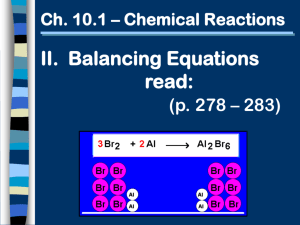
WORKSHEET CHEMISTRY Date: Nov 11, 2021 Points: ____ / ____ Name: ___________________________________ Points: 1. Question: Write sentences that completely describe the chemical reactions listed below. → a) 2KOH(aq) + H2SO4(aq) b) FeO(s) + C(s) [heat] c) Cl2 + 2NaI → 2H2O(l) + K2SO4(aq) + CO(g) 2NaCl + I2 d) P4O10(s) + 6H2O(l) e) C7H16 + 11O2 → Fe(s) __ / __ → → 4H3PO4(aq) 7CO2 + 8H2O 2. Question: Rewrite the following word equations as balanced chemical reactions. a) Chlorine gas reacts with a solution of potassium iodide, resulting in the production of elemental iodine and a solution of potassium chloride. b) Solid silver oxide can be heated to give silver and oxygen gas. c) When metallic iron reacts with hydrochloric acid, bubbles of hydrogen gas and an aqueous solution of iron (III) chloride are formed. d) Heating tin (IV) hydroxide gives tin (IV) oxide and water. e) Magnesium chloride decomposes into its constituents when a strong electrical current is passed through it. __ / __ 3. Question: How many types of reactions do you know? List the most common ones and write a balanced chemical equation as an example for each. __ / __ __ / __ 4. Question: Balance the following chemical reactions. Try to discern the type of reaction. → KOH → a) Ba(OH)2 + NO3 Ba(NO3)2 + H2O b) H2C2O4 + K2C2O4 + H2O c) Pb(NO3)2 + K2CrO4 d) Al + CuSO4 → e) Zn(OH)2 + H3PO4 → PbCrO4 + KNO3 Al2(SO4)3 + Cu → Zn3(PO4)2 + H2O __ / __