8 - Quant Booklet 1 - summary questions (moles and empirical formulae
advertisement

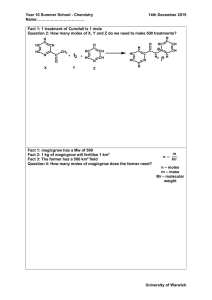
Quantitative Chemistry of Compounds Summary Questions 1 Calculate the formula mass of the following: i) Br2 ii) FeI3 iii) CaSO4 iv) Zn(OH)2 v) Mg(NO3)2 vi) (NH4)3PO4 vii) CuSO4.5H2O viii) CH3(CH2)5CH3 ix) Sodium Sulphate x) Aluminium Hydroxide xi) Lead (1I) Nitrate 2 How many moles are there in each of the following? a) 72 g of Mg d) 1 tonne of NaCl b) 4 kg of CuO e) 20 mg of Cu(NO3)2 c) 39 g of Al(OH)3 3 What is the mass of each of the following? a) 5 moles of Cl2 d) 0.002 moles of (NH4)2SO4 b) 0.2 moles of Al2O3 e) 0.3 moles of Na2CO3.10H2O c) 0.01 moles of Ag 4 How many moles of the following are in 11 g of carbon dioxide? i) Moles of CO2 ii) Moles of carbon atoms iii) Moles of oxygen atoms 5 How many moles of the following are in 5.1 g of aluminium oxide? i) Moles of Al2O3 ii) Moles of Al3+ ions iii) Moles of O2- ions iv) Moles of electrons 6 An experiment was carried out to find the Mr of vitamin C (ascorbic acid). It was found that 1 g contains 0.00568 moles of Vitamin C molecules. Calculate the Mr of vitamin C. 7 a) The molecular formula of some compounds is shown. What is the empirical formula of each one? i) C9H18 ii) B4H10 iii) SO3 b) Work out the molecular formula of the following compounds given the information below? i) empirical formula = P2O5 , ii) empirical formula = CH2, Mr = 284 Mr = 56 c) Work out the empirical formula of the following compound given the information about its composition by mass: K 44.8%, S 18.4%, O 36.8%. 8 Find the empirical formulae of the following compounds using the data given. a) Ca 20 % Br 80 % b) Na 29.1 % S 40.5 % O 30.4 % c) C 53.3 % H 15.5 % N 31.1 % d) C 2.73 g O 7.27 g e) N 15.2 g O 34.8 g 9 An organic compound has the composition 38.7% carbon, 9.70% hydrogen and 51.6% oxygen by mass. The relative formula mass of the compound is 62. Calculate the empirical and molecular formulae of the compound. 10 2.67 g of copper reacts with sulphur to form 4.01 g of copper sulphide. Work out the mass of sulphur that reacted with the copper and then work out the empirical formula of the copper sulphide. 11 A porcelain boat was weighed. After a sample of a metal M, Ar = 119, was placed in the boat, the boat was reweighed. Then the boat was placed in a reduction tube, and heated while a stream of hydrogen was passed over it. The metal oxide was reduced (oxygen removed) to leave the metal M, on its own. The boat was allowed to cool with the hydrogen still passing over it, and then reweighed. Then it was reheated in hydrogen, cooled again and reweighed. The following results were obtained: mass of boat = 6.10 g mass of boat + metal oxide = 10.63 g mass of boat + metal (1) = 9.67 g mass of boat + metal (2) = 9.67 g a) Explain why the hydrogen was passed over the boat while it was cooling. b) Explain why the boat and metal were reheated. c) Find the empirical formula of the metal oxide d) Write a balanced formula equation for the reaction of the oxide with hydrogen. 12 In an experiment to find the number of moles of water of crystalisation in sodium sulfate crystals, Na2SO4.nH2O, 3.22g of of hydrated sodium sulfate crystals were heated gently. When all of the water of crysallisation had been driven off, 1.42g of anhydrous sodium sulfate was left. Find the value for n in the formula.