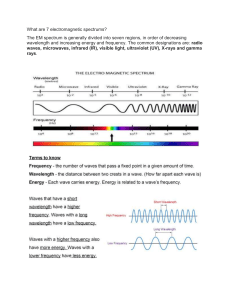
Name ____________________________________________________________________ Date ______________________________________ Period ___________ The following questions pertain to the electromagnetic spectrum and the duality of light (wave and particle). Use your notes to answer each question. 1. The relationship between frequency and wavelength is ( direct / inverse ). 2. The relationship between frequency and energy is ( direct / inverse ). 3. The relationship between wavelength and energy is ( direct / inverse ). 4. Two light waves travel at the same speed. Wave A has four times the frequency of wave B. How does the wavelength of wave A compare with the wavelength of wave B? _____________________________________________ 5. Photon A has twice the frequency of photon B. How do the energies of the two photons compare? _________________________________________________________________________________________________________________________ 6. A Photon has a frequency of 2 x 1014 Hz. If the energy of the photon triples, what is the new frequency? ________________________________________________________________________________________________________________________ 7. What is the effect on the frequency if the wavelength is halved? _______________________________________________ 8. What is the effect on the energy if the frequency is increased by a factor of four? ____________________________ 9. Of the types of radiation below, place them in order of lowest energy to highest. a. UV (ultraviolet) b. Microwaves ________ ________ ________ ________ ________ c. FM, TV lowest highest d. Radio e. Gamma 10. Of the types of radiation below, place them in order of shortest wavelength to longest. a. UV (ultraviolet) b. Microwaves ________ ________ ________ ________ ________ c. FM, TV shortest longest d. Radio e. Gamma 11. What is a photon? ________________________________________________________________________________________ 12. Each photon carries ___________________________ of energy. 13. What is the speed of ALL light? ________________________________________________________________