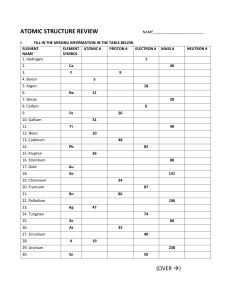
MYP Chemistry Unit Atomic Theory and Structure Atomic theory and structure unit overview This unit involves the following chemistry concepts. • • • • • • • • Atomic theory Organization of the periodic table Atomic structure Bohr models and Lewis dot diagrams Isotopes and ions Quantum mechanical model Electron configuration and orbital-filling diagrams Famous scientists who contributed to atomic theory Unit rationale This unit is designed to investigate the development of atomic theory throughout time. A main focus is atomic structure and the quantum model. Notes are provided for electron configuration and orbital filling in case those concepts are a part of your curriculum. If not, adjust the unit as appropriate. Suggested lesson plan Lesson 1: The nature of science discussion activity Objective: The student will explore the philosophical and theoretical nature of science and answer questions based on a conversation in The God Particle by Leon Lederman. • Essential knowledge Plan 1. Distribute 1.1 The God Particle Discussion Constant re-evaluation in the light of new Questions before students begin to read. data is essential to ensure scientific 2. 1.2 The God Particle extract. Allow 15 knowledge remains current. In this fashion, minutes for students to read and find all forms of scientific knowledge remain answers. flexible and may be revised as new data 3. Conduct discussion activity using one of and new ways of looking at existing data the formats from 1.3 God Particle become available. Discussion Instructions for Teachers. 4. Reflection activity for homework: How do the questions we ask impact on the answers we receive? 1 MYP Chemistry Unit Atomic Theory and Structure Lesson 2: The periodic table Objective: The student will discover that the periodic table is ordered by groups and periods, along with family names, and discuss those who contributed to this design. Essential knowledge • Horizontal rows called periods have predictable properties based on an increasing number of electrons in the outer orbitals. • Vertical columns called groups have similar properties because of their similar valence electron configurations. • The names of groups and periods on the periodic chart are alkali metals, alkaline earth metals, transition metals, halogens, noble gases and metalloids. • Periods and groups are named by numbering columns and rows. Plan 1. Distribute 2.1 Periodic Table (make this two-sided). The first side of the periodic table will be used for colouring family names. Students should also label and number groups and periods. Distribute 2.2 Atomic Theory & Structure Guided Notes. (Notes will be used for the duration of the unit. 2. Complete section I along with 2.3 Organization of the Periodic Table Presentation. 3. Homework: Students will colour-code their periodic tables by family names on one side only. Lesson 3: Atomic theory Objective: The student will discover how the model of the atom has changed over time. The student will also memorize the key scientists who contributed to atomic theory, along with their experiments and discoveries. The student will evaluate the results of each scientist’s experiment. Essential knowledge • Discoveries and insights related to the atom’s structure have changed the model of the atom over time. 1. 2. • The periodic table is arranged in order of increasing atomic numbers. • Major insights regarding the atomic model of the atom and principal scientists include: - particles—Democritus - first atomic theory of matter—John Dalton - discovery of the electron—JJ Thompson - discovery of the nucleus—Ernest Rutherford 3. 4. Plan Distribute 3.1 Element Groups Worksheet as a warm-up. Review the answers. Teach Section II of 2.2 Atomic Theory & Structure Notes with 3.2 Atomic Theory Presentation Slides 1–10. Videos are linked and/or embedded. Headline reflection: All students must write a headline that summarizes the lesson and how it applies to the unit question. Homework: Distribute 3.3 Atomic Theory & Structure Homework Packet. Students must complete section A. 2 MYP Chemistry Unit Atomic Theory and Structure - discovery of charge of electron— Robert Millikan - planetary model of atom—Niels Bohr - periodic table by atomic mass— Demitry Mendeleev - periodic table by atomic number— Henry Moseley - quantum nature of energy—Max Planck - uncertainty principle—Werner Heisenberg - wave theory—Louis de Broglie. Lesson 4: Atomic structure and symbols Objective: The student will discover the subatomic particles of the atom and how each of those particles affects the identity of the element. Key terms include the following: atom, element, proton, neutron, electron, atomic number, atomic mass, mass number, valence electron, atomic mass unit. Essential knowledge • The atomic number of an element is the same as the number of protons. • In a neutral atom, the number of electrons is the same as the number of protons. • All atoms of an element have the same number of protons. • The atomic mass for each element is the weighted average of that element’s naturally occurring isotopes. • Electrons have little mass and a negative (–) charge. They are located in electron clouds or probability clouds outside the nucleus. • Protons have a positive (+) charge. Neutrons have no charge. Protons and neutrons are located in the nucleus of the atom and comprise most of its mass. Plan 1. Distribute 4.1 Atomic Theory Timeline Worksheet as a warm-up. Review. 2. Discuss 2.2 Atomic Theory & Structure Guided Notes Section III using 4.2 Atomic Structure Presentation for help. 3. Complete 4.3 Atomic Structure Investigation. 5. Homework: Distribute 3.3 Atomic Theory & Structure Homework Packet. Students must complete Section B. 3 MYP Chemistry Unit Atomic Theory and Structure Lesson 5: Modelling for Bohr and Lewis Objective: The student will create Bohr models and Lewis dot structures for elements. Essential knowledge • Draw Bohr models and Lewis dot diagrams for atoms. Plan 1. Distribute 5.1 Atomic Structure Practice as a warm-up. http://www.corvallis.k12.mt.us/high/staff/brockh/Chemistry/Ch5/CRM050 6.pdf 2. Give instruction on Bohr models and Lewis structures using 5.2 Teacher Guide for Bohr Models. 3. Complete 5.3 Bohr Model & Lewis Structure Worksheet as class work. 4. Homework: 3.3 Atomic Theory & Structure Homework Packet, sections C and D (only the questions that apply). Lesson 6: Changing an atom’s structure Objective: The student will discover what happens when the number of protons, neutrons, or electrons is changed in an atom. The student will calculate average atomic mass using abundance and mass numbers. Essential knowledge • The atomic mass for each element is the weighted average of that element’s naturally occurring isotopes. 1. • An isotope is an atom that has the same number of protons as another atom of the same element but has a different number of neutrons. Some isotopes are radioactive; many are not. 3. • Atoms can gain, lose or share electrons within the outer energy level. • Loss of electrons from neutral atoms results in the formation of an ion with a positive charge (cation). 2. 4. 5. Plan Distribute and assign 6.1 Cloze Reading Passage. Complete 2.2 Atomic Theory & Structure Guided Notes section III. Distribute 6.2 Solving and Naming Isotopes Worksheet. Distribute 6.3 Another Isotope Worksheet and complete “Average Atomic Mass Problems” section. Leave part A blank for lesson in class. Homework: Complete all questions up to and including section D of 3.3 Atomic Theory & Structure Homework Packet. Also complete 6.4 Practice with Ions & Isotopes. • Gain of electrons by a neutral atom results in the formation of an ion with a negative charge (anion). 4 MYP Chemistry Unit Atomic Theory and Structure Lesson 7: The quantum model part 1—Electron configuration1 Objective: The student will learn how elements fit into shells, subshells, and orbitals on the periodic table. The student will write electron configurations in both longhand and shorthand. The student will discover contributions by Planck, Heisenberg and de Broglie. Essential knowledge • Discoveries and insights related to the atom’s structure have changed the model of the atom over time. • The modern atomic theory model is called the quantum mechanical model. • Vertical columns called “groups” have similar properties because of their similar valence electron configurations. • Electron configuration is the arrangement of electrons around the nucleus of an atom based on their energy level. • It is possible to use an element’s electron configuration to determine the number of valence electrons and possible oxidation numbers. 1. 2. 3. 4. 5. Plan Require students to complete part A of 6.3 Isotopes Worksheet. Show 6.1 Quantum Mechanics Video to introduce the topic. Answer 2.2 Atomic Theory & Structure Guided Notes sections V and VIa. Distribute/teach from 7.2 Learning Electron Configuration Presentation. Students should work alongside the presentation. Another presentation is offered to teach students how to actually write electron configurations (7.3 Writing Electron Configurations). Use this to assist or teach it without this aid. Homework: Complete section D of 3.3 Atomic Theory & Structure Homework Packet and 7.4 Yet Another Atomic Structure Worksheet. Lesson 8: The quantum model part 2—Orbital-filling diagrams Objective: The student will create orbital-filling diagrams showing orbitals and correct filling of electrons using the following rules: Aufbau, Pauli’s exclusion principle and Hund’s rule. Essential knowledge • Electrons are added one at a time to the lowest energy levels first (Aufbau principle). • An orbital can hold a maximum of two electrons (Pauli exclusion principle). Plan 1. Review homework from lesson 7 as a warm-up. 2. Show 8.1 Quantum Mechanics Video 2 to introduce orbital filling. 3. Discuss 2.2 Atomic Theory & Structure Guided Notes section VIb. 1 I have recently discovered that it is just as easy to teach orbital filling first, then electron configuration. If your students are visual learners, this method may work better. Also, they can be taught together in the same class time frame, using this method. I provided students with a blank periodic table, several sticky notes, and four different colors to outline their blocks. The students used sticky notes for important points like the number of orbitals, number of electrons in each subshell, and key rules. 5 MYP Chemistry Unit Atomic Theory and Structure • Electrons occupy equal-energy orbitals so that a maximum number of unpaired electrons results (Hund’s rule). • Energy levels are designated 1–7. Orbitals are designated s, p, d, and f according to their shapes. • Orbitals s, p, d and f relate to the regions of the periodic table. 4. Teach orbital filling, using 8.2 OrbitalFilling Diagrams Presentation to help if you wish. Suggestion: Use the same problems you just reviewed from homework as classwork. 5. Thought-provoker activity: Show 8.3 Double-slit Experiment Video. Allow students to ask questions and lead discussion. You may have to show it twice for students to grasp what’s actually taking place. 6. Homework: Complete 3.3 Atomic Theory & Structure Homework Packet. If necessary, discuss and distribute flame test lab instructions. Lesson 9: The fingerprint of atoms and flame test lab Objective: The student will discover the chemical properties of elements through color changes of light. The student will perform a flame test lab. • • • Essential knowledge The electromagnetic spectrum is the continuous range of light radiation. An atomic absorption spectrum shows the frequencies at which atoms absorb energy. An atomic emission spectrum is the opposite of the absorption spectrum, and shows the frequency at which energy is released. The atomic emission spectrum of an element is like a fingerprint, meaning it is unique to that element. Plan 1. Distribute 9.1 Quantum Model Warm-Up. 2. Conduct 9.2 Flame Test Lab. Lesson 10: Objective test This test may serve as a pre-test for the actual assessment test. Students should receive immediate feedback. Lesson 11: Test assessment .Isotopes Worksheet from Loundoun County Schools 6