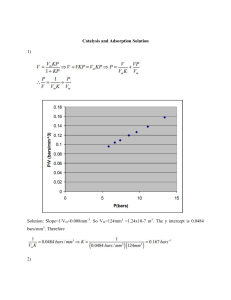
ADSORPTION LESSON FIVE WRU07432 Adsorption Mechanism of adsorption Types of adsorption Applications of adsorption Adsorption isotherms Different types of isotherms Applications of isotherms Summary References Adsorption The term adsorption was introduced by Kayser in 1881. The phenomenon of higher concentration of any species of solid, liquid or gas at the surface than in the bulk of a material, is known as adsorption. Some Important Terms Adsorbent-The solid that takes up a gas, vapour or a solute from a solution, e.g. silica gel, charcoal, clay, etc. Desorption-The process of removal of adsorbed substances from the surface on which it is absorbed. Adsorbate-The gas, vapour or the solute, which is held to surface of the solid. e.g. poisonous gases Some commonly used adsorbents silica Activated charcoal Alumina Bentonite clay Zeolites Bagasse Examples of adsorption Accumulation of dust on our skin when we travel in regions of heavy traffic Adsorption of dyes like methylene blue by charcoal Protection from poisonous gases Solution of raw sugar become colorless when passed over bed of animal charcoal. Air becomes dry in the presence of silica gel. Difference between adsorption and absorption Absorption It is the phenomenon in which the particles of gas or liquid uniformly distributed throughout. Bulk phenomenon Occurs at uniform rate Anhydrous CaCl2 absorbs water. Adsorption It is the phenomenon in which the higher conc. of particles of gas or liquid on surface than in bulk. Surface phenomenon Rapid in beginning but slowly decreases. Silica gel absorbs water vapour. continued…… Absorption Adsorption Mechanism of Adsorption Adsorption is surface phenomenon. It arises because of unbalanced forces on the surface of solids and liquids. The surface is under tension due to unbalanced forces. The surface of the solid or liquid tends to satisfy their residual forces by attracting and retaining the molecules of other species when brought in contact with them. Continued….. Exothermic process ∆𝐺 = ∆𝐻 − 𝑇∆𝑆 Spontaneous Types of Adsorption Physisorption Chemisorption continued…… PROPERTIES PHYSISORPTION CHEMISORPTION Bonding Weak, long range forces, van der Waals interactions. Strong, short range forces, Chemical bonding involving orbital overlap and charge transfer. Enthalpy 20-40 kJ mol-1 80-240 kJ mol-1 Saturation Multi-layer Mono-layer Nature reversible irreversible Effect of temperature Occurs at low temperature and decreases with increase in temperature. Occurs at high temperature and increases with increase in temperature. Effect of pressure Increases with increase in pressure of adsorbate. Decreases with increase of pressure Activation energy Not needed High activation energy is needed. Surface specificity No Yes Factors affecting adsorption Nature of gas - easily liquefiable gases easily gets adsorbed. Nature of adsorbent- More the surface area per unit mass of the adsorbent, more will be the adsorption Temperature- adsorption increases as temperature decreases. Pressure- magnitude of adsorption increases with increase in pressure. APPLICATIONS OF ADSORPTION Gas masks Adsorption indicators Chromatographic separation Removal of coloring matter Heterogeneous catalysis Controlling humidity Curing diseases Froth flotation process Production of high vacuum Purification Adsorption equilibria The adsorbent and adsorbate are contacted long enough, an equilibrium will be established between the amount of adsorbate adsorbed and the amount of adsorbate in solution. Isotherms: m, Pdiagrams at constant temperature Adsorption isotherm The process of adsorption is studied through graphs know as adsorption isotherms. Graph between the amounts of adsorbate (x) adsorbed on the surface of adsorbent (m) and pressure (P) at constant temperature. Continued… In adsorption, adsorbate gets adsorbed on adsorbent. The direction of equilibrium would shift in that direction where the tension can be relieved. In excess of pressure to the equilibrium system, the equilibrium will shift in the direction where the number of molecules decreases. Number of molecules decreases in forward direction. Therefore, forward direction of equilibrium will be favored. Basic Adsorption isotherm Limited numbers of vacancies on the surface of the adsorbent. After saturation pressure Ps, adsorption does not occur anymore. At high pressure a stage is reached when all the sites are occupied and further increase in pressure does not cause any difference in adsorption process. At high pressure, Adsorption is independent of pressure. Types of isotherms Temkin isotherm Langmuir isotherm Dubinin– Radushkevich isotherm Freundlich isotherm BET isotherm Langmuir Adsorption isotherm History In 1916, Dr. Irving Langmuir presented this model. Langmuir was awarded the Nobel Prize in 1932 Adsorption can be physisorption or chemisorption Estimate the adsorption capacity of adsorbent used Basic assumptions of Langmuir Model The surface is homogeneous All sites are equivalent Mono-layer adsorption only No interactions between adsorbate molecules on adjacent sites Heat of adsorption is constant and equivalent for all sites Adsorbate molecules have tendency to get adsorb and desorb from surface Langmuir Adsorption isotherm The linear form of the Langmuir isotherm bqmax qe = 1 + bCe Where qe= Quantity of adsorbate adsorbed per unit weight of adsorbent at equilibrium (mgg-1) Ce = Concentration of adsorbate at equilibrium in solution after adsorption (mgl-1) qmax = Maximum adsorption capacity(mgg-1) b = Langmuir adsorption equilibrium constant (lmg-1) Continued…. Plot of original equation Continued…. The rearranged form of the Langmuir isotherm 1 1 1 = + qe Ce bqmax qmax Where qe= Quantity of adsorbate adsorbed per unit weight of adsorbent at equilibrium (mgg-1) Ce = Concentration of adsorbate at equilibrium in solution after adsorption (mgl-1) qmax = Maximum adsorption capacity(mgg-1) b = Langmuir adsorption equilibrium constant (lmg-1) Continued…. 1/qe Isotherm is plotted between 1/qe versus 1/Ce and the slope of the graph gives the value of qmax and intercept gives the value of b. 0.06 0.05 0.04 0.03 0.02 0.01 0 y = 0.2597x + 0.0018 R² = 0.9637 0 0.05 0.1 0.15 1/Ce 0.2 0.25 Limitations of Langmuir isotherm Holds only at low pressure. Multilayer adsorption is possible. Ignores adsorbate-adsorbate interactions. Fails to account for the surface roughness of the adsorbent. Saturation value of adsorption depends upon temperature. Freundlich Adsorption Isotherm 1909, Dr. Herbert Freundlich Estimate the sorption intensity of the adsorbent towards the adsorbate. This isotherm is an empirical expression that accounts for surface heterogeneity. Assumptions of Freundlich Isotherm Surface roughness Adsorbateadsorbate interactions Inhomogeneity Freundlich Adsorption Isotherm The linear equation x/m = k.P1/n (n > 1) Or x/m = k.C1/n (n > 1) where x is the mass of the gas adsorbed on mass m of the adsorbent P is pressure, C is equilibrium concentration of adsorbate in solution, k and n are constants. Continued… The linear form of Freundlich isotherm is 1 logqe = log K f + logPe n or 1 logqe = log K f + logCe n Where qe = Extent of adsorbate adsorbed per unit weight of adsorbent at equilibrium (mgg-1) Pe = Equilibrium pressure of adsorbent in solution after adsorption Ce = Equilibrium concentration of adsorbent in solution after adsorption (mgl-1) Kf = Freundlich constant indicating adsorption capacity n = Empirical constant Continued…. Kf approximately indicates adsorption capacity 1/n is a function of the strength of adsorption n = 1, the boundary between the two phases is independent of the concentration. 1/n below 1 indicates a normal adsorption. n varies with the heterogeneity of the adsorbent and range for favorable adsorption is of 1-10. 1/n = 0, x/m = constant, the adsorption is independent of pressure. 1/n = 1, x/m = k P, i.e. x/m ∝ P, the adsorption varies directly with pressure. Continued… The parameters of the Freundlich isotherm can be determined by plotting logqe versus logCe. 5 log qe 4.5 4 3.5 3 y = 0.9452x + 1.3982 R² = 0.9637 2.5 2 1 2 log Ce 3 4 Limitations of Freundlich Isotherm Freundlich equation is purely empirical and has no theoretical basis. The equation is valid only upto a certain pressure and invalid at higher pressure. The constants K and n vary with temperature. Frendilich’s adsorption isotherm fails at high concentration of the adsorbate. Differences between Freundlich and Langmuir adsorption isotherms FRENDLICH ADSORPTION ISOTHERM • Tells about the quantity of gas adsorbed by unit mass of solid adsorbent with pressure. • Represented by formula x/m = KP1/n • Heterogeneity of adsorption site. • No information about adsorption and desorption. LANGMUIR ADSORPTION ISOTHERM • Tells about the number of active site undergoing adsorption and pressure. • Represented by formula θ = 𝐾𝑃ൗ1+ KP • Homogeneity of adsorption site. • Based on adsorption and desorption equilibrium. APPLICATIONS OF ADSORPTION ISOTHERMS Spontaneity Exothermicity Percentage removal of adsorbate Langmuir parameters- maximum adsorbent uptake and affinity between adsorbent and adsorbate CONTINUED……. The extent of adsorption increases with the increase of surface area per unit mass of adsorbent at given temperature and pressure. Types of adsorption physisorption and chemisorption Factors affecting adsorption temperature, pressure, nature of adsorbent and adsorbate Adsorption isotherm- It is the graph between the amounts of adsorbate (x) adsorbed on the surface of adsorbent (m) and pressure (P) at constant temperature. Types of adsorption isotherms-Langmuir isotherm, Freundlich isotherm, BET isotherm, DR isotherm, Temkin isotherm ACTIVATED CARBON Water contains dissolved organic matter that cannot be removed with floc formation, floc removal or sand filtration. These dissolved organic compounds are: ➢ Odor, taste and color-producing compounds ➢ Organic micropollutants (pesticides, hydrocarbon compounds) Activated carbon adsorbs (part of) the organic matter and is mainly used to treat drinking water produced from surface water. ACTIVATED CARBON Activated carbon is a substance with a high carbon concentration ➢ The activated carbon filters of the drinking water production plant have the objective ➢ ❖ to improve the taste of the water, ❖ to reduce the regrowth of bacteria in the piped network, and ❖ to remove toxic substances from the water. ➢ The carbon filters are placed after the floc formation, sedimentation and rapid sand filtration to avoid rapid clogging ACTIVATED CARBON Under high temperatures this material becomes carbonated, meaning that the carbon partly transforms into carbon monoxide and water. ➢ This is how the carbon gets its open structure ➢ The internal surface area of the activated carbon is several times larger than the external surface area ➢ ACTIVATED CARBON macro pores > 25 nm meso pores 1 - 25 nm micro pores < 1 nm The open structure of activated carbon ACTIVATED CARBON Depending on the size of activated carbon, they are divided into Granular ➢ Activated powder ➢ Students should discuss the problems associated with the use of Powder activated carbon (PAC) and granular activated carbon (GAC) during adsorption TUTORIAL The following data were gathered in a laboratory study of the adsorption of trichlorophenol (MW= 197.5 g/mol) on a commercial powdered activated carbon for the following conditions: Initial concentration of solute: 470 mg/L; Volume of water: 500 mL Fit these data to the Langmuir and Freundlich isotherms, and determine the constants of both the models. Find the maximum adsorption capacity