Biochemistry Exam: Protein Structure & Amino Acids
advertisement

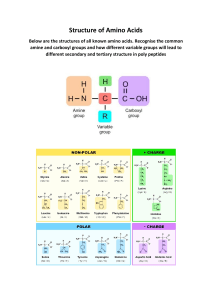
Unit 2 Biochem Exam https://quizlet.com/494227013/unit-2-biochem-exam-flash-cards/ The secondary structure of a protein depends on which of the following types of bonds/interactions hydrogen bonds β-sheets characteristically distribute hydrophobic side chains on both sides of the sheet, and ____ β-sheets are usually arranged with all their hydrophobic residues on one side of the sheet parallel, antiparallel Proteins destined for an extracellular location are characteristically glycoproteins This is the structure of selenocysteine. How is this amino acid classified based on its functional group associated with selenium? hydrophilic polar How many residues are present in vasopressin or oxytocin 9 Antiparallel β-sheets have usually all of their hydrophobic residues on one side of the sheet. What is the most important property of the R groups of the amino acids their polarity According to the chart, where is glycine found in the zwitterion form 6.06 Secondary structure is associated with which of the following hydrogen bonding within the backbone Identify which of the following is not an example of secondary structure of proteins a. ß-pleated sheet b. α-helix c. hydrophobic folding d. random coil Which of the following contributes to the tertiary structure of proteins hydrogen bonding between the side chains Which of the listed amino acids is classified as a basic amino acid lysine Fibrous proteins contain polypeptide chains ____ producing long fibers or large organized approximately parallel along a single axis β-Turns in a peptide chain form a tight loop with hydrogen bonding of the carbonyl oxygen with: amide proton of the residue three positions down the chain. Which of the following side chains are hydrophobic none of these You know that the protein you want to purify from a natural source forms a multimer with multiple sub-units giving a molecular weight in solution much bigger than visualised denatured on SDS-PAGE. There is only a small amount of the target protein in the total protein sample. Which of the following is an appropriate purification strategy IEX followed by SEC Which of the following amino acids has a net negative charge at physiologic pH (~7.4)? Glutamic Acid Which of the peptides would absorb light at 280 nm Ala-Ala-Trp α-Helix and β-strand are components of ____ structure secondary Which of these techniques is often considered a suitable "polishing" step in a protein purification strategy Size-exclusion chromatography (SEC) For an application where you require a sample of your target protein at high purity, what would be a good purification strategy? Assume that your starting point is E. coli cells in which the target protein fused to an affinity tag has been over-expressed AC followed by ion-exchange (IEX) followed by SEC Which of the following side chains are hydrophobic? nonpolar Which of the following is true of an amino acid at a pH below its isoelectric point We cannot predict its charge Which of the following contains a disulfide bond cytesine In which of the following do both amino acids have a polar but neutral side chain cysteine and serine Protein synthesis proceeds in which direction from N-terminus to C-terminus Which of the following is true about zwitterions they are electrically neutral Which of the following is associated with the quaternary structure of proteins the interaction of protein subunits Which of the following is an allosteric protein hemoglobin Which of the following amino acids has the lowest pI glutamic acid . Which of the following is alanylglycine? What is the net charge of histidine at pH 2 +2 What is the name for proteins which assist other proteins in attaining their correct secondary and tertiary structures chaperones What type of biopolymer contains an amide as a connector between monomers protein In which of the following do both amino acids have a basic side chain histidine and lysine In the tertiary structure of a protein, an electrostatic interaction could form between the Rgroups of which two amino acids Glu and Arg Which of the following statements about Nuclear Magnetic Resonance (NMR) is correct The majority of known protein structures have been determined using NMR. The peptide bond has partial ____ character double bond Which amino acid is least hydrophobic glutamine The intrinsic properties of the 20 amino acids include all EXCEPT α-Carbon is symmetric. Which of the following is a type of secondary structure a helix and beta pleated sheet (both) What is the starting point for selection of a suitable IEX matrix for purification of a recombinant protein Prediction of isoelectric point (pI) from the amino acid sequence Which type of interaction is not associated with tertiary structure? None, they are all associated with tertiary structure 46. For the peptide, Ala-Asp-Gly-Leu-Phe, what would be the expected charges at pH 2 and 8? a.+2, +1 b.+2, 0 c.+1, -1 d.-1, 0 c.+1, -1 Which functional group is involved in linking together the amino acids of proteins? a.amine b.anhydride c.carbonyl d.carboxyl amine Which of the following statements about the use of mass spectrometry in protein investigation is correct a. Ionic fragments are separated according to their mass-to-charge ratio in Mass spectrometry. b .Mass spectrometry involves ionized molecules in the liquid phase. c .Mass spectrometry is used for analysing the three-dimensional shape of peptides. d. Mass spectrometry involves the separation of ionic fragments on a gel. What type of interaction is depicted by the dashed line? a.hydrogen bonding b.covalent bonding c.hydrophobic interactions d.salt bridges e.metal ion coordination Alpha helices are stabilized primarily by: a.hydrogen bonds between the main chain peptide bond component atoms. b.electrostatic interactions between R-groups. c.hydrophobic interactions between the α-carbons of the main chain. d.hydrogen bonding between the R-groups. e.hydrophobic interactions between R-groups and the solvent water. Which of the following is true of all α-amino acids? a.All α-amino acids are also D-amino acids. b.All α-amino acids are also L-amino acids. c.The amino group is attached to the carbon of the carboxyl group. d.The amino group and the carbon of the carboxyl group are attached to the same carbon atom. The denaturing agent urea affects which of the following? a.hydrogen bonding b.disulfide bonds c.hydrophobic interactions d.salt bridges Electrostatic bonding between separate subunits of hemoglobin is an example of which of the following? a.quaternary structure b.tertiary structure c.secondary structure d.primary structure Which of the following amino acids absorbs UV radiation at 280 nm all of these The unique cyclic structure of which of the following amino acids plays a central role in the formation of alpha helices and beta sheets? a.proline b.glycine c.lysine d.cysteine 56. ____ amino acids are almost never found in the interior of a protein, but the protein surface may consist of ____ amino acids. a.Nonpolar, both polar and nonpolar b.Nonpolar, mostly nonpolar c.Polar, both polar and nonpolar d.Polar, only polar e.Polar, only nonpolar A polypeptide with a net positive charge at physiologic pH (~7.4) most likely contains amino acids with R groups of what type? Basic R group Which of the following amino acids would generally not be found in an α-helix? a.Ala b.His c.Leu d.Ser e.Met . Proteins with two different polypeptide chains are: a.monomeric proteins. b.trimeric proteins. c.homodimeric proteins. d.heterodimeric proteins. Planarity of the peptide bond means that no rotation occurs about the _____ bond while rotation is allowed about the ____ and ____ bonds. a.C(O)-N; Cα-Cβ; N-Cα b.C(O)-N; Cα-C(O); N-Cα c.Cα-C(O); C(O)-N; N-Cα d.N-Cα; Cα-C(O); C(O)-N C(O)-N; Cα-C(O); N-Cα 61. Where Cα is the α-carbon, N represents the amide nitrogen and Co is the carbonyl carbon of amino acids in a peptide, the peptide backbone of a protein consists of the repeated sequence: a.−Cα−N−Co− b.−N−Co−Cα− c.−N−Cα−Co− d.−Co−Cα−N− Which of the following statements about protein separation by gel filtration are correct? Please select all that apply a. When a mixture of proteins is separated by gel filtration, the smallest molecular weight protein is eluted first. b. When a mixture of proteins is separated by gel filtration, the largest molecular weight protein is eluted first. c. When a mixture of proteins is separated by gel filtration, the largest molecular weight protein is eluted last. d. When a mixture of proteins is separated by gel filtration, the smallest molecular weight proteins flow around the beads. In the tertiary structure of a protein, a hydrophobic interaction could form between the R-groups of which two amino acids? a.Ala and Ser b.Asn and Tyr c.Leu and Val d.Val and His Which of the following statements about column chromatography is correct? a.Affinity chromatography involves the attachment to the column matrix of ionic groups known that bind to the wanted protein. b.In reverse phase chromatography the wanted protein can be selectively eluted by solutions of different hydrophobicities or ionic strengths c.Ion-exchange chromatography separates proteins according to their size. d.Gel-filtration chromatography separates proteins on their ability to bind to specific groups on the column matrix. In reverse phase chromatography the wanted protein can be selectively eluted by solutions of different hydrophobicities or ionic strengths How many tripeptides can be made using the 20 standard amino acids? a.60 b.400 c.6840 d.8000 If an aspartic acid residue were present in the interior of a globular protein, it would most likely be _________. tightly associated with the R-group of a lysine residue Which of the following side chains are hydrophilic? a.acidic b.basic c.polar but neutral d.all of these In the tertiary structure of proteins which amino acid is involved in forming covalent bonds? a.alanine b.aspartic acid c.cysteine d.tryptophan Which of the following pairs of amino acids can form a salt bridge? a.alanine and glutamic acid b.aspartic acid and lysine c.leucine and phenylalanine d.serine and tyrosine Which of the following amino acids can coordinate to a metal ion? a.arginine b.glutamic acid c.isoleucine d.phenylalanine glutamic acid Which of the following correctly describes the relationship between the primary structure of a protein and protein function? a.Changing one amino acid always affects protein function. b.Changing one amino acid may or may not affect protein function. c.Changing one amino acid never affects protein function. d.There is no relationship between primary structure and protein function. Which of the following is true of the isoelectric point of amino acids? a.The isoelectric points of all amino acids are the same. b.It is the pH at which an amino acid conducts electricity. c.It is the pH at which an amino acid has equal numbers of positive and negative charges. d.All of the above are true. Which of the following methods could be used to check the molecular weight of your purified protein? a.SDS-PAGE b.Mass spectrometry c.Analytical SEC d.All of the above. Which amino acid has an amide as a part of its R group? a.glutamine b.glutamate c.glycine d.glutamic acid e.histidine . For the peptide Ala-His-Glu-Val-Asp-Cys-Lys-Leu, what is the net charge at pH 3? a.-1 b.0 c.+1 d.+2 e.+3 The amino and carboxyl groups of amino acids react in a head-to-tail fashion, eliminating water, and forming a covalent ____ linkage typically referred to as a(n) ____ bond. a.ester, aromatic b.anhydride, phosphoanhydride c.amide, peptide d.dehydration, hydrogen 78. Which of the following statements about SDS polyacrylamide gel electrophoresis is correct? a.Wanted proteins can be tested for their biological activity after separation by SDS polyacrylamide gel electrophoresis. b.Proteins are solubilized but not denatured when separated by SDS polyacrylamide gel electrophoresis. c.SDS polyacrylamide gel electrophoresis separates proteins on the basis of charge. d.SDS polyacrylamide gel electrophoresis separates proteins on the basis of size. All of the statements about the tertiary structure of the enzyme triose phosphate isomerase are correct EXCEPT: b.Its α-helices are in the interior core of the molecular structure. Secondary and higher orders of structure are determined by all EXCEPT: a.hydrophobic interactions. b.ionic bonds. c.van der Waals forces. d.hydrogen bonds. e.peptide bonds. Which of the following statements about the use of mass spectrometry in protein investigation are correct? a.Mass spectrometry is used to rapidly determine the molecular weight of a protein. b.Mass spectrometry is not used to determine the sequence of peptides. c.Mass spectrometry cannot be used to investigate post-translational modifications d.Mass spectrometry involves separating ionic fragments on a gel. Which of the following amino acids will exist as a -2 ion at very high pH? a.arginine b.glutamic acid c.isoleucine d.none of these You have a protein that runs at 33 kD on an SDS-PAGE gel but at about 100 kD on mass spectometry. What does this imply about the structure of the native protein? the protein is natively a trimer 84. The amino acid with a side-chain pKa near neutrality and which therefore plays an important role as proton donor and acceptor in many enzyme catalyzed reactions is: a.histidine. b.cysteine. c.proline. d.serine. Given the following chart, what charge will histidine have at a pH of 10? a.+2 b.+1 c.0 d.-1 What is the overall net charge on the peptide lys-lys-ser-glu at pH 7.0? a.+2 b.+1 c.0 d.−1 Electrophoretic separation of leucine from a protein sample would be least effective at which of the following pH values? a.1 b.2 c.3 d.6.7 An electrostatic interaction might occur within a protein between which of the following amino acid pairs at typical physiological pH? a.Ser/Asn b.Asp/Glu c.Arg/Cys d.Lys/Asp Edman degradation will: a .determine the C-terminal amino acid by using a carboxypeptidase. b. cleave the protein into a multitude of smaller peptides. c. compare overlapping sets of peptide fragments. d. determine the N-terminal amino acid. Which level of protein structure is not affected by denaturation? a. primary b. secondary c. tertiary d. quaternary Which of the following statements about Western Blotting is correct? a. The detection of a particular protein by Western Blotting relies on the very specific interaction between the protein and its antibody. b. The detection of a particular protein by Western Blotting relies on labeling the protein with a specific dye. c. The detection of a particular protein by Western Blotting relies on labeling the antibody with a specific dye. d. The detection of a particular protein by Western Blotting relies on the denaturation of the protein. Flexible, disordered segments of proteins are commonly high in the amino acid: a. leu b .lys c .ser d .pro . Electrostatic interactions among amino acid residues on proteins may be damped out by high concentrations of: a .water. b .organic solvents. c. salts. d .heat. Which of the following statements about isoelectric focusing is correct? a.Proteins separated by isoelectric focusing can be tested for biological activity. b Proteins separated by isoelectric focusing cannot be tested for biological activity. c.The separation of proteins by isoelectric focusing is only based on charge. d.The separation of proteins by isoelectric focusing is only based on size. Which of the following IS NOT a characteristic of globular proteins? a.Insoluble in water. b.Roughly spherical. c.Folded so that the hydrophobic amino acids are in the interior of the molecule. d.Hydrophobic side chains are exposed to the water. 96. Your purification strategy of combinations of chromatography steps gives a protein preparation with a single band on SDS-PAGE. Which of the following would be best for determining the protein concentration (as mg/ml or molarity)? a. Measure a UV absorbance scan and use the absorbance at 280nm with the molar extinction coefficient (predicted from the amino acid sequence) b. Determine amino acid composition after hydrolysis to amino acids c. Colorimetric assay using Bradford or BCA assays d. "Guesstimate" the concentration from the intensity of bands on SDS-PAGE gels Measure a UV absorbance scan and use the absorbance at 280nm with the molar extinction coefficient (predicted from the amino acid sequence Which of the following denaturing agents affects hydrophobic regions? a.detergents b.heat c.heavy metals d.urea . A Ramachandran plot shows: a.the amino acid residues which have the greatest degree of rotational freedom. b.the sterically allowed rotational angles between R groups and α-carbons in a peptide. c.the sterically allowed rotational angles between Cα and the amide nitrogen (Cα−N) as well as between Cα and the amide carbonyl carbon (Cα−CO). d.the sterically allowed rotational angles about the amide nitrogen (NH) and CO. The preponderance of protein sequence information is now derived from: :يتم اآلن اشتقاق رجحان معلومات تسلسل البروتين من a.chemical sequencing (Edman method). b.mass spectrometry. c.mass spectrometry-mass spectrometry. d.translating the nucleotide sequence of genes into codons, and thus amino acid sequence. How many amino acids have an amide side chain? a.1 b.2 c.3 d.4 How many amino acids have basic side chains? a.1 b.2 c.3 d.4 In the majority of α-helixes, each peptide carbonyl is hydrogen bonded to the peptide N−H group ____ residues farther ____ the chain. a.2, down b.4, up c.3, down d.2, up How many amino acids have acidic side chains? a.1 b.2 c.3 d.4 Amino acid side chains capable of forming hydrogen bonds are usually located on the protein ____ and form hydrogen bonds primarily with the ____. a. surface, water solvent b. interior, water solvent c. surface, other amino acid side chains d. interior, other amino acid side chains Which of these spectroscopic methods identifies the secondary structure of a protein? a.circular dichroism b.X-ray crystallography c.NMR d.cryo-EM When will a protein have the lowest solubility? a.when the pH is very low b.when the pH = 7 c.when the pH is very high d.at its isoelectric point Nickel-NTA (Ni-NTA) chromatography is a popular affinity chromatography method for the purification of histidine-tagged proteins. However, SDS-PAGE of the eluted protein can show bands in addition to your target protein. How might you improve this purification step? a.Add a low concentration of imidazole to the binding buffer b.Gradient elution with increasing imidazole concentration on a chromatography machine c.Include protease inhibitors during purification d.All of the above Which of these chromatography types are suitable as a "capture" step in the purification of non-tagged proteins? a.IEX and HIC b.SEC c.Dialysis d.Ammonium sulphate precipitation ____ between tightly packed amino acid side chains in the interior of the protein are a major contribution to protein structure. a.Hydrogen bonds b.Electrostatic interactions c.Covalent ester bonds d.Van der Waals interactions Which of the following amino acids is not optically active? a.tyrosine b.glycine c.tryptophan d.phenylalanine The outward face of a(n) ____ consists mainly of polar and charged residues, whereas the inner face contains mostly nonpolar, hydrophobic residues. a.β-sheet b.configuration c.β-turn d.amphiphilic helix Which of the following pairs of amino acids has hydrophobic interactions? a.alanine and glutamic acid b.aspartic acid and lysine c.leucine and phenylalanine d.serine and tyrosine Which of the following IS NOT a characteristic of a protein's overall conformation Achieved by breaking and reforming covalent bonds 115. If the pI of a peptide is 4.6, ____ might be present while ____ would probably be absent. a .Glu / Lys b .His / Ser c. Arg / His d. Asp / Gly Hemoglobin is an example of a(n): a.enzyme. b.regulatory protein. c.transport protein. d.storage protein. Which of the following amino acids will exist as a -2 ion at very high pH? a.arginine b.glutamic acid c.isoleucine d.none of these Which of the following is not a commonly used technique for protein isolation and purification? a.gas-liquid chromatography. b.ion exchange chromatography. c.electrophoresis. d.solubility ("salting in" and "salting out"). Collagen is an example of a(n): a.enzyme. b.regulatory protein. c.transport protein. d.structural protein. What would hinder the formation of alpha-helix in proteins? a. glycine residues b. positive charges in the middle c. nonpolar R groups in the helix d. sequences of negative or positive charges in the middle of the helix . Which of the following properties of a protein is least likely to be affected by changes in pH? a.primary structure b.secondary structure c.tertiary structure d.charge Which of the following statements about two-dimensional gel electrophoresis are correct? Please select all that apply. a. SDS gel electrophoresis and isoelectric focusing both require a pH gradient. b. SDS gel electrophoresis and isoelectric focusing both separate native proteins. c. SDS gel electrophoresis and isoelectric focusing both make use of the migration of proteins in an electric field. d. SDS gel electrophoresis and isoelectric focusing together make up the process of threedimensional gel electrophoresis. Why should the core of most globular and membrane proteins consist almost entirely of α-helix and β-sheets a. Hydrogen bonded structures must be kept away from water solvent. b. Highly polar N−H and C=O moieties of the peptide backbone must be neutralized in the hydrophobic core of the protein. c. Hydrogen bonding only occurs in the core of proteins. d. Trapped water stabilizes the helix and sheet structures. Which of the following side chains are hydrophilic? a.acidic b.basic c.polar but neutral d.all of these Which of the following mutations would probably be least likely to impact the function of the protein? a.Lys to Ser b.Ala to Asp c.His to Pro d.Val to Ile A β-barrel would most likely be composed of ____. a.parallel β-sheets connected by regions of α-helix b.parallel β-sheets connected by β-turns. c.parallel β-sheets connected by regions of random coil. d both a and c are correct. . What is a residue? a.An amino acid which has been severed from a protein. b. Any individual amino acid in a protein or polypeptide. c.The portion of a protein which remains after one amino acid has been removed from the Cterminus. d.The portion of a protein which remains after one amino acid has been removed from the Nterminus. 130. In which of the following do both amino acids have a nonpolar side chain? a.cysteine and glycine b.cysteine and methionine c.histidine and methionine d.phenylalanine and tryptophan The secondary structure of a protein depends on which of the following types of bonds/interactions a.hydrogen bonds b.covalent bonds c.disulfide bonds d.hydrophobic interactions When will a protein have a net positive charge? a.always b.never c.when the pH is above its isoelectric point d.when the pH is below its isoelectric point What does SDS do in an SDS-PAGE gel? a.denatures the protein b.breaks disulfide bonds c.coats the protein in positive charge d.breaks up the protein into amino acids What properties of a protein does hydrophobic interaction chromatography exploit for purification? a. Charged amino acids b. Hydrophobic amino acids on the protein surface c. Molecular weight d. Enzyme activity When the peptide (AEFFLAMEP) forms an α-helix, which amino acid residue would be closest to being in the same position on the same face of the helix as is the initial alanine residue? a.F(3) b.A(6) c.E(8) d.P(9) e.L(5) In which of the following do both amino acids contain a sulfur atom? a.cysteine and glycine b.cysteine and methionine c.histidine and methionine d.phenylalanine and tryptophan Glutamic acid has pKa values of 2.2, 4.3, and 9.7. Calculate the isoelectric point for glutamic acid. a.3.25 b.4.3 c.5.4 d.7.0 140. What is the approximate pI of most amino acids? a.3 b.6 c.7 d.10 When the amino acids R and and D are found in close proximity to each other on protein, what type of interaction is possible? a.electrostatic b.disulfide bond c.hydrophobic interaction d.convalent polar Which of the following pairs of amino acids can form hydrogen bonds? a.alanine and glutamic acid b.aspartic acid and lysine c.leucine and phenylalanine d.serine and tyrosine You find that your protein sample loses activity during storage. What can you do about this? a.Add an additional purification step b.Use a protease inhibitor during purification steps c.Perform each step as quickly as possible, in a cold-room d.All of the above all of the above ] Which of the following is glycylalanine Which of the following amino acids will exist as a + 2 ion at very low pH? a. arginine b. glutamic acid c. isoleucine d. none of these