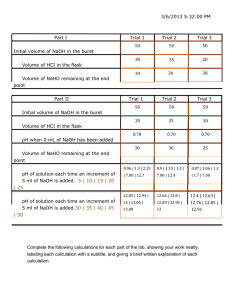
Acid Base Titration Pre-Lab questions: 1. Phenolphthalein in solution of basic pH gives a _____________ color. 2. Phenolphthalein in a solution of acidic pH gives a __________________ color. 3. Write the neutralization reaction that occurs when solutions of HCl and NaOH are mixed. 4. How does the number of H+ ions compare with the number of OH- ions in an acid solution? __________________________________________________________________________ 5. How does the number of H+ ions compare with the number of OH- ions in a basic solution? __________________________________________________________________________ 6. How does the number of H+ ions compare with the number of OH- ions in a neutral solution? __________________________________________________________________________ 7. Does the volume of the acid need to equal the volume of base in neutralization? Explain. ___________________________________________________________________________ ___________________________________________________________________________ Materials: Burette Ring stand Erlenmeyer flask Unknown molarity of sodium hydroxide funnel 1.0 M HCl phenolphthalein Procedure: In this experiment the Molarity of the NaOH solution will be determined. This is done by using the NaOH solution as a titrate and titrate standard acid solution until you reach the endpoint. 1. Rinse the buret three times with deionized water. Rotate the stopcock to let the di-water run out into the sink. 2. Rinse the buret again once with about 5 mL of the sodium hydroxide solution. Rotate the stopcock to let the sodium hydroxide solution to run out into the sink. 3. Clamp the buret in position and fill with approximately 40 mL of the sodium hydroxide solution, using the funnel 4. Place the sodium hydroxide solution beaker under the buret and open the stopcock to allow any air bubbles to escape from the buret. Then fill the buret to between the 0 and 10 mL mark. 5. Record the precise level of the sodium hydroxide solution into the data table. 6. Measure 20.0 mL of 1.0M HCl into an Erlenmeyer flask. 7. Add 3 drops of phenolphtalien to the Erlenmeyer flask. 8. Set the flask under the buret and add the sodium hydroxide solution, swirling the flask constantly. 9. As you approach the endpoint, a pink spot will start to appear. 10. Slow down the buret and proceed drop wise after this point. 11. Stop as soon as the first permanent pale pink color appears. This is the endpoint. 12. Record the volume of sodium hydroxide that was used. 13. Repeat two more times. More trials can be run if % error is too high. Data: Trial number Molarity of acid (M) Volume of acid (mL) Volume of base (mL) Start: Difference: #1 Finish: Start: Difference: #2 Finish: Start: Difference: #3 Finish: Start: Difference: Extra Finish: Start: Extra Extra Difference: Finish: Start: Difference: Finish: Calculations: Must show all work Calculate the Molarity of the NaOH for Trial number Work : Molarity of Base (M) MAVA = MBVB 1 2 3 Extra Extra Extra Must show all work: Average Molarity for 3 trails _________________________ Correct Molarity of the base given by the instructor________________________ 2. Calculate % error : % error = ( Ι Average Molarity – Correct Molarity Ι ) x 100 = Correct Molarity Must show all work: Lower than 3% error = +5 on lab Higher than 7% = - 5 on ab