Intermolecular Forces Worksheet: Identify Strongest Force
advertisement

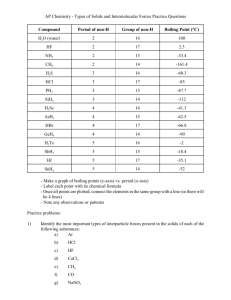
Types of Intermolecular Forces W 317 What is the strongest intermolecular force present for each of the following molecules? 1) hydrogen (H2)_____________________________________________ 2) carbon monoxide (CO) ______________________________________ 3) silicon tetrafluoride (SiF4)_____________________________________ 4) nitrogen tribromide (NBr3) _____________________________________ 5) water (H2O)________________________________________________ 6) acetone (CH2O) ____________________________________________ 7) methane (CH4) _____________________________________________ 8) benzene (C6H6)_____________________________________________ 9) ammonia (NH3) _____________________________________________ 10) methanol (CH3OH) __________________________________________ Everett Community College Tutoring Center Student Support Services Program What is the strongest intermolecular force present for each of the following molecules? 1) hydrogen (H2) London dispersion forces 2) carbon monoxide (CO) London dispersion forces 3) silicon tetrafluoride (SiF4) London dispersion forces 4) nitrogen tribromide (NBr3) dipole-dipole forces 5) water (H2O) hydrogen bonding 6) acetone (CH2O) dipole-dipole forces 7) methane (CH4) London dispersion forces 8) benzene (C6H6) London dispersion forces 9) ammonia (NH3) hydrogen bonding 10) methanol (CH3OH) hydrogen bonding