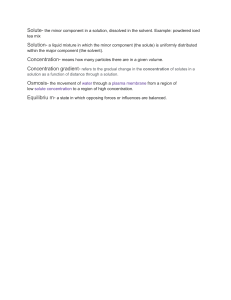
SOLUTE AND SOLVENT IN A SOLUTION Solution - is a homogeneous mixture of one or more liquids or with one or more substances. Solvent - is a substance with dissolving capability, thus can dissolve with other substances. Solute - is the substance that dissolves in solvent in order to form a solution. The difference between solvent and solute is that solute is the one to be dissolved and the solvent is responsible for dissolving it. Like for example in a mixture of lemon juice powder and water, the solvent is the water (liquid) and the solute is the lemon juice powder (solid) after mixing, it forms lemonade juice solution. Water is the universal solvent. - Some solutes dissolve easily than others. This property of solutes is called solubility. - But not all solutes dissolve in all solvent. - A solute is considered soluble if an appreciable amount of it can be dissolved in a given amount of the solvent. - A solute considered insoluble if very little of it dissolves in a given amount of the solvent. - Some substances dissolved faster than the others smaller particles dissolve faster than substances with bigger particles. Factors/conditions Affecting Solubility of Solutes in a Solvent 1. Manner of stirring - the way you stir a glass with salt increases the move of the solvent, thus enabling solubility of the solution. This condition affects the solubility of solutes in a solvent. Factors/conditions Affecting Solubility of Solutes in a Solvent 2. Temperature of the solvent - solubility increases with temperature. When you compare hot and cold water, it was hot water with high temperature dissolved the sugar faster. Factors/conditions Affecting Solubility of Solutes in a Solvent 3. Size of the solute particles - the size particles affect the solubility. The larger the molecules of the solute are, the more difficult it is for solvent to Write the correct answer in the required box to complete the missing term.