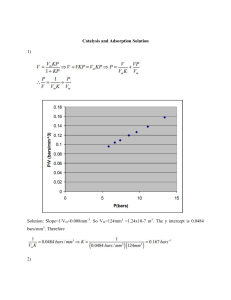
Instruction Manual ADSORPTION IN PACKED BED Adsorption In Packed Bed 1 ADSORPTION IN PACKED BED 1. AIM: Determination of adsorption isotherm of acetic acid on activated charcoal. To determine the adsorption constant (k) and the maximal adsorbed substance amount of acetic acid per gram of charcoal (Amax) of Langmuir isotherm. 2. INTRODUCTION: Adsorption is a process that occurs when a gas or liquid solute accumulates on the surface of a solid or a liquid (adsorbent), forming a molecular or atomic film (the adsorb-ate). It is different from absorption, in which a substance diffuses into a liquid or solid to form a solution. The term sorption encompasses both processes, while desorption is the reverse process. 3. THEORY: Adsorption is operative in most natural physical, biological, and chemical systems, and is widely used in industrial applications such as activated charcoal, synthetic resins and water purification. Similar to surface tension, adsorption is a consequence of surface energy. In a bulk material, all the bonding requirements (be they ionic, covalent or metallic) of the constituent atoms of the material are filled. But atoms on the (clean) surface experience a bond deficiency, because they are not wholly surrounded by other atoms. Thus it is energetically favorable for them to bond with whatever happens to be available. The exact nature of the bonding depends on the details of the species involved, but the adsorbed material is generally classified as exhibiting physisorption or chemisorptions. Physisorption or physical adsorption is a type of adsorption in which the adsorbate adheres to the surface only through Van der Waals (weak intermolecular) interactions, which are also responsible for the non-ideal behavior of real gases. Chemisorptions is a type of adsorption whereby a molecule adheres to a surface through the formation of a chemical bond, as opposed to the Van der Waals forces which cause physisorption. Adsorption is usually described through isotherms, that is, functions which connect the amount of adsorbate on the adsorbent, with its pressure (if gas) or concentration (if liquid). One can find in literature several models describing process of adsorption, namely Freundlich isotherm, Langmuir isotherm, BET isotherm, etc. We will deal with Langmuir isotherm in more details. Adsorption In Packed Bed 2 Langmuir isotherm In 1916, Irving Langmuir published an isotherm for gases adsorbed on solids, which retained his name. It is an empirical isotherm derived from a proposed kinetic mechanism.It is based on four hypotheses: 1. The surface of the adsorbent is uniform, that is, all the adsorption sites are equal. 2. Adsorbed molecules do not interact. 3. All adsorption occurs through the same mechanism. 4. At the maximum adsorption, only a monolayer is formed: molecules of adsorption do not deposit on other, already adsorbed, molecules of adsorb-ate, only on the free surface of the adsorbent. For liquids (adsorb-ate) adsorbed on solids (adsorbent), the Langmuir isotherm (Fig. 1) can be expressed by: Where m is the substance amount of adsorb-ate adsorbed per gram (or kg) of the unit of m is -1 mol.g .resp. mol. kg-1 Amax is the maximal substance amount per gram (or kg) of the adsorbent. The unit of Amax is mol.g-1, resp. mol.kg -1 k is the adsorption constant (mol-1.dm3), c is the concentration of adsorbate in liquid (mol.dm-3). In practice, activated carbon is used as an adsorbent for the adsorption of mainly organic compounds along with some larger molecular weight inorganic compounds such as iodine and mercury. Activated Carbon - Activated carbon can be manufactured from carbonaceous material, including coal (bituminous, sub-bituminous, and lignite), peat, wood, or nutshells (i.e., coconut).The manufacturing process consists of two phases, carbonization and activation. The carbonization process includes drying and then heating to separate by-products, including tars and other hydrocarbons, from the raw material, as well as to Adsorption In Packed Bed 3 drive off any gases generated. The carbonization process is completed by heating the material at 400–600°C in an oxygen-deficient atmosphere that cannot support combustion. Granular Activated Carbon (GAC).GAC can be either in the granular form or extruded. GAC is designated by sizes such as 8 × 20, 20 × 40, or 8 × 30 for liquid phase applications and 4 × 6, 4 × 8 or 4 × 10 for vapor phase applications. A 20 × 40 carbon is made of particles that will pass through a U.S. Standard Mesh Size No. 20 sieve (0.84 mm) (generally specified as >85% passing) but be retained on a U.S. Standard Mesh Size No. 40 sieve (0.42 mm) (generally specified as >95% retained). AWWA (1992) B604 uses the 50-mesh sieve (0.297 mm) as the minimum GAC size. The most popular aqueous phase carbons are the 12 × 40 and 8 × 30 sizes because they have a good balance of size, surface area, and head loss characteristics. The 8 × 30 size is the most commonly used for most applications. For all three variations of the name "activated" is sometimes substituted by "active." By any name, it is a material with an exceptionally high surface area. Just one gram of activated carbon has a surface approximately 500 m² (for comparison, a tennis court is about 260 m²). The three main physical carbon types are granular, powder and extruded (pellet). All three types of activated carbon can have properties tailored to the application. Activated carbon is frequently used in everyday life, in: industry, food production, medicine, pharmacy, military, etc. In pharmacy, activated charcoal is considered to be the most effective single agent available as an emergency decontaminant in gastrointestinal tract. It is used after a person swallows or absorbs almost any toxic drug or chemical. EXPERIMENTAL SET UP The experimental set up consists of a three Glass Columns having different dia and height, filled with activated carbon. A common feed inlet (manifold) is provided at the bottom of each column. Feed is supplied from feed tank by means of pump and rotameter. Flow rate can be varied by rotameter. A bye-pass arrangement is also provided to maintain level in feed tank. Adsorption process can be studied by varying bed height and feed flow rate in each of the three columns turn-wise. The whole set-up is mounted on a powder coated sturdy MS frame. GLASS COLUMNS BOROSILICATE: ID 25mm height 1000mm, ID 40mm height 500mm and ID 50mm and height 300mm, Rotameter Make Eureka 2-20LPH. Pump make Promivac Model PMP-15, Max head 2.5 mtrs, RPM 2800, single phase 220 Volts @ 50 Hz, sump tank capacity 20 ltrs apprx. 4. EXPERIMENTAL PROCEDURE: 1. First prepare the aqueous solution of acetic acid and water. 2. Switch ON the set up and ensure pump discharge by fully opening by pass valve. 3. Set the desired flow rate in Rota meter by partially closing by pass valve. Adsorption In Packed Bed 4 4. Open the feed valve provided at the back of column 1 (a common manifold has been provided for feed routing to all the three columns). 5. Maintain constant flow rate in Rota meter (in case of fluctuation adjust with needle valve) after feed starts to come out from top of column, collect the sample in flask. 6. Add 4-5 drops of phenolphthalein indicator in added in each flask and titrate by standard NaOH solution. 7. Once the endpoint has been reached, read the burette. The volume of the base X i0 (ml) that was required to reach the endpoint. 8. Read the value of the each flask and write the value respectively in table 2. 9. Repeat procedure for different flow rates and bed heights. Table 1 Sample flask No. 1 2 3 4 5 6 Volume V (ml) c i0 Concentration of acetic acid before the adsorption reaction ci Concentration of acetic acid after the adsorption reaction mi Amount of acetic acid adsorbed per gram of charcoal X i0 The volume of the titrant (NaOH) Table 2 Flask No. X i0 c i0 Xi ci mi 1/ci l/mi (mol/dm3) (ml) (mol/dm3) (mmol/g) (dm3 /mol) (g/mmol) (ml) 1 2 3 4 5 Adsorption In Packed Bed 5 6 10. After completing taking readings of Column 1, start feed supply to column NO 2 and close feed valve of column NO 1. 11. Repeat procedure from 4-5 as mentioned above for different flow rates and bed heights. 12. Add 4-5 drops of phenolphthalein indicator in added in each flask and titrate by standard NaOH solution. 13. Once the endpoint has been reached, read the burette. The volume of the base X i0 (ml) that was required to reach the endpoint. 14. Read the value of the each flask and write the value respectively in table 2. Table 3 Sample flask No. 7 8 9 10 11 12 Volume V (ml) c i0 Concentration of acetic acid before the adsorption reaction ci mi Concentration of acetic acid after the adsorption reaction Amount of acetic acid adsorbed per gram of charcoal The volume of the titrant (NaOH) X i0 Table 4 Flask No. X i0 c i0 Xi ci mi 1/ci l/mi (mol/dm3) (ml) (mol/dm3) (mmol/g) (dm3 /mol) (g/mmol) (ml) 7 8 9 10 11 12 Adsorption In Packed Bed 6 15. After completing taking readings of Column 2, start feed supply to column NO 3 and close feed valve of column NO 2. 16. Repeat procedure from 4-5 as mentioned above for different flow rates and bed heights. 17. Add 4-5 drops of phenolphthalein indicator in added in each flask and titrate by standard NaOH solution. 18. Once the endpoint has been reached, read the burette. The volume of the base X i0 (ml) that was required to reach the endpoint. 19. Read the value of the each flask and write the value respectively in table 2. 20. After completing experiment switch off pump and disconnect set-up from mains. 21. Drain the set-up. Table 5 Sample flask No. 7 8 9 10 11 12 Volume V (ml) c i0 Concentration of acetic acid before the adsorption reaction ci Concentration of acetic acid after the adsorption reaction mi Amount of acetic acid adsorbed per gram of charcoal X i0 The volume of the titrant (NaOH) Table 6 Flask No. X i0 (ml) c i0 Xi ci mi 1/ci l/mi (mol/dm3) (ml) (mol/dm3) (mmol/g) (dm3 /mol) (g/mmol) 7 8 9 10 11 12 Adsorption In Packed Bed 7 4. DATA TREATMENT: 1. Determination of the concentration of acetic acid before (ci0) and after (ci) Adsorption: ci0 X i0 cT , V (2) 0 where X i is the volume of the titrant (NaOH), cT is the concentration of the titrant, V is the volume of the analytic (acetic acid according to Tab. 2), i=1-6 is the number of flask. Calculate the concentration of acetic acid after adsorption (ci), using the Eq. 2 and data form Tab. 3 after adsorption. 2. Determination of the substance amount of acetic acid adsorbed per gram of the charcoal m (mol.g-1) in individual flask: mi (ci0 ci )V A , g (3) 0 Where ci , ci are the concentrations of acetic acid before and after adsorption, respectively. VA is the volume of the liquid phase in the mixture charcoal – acetic Acid, g is the mass of the adsorbent – charcoal (in grams), i=1-6 is the number of flask. Eq. 3 supposes that VA is the same for i=1-6, and also the mass of the charcoal (g). Write down the obtained values of m i to the Tab. 3. 3. Determination of k and Amax: The Eq. 1 one can rearrange into a form: 1 1 1 1 , m Amax k c Amax thus (4) 1 1 f should be a straight line. m c 1 1 f , where c is the concentration of acetic acid after m c 1 adsorption. Fit the experimental points with a linear function. The slope represents the value of , and Amax k Use MS Excell to create the dependence the intercept corresponds to 1 Amax Calculate Amax and k from the slope and the intercept. Adsorption In Packed Bed 8 5. UTILITY REQUIRED: 1. Power supply: Single Phase, 220 Volts, @50 Hz, 5 Amp with Earth. 2. Water (for preparing concentration). 3. Drain. Control Panel Comprises of: Standard Make On/Off Switch, Mains Indicator, etc. CHEMICALS REQUIRED:Acetic acid, NaOH, phenolphthalein indicator, activated charcoal granular form (supplied with set-up) 6. PRECAUTIONS & MAINTENANCE INSTRUCTIONS: 1. Do not run the pump at low voltage i.e. less than 180 Volts. 2. Never fully close the Delivery line and By-Pass line Valves simultaneously. 3. Always keep apparatus free from dust. 4. To prevent clogging of moving parts, Run Pump at least once in a fortnight. 5. Always use clean water. 6. If apparatus will not in use for more than one month, drain the apparatus completely. 7. TROUBLE SHOOTING: 1. If pump gets jam, open the back cover of pump and rotate the shaft manually. 2. If pump gets heat up, switch off the main power for 15 minutes and avoid closing the flow control valve and by pass valve simultaneously during operation. Adsorption In Packed Bed 9