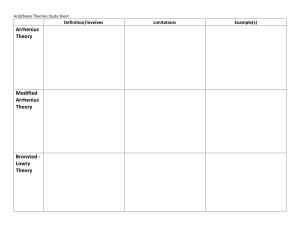
Introduction in acid and base Chapter 3 Lesson 1 Plan of lesson Period 1 properties’ of acid and base . Period 2 the Bronstedlowry model . Period 3 lewis model Period 1 objective properties of acid and base Hydronium and hydroxide ions Arrhenius model properties of acid and base Physical properties Chemical properties water self ionization Acid and base solution Arrhenius model Period 2 the Bronsted-lowry model • The objective Bronsted lowry acid and base Conjugate acid and base . For the following reaction, which of the following is a conjugate acid-base pair? HC2O4-(aq) + H2O(l) H3O+(aq) + C2O42–(aq) a. HC2O4– and H2O b. HC2O4– and H3O+ c. H2O and C2O42– d. HC2O4– and C2O42– Amphoteric substance Monoprotic and polyprotic acid