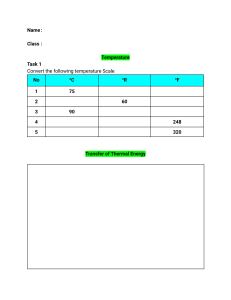
Name: Class: Date: HEAT TRANSFER BY CONDUCTION, CONVECTION & RADIATION Temperature and Thermal Energy All particles of matter have kinetic energy (KE) and are in constant motion, even at colder temperatures. Temperature is a measurement of the average KE of the particles in an object at a particular location. On the other hand, thermal energy is the total energy of ALL the particles in an object. Temperature is like counting the average amount of money each person has in his or her pocket. Thermal energy is like counting the total money that everyone in the room has by adding it all up. Heat Heat is defined as the movement of thermal energy from a warmer location to a colder location when a temperature difference exists. The bigger the temperature difference, the faster heat will flow. Heat will continue to flow from the warmer location to the colder location until both locations are at the same temperature. This is called thermal equilibrium. Heat moves by 3 different processes: conduction, convection, and radiation. Three Methods of Heat Transfer Method 1: Conduction Conduction is the process where heat is transferred from one particle to another by direct physical contact. If you have ever picked up a cup of hot chocolate and it warmed your hands, you have experienced conduction. Conduction occurs any time objects at different temperatures are in direct contact, or when one part of an object is warmed up to a different temperature compared to the rest of the object. Since the particles are very close together, the fast-moving hotter particles collide with the slower-moving cooler particles, transferring KE to them. This makes the cooler particles move more, increasing their temperature. As long as the objects are touching, conduction will continue until the temperatures of the locations are equal. Conduction occurs mainly in solids, but it can also occur between solids and liquids. A thermal conductor is a material that conducts heat easily. Conductors have a high density, meaning that their particles are closely packed together. The higher the density, the better the conductor. Metals such as steel, gold and copper, which have high densities, are good thermal conductors. A thermal insulator is a material that does not conduct heat easily. Insulators have a low density, meaning that their particles are loosely packed together. The lower the density, the better the insulator. Materials such as wood, wool, cotton and cork are good thermal insulators. Air is the best insulator, so materials that trap air, such as Styrofoam or aerogel, are particularly good thermal insulators. Method 2: Convection Convection is the process where heat is transferred by the bulk movement of particles from a warmer location to a colder location. If you have ever put your hand over a pot of boiling water and felt the heat rising up, you have experienced convection. Convection can only occur in fluids, which include liquids and gases, because their particles are free to move, unlike the particles in solids. The repeated rising of heated fluid particles and sinking of cooled fluid particles creates a circular motion called a convection current. Convection currents in many situations, such as pots of water on a stove, hot air from a furnace flowing throughout a home, the movement of giant air masses in the atmosphere to create weather patterns, as well as the slow movement of the Earth’s mantle causing plate tectonics. Method 3: Radiation Thermal radiation is the process where heat is transferred by electromagnetic (EM) waves. Thermal radiation is also called infrared radiation. Unlike conduction and convection, thermal radiation does not require particles to transfer thermal energy. For example, all of the Sun’s thermal energy must travel through 150 million kilometers of outer space to reach Earth. Since outer space is a vacuum, which is an area devoid of particles, conduction and convection cannot there since as both processes require particles to work. While the Sun is our biggest source of thermal radiation, all objects give out (emit) and take in (absorb) thermal radiation by some amount. The hotter an object is, the more thermal radiation it gives out. Different materials absorb heat radiation at different rates. The color and luster of a material are properties that both affect the amount of thermal radiation that is absorbed or emitted. In general, black or other dark-colored objects absorb thermal radiation better than light-colored objects. By the same token, black or dark objects also emit thermal radiation better than light-colored objects. In addition, dull surfaces absorb and emit thermal radiation faster than shiny surfaces, which reflect thermal radiation. This is why most care engines are painted a dull black color; to better emit the tremendous thermal energy they produce. Table: Comparison of Different Materials Ability to Absorb and Reflect Thermal Radiation Color Luster Ability to Absorb Ability to Emit Radiation Radiation Dark Dull / Matte Good Good Light Shiny Poor REVIEW: HEAT TRANSFER BY CONDUCTION, CONVECTION & RADIATION definitions. 1. Contrast “temperature” and “heat” by stating their basic Poor Temperature “Temperature is a measurement of the average Kinetic energy of the particles in an object at a particular location.” Heat the movement of thermal energy from warm to cold when a temperature difference exists 2. A cup of water and a bathtub full of water are at the same temperature, but have very different thermal energies. Which one has more thermal energy? Explain why. The bathtub because it contains more particles, and since thermal energy is the total, the bathtub has more thermal energy. 3. In which direction does thermal energy always move? Explain your answer. It goes both up (hot air rises) and to cold objects. Since hot air is less dense than cold air, it rises, while cold air sinks. 4. When does thermal energy stop moving? Explain your answer. Thermal energy stops moving when both the original cold thing that it was transferring to, and itself are the same temperature; Thermal energy stops moving (to a colder object) when both the hot object and the cold object it was transferring to are the same temperature. 5. Match the type of heat transfer with its correct definition. _ii___ a. conduction i. The process where heat is transferred by electromagnetic (EM) waves. iii____ b. convection ii. The process where heat is transferred from one particle to another by direct physical contact i____ c. thermal radiation iii. The process where heat is transferred by the bulk movement of particles in a fluid. 6. Fill in the spaces in the table below by writing either conduction, convection, or radiation. Example Type of Heat Transfer a. A lake heated by warm water rising from a hot spring on its bottom. Convection b. Sunlight melts a wax crayon left outside on a warm sunny day. Radiation c. A gas burner set on a hot stove heats the bottom of a pot. Conduction d. An ice cube feels cold when you hold it in your hand. Conduction e. A kite rises high above a hot, sandy beach. Convection f. You feel the warmth from a fireplace when sitting in front of it. Radiation