SCH4U Chemistry Exam Review: Equilibrium, Structure, Organic, Electro
advertisement

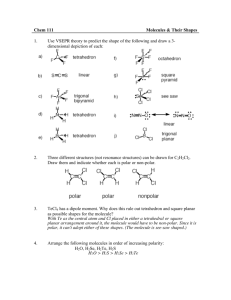
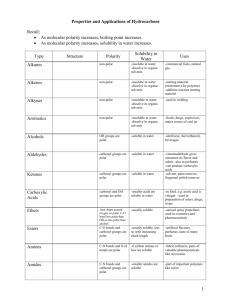
SCH4U: Exam Review - Answers to non-calculation/non-text problems Unit 2: Chemical Equilibrium a) 1a) Solubility Equilibrium least soluble soluble AgI most Ag2CrO4 1b) AgI(s) �Ag+(aq) + I-(aq) b) 3a) Acid-Base Equilibrium Neutral Ag2CO3 Acidic KNO3 AgCl AgNO3 Basic N2H5Cl NH4I LiC6H5COO LiCN 3b) Acidic 1+ � N H (aq) + H O1+(aq) N2H1+ 2 4 5 (aq) + H2O(l) 1+ 3 NH4 (aq) + H2O(l) � NH3(aq) + H3O (aq) Basic C6H5COO1-(aq) + H O(l) � C 6H 5COOH(aq) CN1-(aq) + H2O(l) HCN(aq) + OH1-(aq) + OH1-(aq) Unit 3: Atomic Structure and Classification of Solids 1) Bohr introduced the idea of electrons with quantized energy resulting in energy levels of fixed energy DeBroglie wave/particle duality - electrons can be considered as both having particle and wave like properties Heisenberg uncertainty principle - the exact location of an electron cannot be known with any degree of certainty, lead to the concept of orbitals Schrodinger developed wave equation that can be used to predict regions of probability of finding electrons Pauli Pauli exclusion principle - only two spin paired electrons can occupy the same orbital 2a) 2b) 2c) An orbital is a region or volume of space around an atomic nucleus in which there is a 95% chance of finding an electron. Maximum # electrons = 2n2 n=2 2 sublevels: 2s and 2p 2s = one spherical shaped “s” orbital 2p = three dumbbell shaped “p” orbitals along x,y and z axes n=4 4 sublevels: 4s, 4p, 4d, 4f 4s = one spherical shaped “s” orbital 4p = three dumbbell shaped “p” orbitals along x,y and z axes 4d = five “d” orbitals 4f = seven “f” orbitals -23a) 4) 3b) 27 Co 1s2 2s2 2p6 3s2 3p6 4s2 3d7 n l ml ms 1s 1 0 0 ±½ 2s 2 0 0 ±½ 2p 2 1 -1 +½ 2 1 0 +½ 2 1 +1 +½ Fe2+ 1s2 2s2 2p6 3s2 3p6 3d6 5) Compound General formula Shape Polarity PCl3 AX3E trig. Pyramidal polar SiF4 AX4 tetrahedral non-polar CO32- AX3 planar triangle N/A H2S AX2E2 bent slightly polar XeCl4 AX4E2 square planar non-polar PO43- AX4 tetrahedral N/A O3 AX2E bent non-polar CO AX linear polar NO31- AX3 planar triangle N/A SF4 AX4E see-saw polar 6) Fluoromethane is a polar molecular solid while methane is a non-polar molecular solid. The DDF between CH3F molecules will be much stronger than the LDF between CH4 molecules. As a result, CH3F will have a higher BP than methane. 7) Iodine is a non-polar molecule and will therefore be more soluble in a non-polar solvent like tetrachloromethane rather than the very polar water. 8) ethanal: 3 o a,b,d) C1 = sp hybridization, tetrahedral arrangement, 109.5 C2 = sp2 hybridization, planar triangular arrangement, 120o c) [C1(2sp3 ) -ó- H(1s)] x3 C1(2sp3 ) -ó- C2(2sp 2) C2(2sp2) -ó- H(1s) C2(2sp2) -ó- O(2p) C2(2p) -ð- O(2p) -39) Substance type of solid identity A molecular (polar) methanol B network quartz C molecular (non-polar) sulphur D metallic aluminum E ionic potassium chloride F molecular (non-polar) methane G ionic calcium oxide Unit 4: Organic Chemistry 1) 2) a) 3-ethyl-4methyl-2-hexene c) 3,4,5-trimethylheptane e) isopropylcyclopentane g) 2,4-cyclopentadiene-1-ol i) 1,3-dichlorobenzene (m-dichlorbenzene) k) 1-ethoxypropane m) 2-hexanone o) ethylbenzoate q) N-ethyl-N,2,4-trimethyl-3-pentanamine b) 3,5-dimethylcyclohexene d) 3-methyl-1,3-pentadiene f) 3-methylbutanal h) cyclohexanone j) 2,3-butanediol l) methylpropanoate n) butanoic acid p) N,3-dimethyl-2-butanamine r) N-isopropylbutanamide -43) a) 3-methyl-1-butyne + 2Cl2 = 1,1,2,2-tetrachloro-3-methylbutane b) methane + excess O2 = CO2 + 2H2O c) 2-methyl-3-pentanol + KMnO4 = 2-methyl-3-pentanone d) methylpropene + HBr = 2-bromo-methylpropane e) 2-methylbutanal + KMnO 4 = 2-methylbutanoic acid f) cyclopentene + H2O (H2SO 4 catalyst) = cyclopentanol g) dimethyl-1,3-propandiol + KMnO 4 = dimethylpropanedial = dimethylpropanedioic acid h) ethanoic acid + phenol = phenylethanoate + water a) c) d) e) f) g) h) 4) propanone 2-propanol 2-chloropropane propanoic acid propane dipole-dipole H-bonding dipole-dipole H-bonding LDF highly soluble highly soluble slightly soluble highly soluble insoluble lowest BP highest BP propane < 2-chloropropane < propanone < 2-propanol < propanoic acid -5Unit 5 - Electrochemistry 1) red ½ rxn: Cl2 + 2e1- = 2Cl1- Er = 1.36 V ox ½ rxn: Zn = Zn2+ + 2e1cell: Cl2 + Zn = Zn2+ + 2Cl1- Eox = 0.76 V red ½ rxn: Pb2+ + 2e1- = Pb Er = - 0.13 V ox ½ rxn: 2Na = 2Na1+ + 2e1- Eox = 2.71 V cell: Ecell = 2.12 V Pb2+ + 2Na = 2Na1+ + Pb Ecell = 2.58 V red ½ rxn: Au3+ + 3e1- = Au Er = 1.50 V ox ½ rxn: 3Ag = 3Ag1+ + 3e1- Eox = - 0.80 V cell: Au3+ + 3Ag = Au + 3Ag1+ Ecell = 0.70 V 2) red ½ rxn: 2Ag1+ + 2e1- = 2Ag E = 0.80 V r 2+ 1E = 2.37 V = Mg + 2e ox ½ rxn: Mg = 1+ 2+0x cell: 2Ag + Mg 2Ag + Mg Ecell = 3.17 V 3) a) b) c) 1+ 12+ � 5H2C2O4 + 6H + 2MnO4 10CO2 + 2Mn + 8H2O � 2CrO 2- + 3Br1- + 5H O + 2OH1312Cr(OH)6 + 3BrO 4 2 � 6K O + N 2KNO3 + 10K 2 2