptic to be used in ntury phenol was the first antiseptic to be used in phenoxide ion,phenoxide ion, ethoxide ion, with ethoxide ion, with ManyLister. people usedpeople to die when their (–) (–) seph Many used to die when their negative charge with negative – negative charge with negative or or O O CH CH dme after an operation. However, when Lister 3 2 – over spread over concentrated onconcentrated on infected after an operation. However, when Lister O spreadcharge CH3CH2 charge the oxygen the whole ion eatre and recovery wards with phenol (–) the oxygen the whole ion perating theatre and recovery wards with phenol ically reduced. Nowadays, derivatives ere c todramatically be used inreduced. Nowadays, derivatives This delocalisation reduces the charge density of the Th is delocalisation reduces the charge density of the use phenol itself is toxic and caustic. ethoxide ion, with phenoxide ion, used because phenol itself is toxic and caustic. ethoxide ion, with phenoxide ion, ie when their negative charge on the phenoxide ion compared with ® 6-trichlorophenol, found in TCP and negative charge on the phenoxide ion compared with negative charge with negative ® − − + such as 2,4,6-trichlorophenol, found in TCP and negative charge with negative 26_17 Cam/Chem AS&A2 −H5O (aq). Therefore − + not OH (aq) or C H (aq) ions are wever, when Lister 2 without the nasty side-effects of phenol. concentrated on charge spread over OH (aq) or C H O (aq). Th erefore H (aq) ions are not 2 5 ill bacteria without the nasty side-effects of phenol. 433 concentrated on charge spread over Barking Dog Art as strongly attracted to the phenoxide ion as they are to The negative charge of the anion can be delocalised over the benzene ring. Figure 25.17 shows various ways in which this can be represented. 17 Acid Nature –O We will consider the stability of the anion/conjugate base. 17 Representations of the phenol O O O – – The lone pair on the O— in the phenoxide ion overlaps with the delocalised – system in the benzene ring. This spreads out the negative charge in the ion. – or O (–) O or (–) (–) ( –) 26_17 Cam/Chem AS&A2 Barking Dog Art 433 18 Acid Nature The O is thus less negative and the H+ is not attracted back as strongly. The anion is thus stabilised and phenol is a stronger acid than ethanol i.e. the backward reaction is favoured less. In ethanol this delocalisation does not occur and indeed the negative charge on the O in the ethoxide ion is increased by the electron-donating alkyl group. 19 Acid Nature The phenoxide ion (C6H5O−) is more stable than the ethoxide ion (C2H5O−) and hence its formation is favoured. In the phenoxide ion, the charge is delocalised and is stable. Thus phenol reacts with alkalis unlike alcohols. Compound Dissociation in water pKa phenol C6H5OH(aq) ⇌ C6H5O−(aq) + H+(aq) 10.0 water H2O(l) ⇌ H+(aq) + OH−(aq) 14.0 ethanol C2H5OH(aq) ⇌ C2H5O− (aq) + H+(aq) 16.0 an acid, phenol reacts with alkalis such as sodium hydroxide. Hydroxide 20 Phenol In Alkalis + + om sodium hydroxide remove H (H3O ) ions, displacing the above – brium to the right and forming a solution of sodium phenoxide, C H O 6 5 Although phenol is only slightly soluble in water, it dissolves well in an alkali. Because of this reaction, phenol is much more soluble in aqueous sodium Phenol will react with an alkali to give a salt plus water: xide than it is in water. − − NaOH + OH – O Na + + H2O sodium phenoxide ol was once known as ‘carbolic acid’, and it was used as one of the earliest ptics. The Edinburgh doctor, Joseph Lister first used it in the 1860s to nt wounds going septic after surgery. Unintentionally, it had been used antiseptic even before this, because the old-fashioned way of treating 21 Acidic Strengths Table 10.2 Acidity of alcohols, phenol and carboxylic acids Add blue litmus Add sodium hydroxide Add sodium carbonate Alcohols Stays blue No reaction No reaction Phenol Goes red Salt formed No reaction Carboxylic acids Goes red Salt formed CO2 evolved Phenol as an alcohol Phenol does not react as an alcohol as readily as aliphatic alcohols, such as ethanol or propan-2-ol. For example it does not react with carboxylic acids to form esters. This is because the lone pair of electrons on the oxygen atom is drawn into the ring, making the oxygen less susceptible to electrophilic attack. Reaction with acyl chlorides
 0
0
advertisement
Related documents
Download
advertisement
Add this document to collection(s)
You can add this document to your study collection(s)
Sign in Available only to authorized usersAdd this document to saved
You can add this document to your saved list
Sign in Available only to authorized users