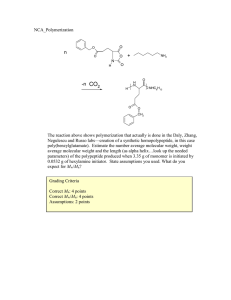
SUSPENSION POLYMERISATION Group member: Khairul Hakimi Bin Ma’amor (2020883664) Tengku Ahmad Faiz Bin Tengku Shamsul Arjunaidi (2020469766) Ismaizzamuddin Bin Ismadi (2020478422) Muhammad Nur Hazim Bin Nor Azhar (2020601366) What is Suspension Polymerization?? Polymerization: A process of reacting monomer molecules together. Suspension Polymerization: ● A heterogeneous radical polymerization process ● Used mechanical agitation to mix a monomer or mixture of monomers in a liquid phase. ● Basically, used in the production of many commercial resin. IUPAC definition: 1. The initiator is located mainly in the monomer phase. 2. Droplets in suspension polymerization have 10 micrometer diameter. SUSPENSION POLYMERISATION PROCESS Process of suspension polymerization : a) FORMULATION (MONOMER ,STABILIZER ,DISPERSING MEDIUM(EXAMPLE WATER ),MONOMER INSOLUBLE INITIATOR,EXAMPLE PEROXIDE,AZO COMPOUND). b) PROCESS POLYMERIZATION : DROPLET ARE DISPERSED IN WATER ACT AS SEPARATE MICROREACTOR,INIATE BY ORGANIC INITIATOR TO START POLYMERISATION .MONOMER CONVERT TO POLYMER ,DROPLET TRANSFER TO STICKY VISCOUS (MONOMER/POLYMER) GRADUALLY BECOME SPHERICAL SOLID POLYMER(PARTICLE HAVE SIZE 100 OF MICROMETER).POLYMERIZATION SETTLE OUT WHEN AGITATION STOP. PROCESS SUSPENSION FLOWCHART Aquaus phase🡪 monomer mix diluent and water and iniator Suspension polymerization. phase 🡪polymer convert to polymer +monomer until finish all monomer ③ ③ Polymer +water(powder or bead polymer) SUSPENSION POLYMERIZATION PROFILE (CON%,PRESSURE,TEMPERATURE). ① ③ ② As with any other reaction, temperature changes the rate constants associated with the polymerization. ... As the rate of termination increases, the degree of polymerization decreases, and the polymers will be shorter. • LIQUID PHASE FOR SUSPENSION:LOW VISCOSITY ,HIGH THERMAL CONDUCTIVITY AND LOW TEMPERATURE VARIATION OF VISCOSITY. • TWO TYPE OF SUSPENSION POLYMERIZATION 🡪 a) BEAD : FOR NON POROUS POLYMER PARTICLE WHICH POLYMER USE SOLUBLE IN MONOMER. b) POWDER: POROUS PARTICLE WHICH POLYMER INSOLUBLE IN MONOMER. c) HE POLYMER BEADS THAT RESULT CAN RANGE IN SIZE FROM 100 NM TO 5 MM. THE SIZE IS CONTROLLED BY THE STIRRING SPEED, THE VOLUME FRACTION OF MONOMER, THE CONCENTRATION AND IDENTITY OF THE STABILIZERS USED, AND THE VISCOSITIES OF THE DIFFERENT COMPONENTS. THE FOLLOWING EQUATION DERIVED EMPIRICALLY SUMMARIZES SOME OF THESE INTERACTIONS: PARAMETER EFFECT PARTICLE SIZE POLYMER FOR SUSPENSION PROCESS. • D= IS THE AVERAGE PARTICLE SIZE • K INCLUDES PARAMETERS RELATED TO THE REACTION VESSEL DESIGN, • DV IS THE REACTION VESSEL DIAMETER • DS IS THE DIAMETER OF THE STIRRER, • R IS THE VOLUME RATIO OF THE MONOMER TO THE LIQUID MATRIX, • N IS THE STIRRING SPEED • ΝM AND ΝL ARE THE VISCOSITY OF THE MONOMER PHASE AND LIQUID MATRIX RESPECTIVELY • Ε IS THE INTERFACIAL TENSION OF THE TWO PHASES, • CS IS THE CONCENTRATION OF STABILIZER. ADVANTAGES ● Easy to control temperature and viscosity . ● Processing and purification of polymer much easier. ● Great heat transfer medium. ● The purify of obtained polymer is higher. DISADVANTAGES ● This method can be adopted with water insoluble monomers. ● hard to control the polymer size. ● Highly agitation sensitive ● Large volume of reactor is taken up by water Chemicals needed suspension polymerisation For a typical suspension polymerisation, the following chemicals are required: - Monomer(s) that are insoluble in water such as (styrene, methyl methacrylate and others) An Initiator, soluble in the monomer (such as dibenzoyl peroxide) Stabiliser (such as PVA, gelatin, cellulose are used together with inorganic stabilisers as well such as kaolin, magnesium silicate, aluminium hydroxide, calcium hydroxide, and magnesium hydroxide if necessary) Water as dispersion agent (continuous phase) Protective colloids (such as polyvinyl alcohol and partially saponified polyvinyl acetate, polyvinyl pyrrolidone, and methyl cellulose). Suspension agents (such as barium sulfate, calcium carbonate, talcum, and hydroxyl apatite). Examples of products - Polyvinyl chloride (commonly used in pipes, wiring cables and other household items) Polyvinyl acetate (commonly used in adhesives like wood glue and many others) Polystyrene Styrene-divinyl benzene copolymer beads END OF SLIDE