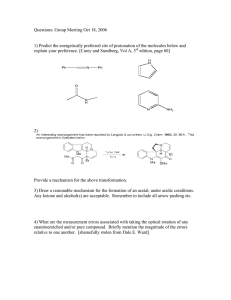
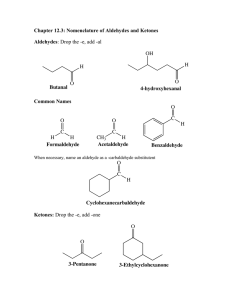
Aldehydes & Ketones Lecture #14 3 March 2021 Announcements – Lecture 14 – 3 March 2021 • Pre-Lecture HW 15 is due Friday (Mar. 5th) by 1 p.m. • Our next problem session is Friday (Mar. 5th) from 3:10 to 4:00 p.m. on Zoom • Expect the Chapter 19 slides and videos to be posted by tomorrow night. The first two episodes are up now. How Does a Toilet Work? Exam-Style Problem: Nomenclature of Aldehydes & Ketones Problem: Name the following compounds. 2-ethylbutanal 4-chlorobenzaldehyde or p-chlorobenzaldehyde (4E,6R)-6-methyl-4-octen-2-one Chapter #19 Lecture Slides Aldehydes & Ketones Part #3 – Hydration and Acetal Formation & Hydrolysis Hydration of Aldehydes and Ketones 1. Aldehydes and ketones form hydrates in the presence of water. • Small aldehydes have K ≈ 1. Ketones have K < 1. • This reaction can be catalyzed by acid or base. Developing Your Chemical Intuition: Acid- and Base-Catalyzed Mechanisms do not invoke strongly acidic intermediates under basic conditions similarly do not invoke strongly basic intermediates under acidic conditions the reason why is that proton transfers are among the fastest reactions in organic chemistry Hydration of Aldehydes and Ketones 2. Base-catalyzed mechanism for the hydration of aldehydes and ketones. • The nucleophile is hydroxide. Hydroxide directly attacks the unactivated carbonyl group. • Don’t invoke strongly acidic intermediates under basic conditions. Here, do not protonate the carbonyl group. Invoke basic and neutral intermediates. Hydration of Aldehydes and Ketones 3. Acid-catalyzed mechanism for the hydration of aldehydes and ketones. • The nucleophile is water. Water attacks the activated (protonated) carbonyl group. • Don’t invoke strongly basic intermediates under acidic conditions. Here, do not attack the protonated carbonyl group with hydroxide. Invoke acidic and neutral intermediates. Acetal Formation 1. Analogous to hydration, aldehydes and ketones can react with alcohols to form acetals. • The acid catalyst used should not be aqueous because acetals hydrolyze in the presence of water and water will shift the equilibrium left. • Popular acid catalysts soluble in organic solvents that do not introduce water include: Acetal Formation 2. Mechanism for acetal formation Acetal Formation 3. Diols form cyclic acetals. • Note the change in entropy is less unfavorable for two molecules adding to form one rather than three molecules reacting to form one. Acetal Formation 4. The additions of oxygen nucleophiles to carbonyl groups is reversible, so these reactions are governed by equilibrium constants. 5. You can drive the equilibrium right (to the acetal) by removing water. You can drive the equilibrium left (to the aldehyde/ketone) by adding water. Acetal Formation 6. Note that hemiacetals are intermediates on the way to acetal formation in acid. • Hemiacetals are usually hard or impossible to isolate because they are not usually stable (because they’ll prefer to form the ketone/aldehyde or acetal, depending on the conditions). Acetal Formation 7. An exception is that aldehydes with a hydroxyl group five or six atoms away usually favor cyclic hemiacetals in water over the free aldehyde. • The entropic cost is not as high when hemiacetal formation is intramolecular. • Hemiacetals are very important for understanding the chemistry of carbohydrates (sugars): Acetal Formation 8. While hemiacetals will react to form acetals in acidic conditions, they will not in basic conditions. • There is no way to make the hydroxyl group into a good leaving group in base. All that can happen is the reverse reaction to re-form the ketone starting material. Acetal Formation 9. Similarly, acetals are stable to strong base. • While protonation would make the ethoxy groups into good leaving groups, there is no equivalent for basic conditions. • This makes acetals great protecting groups for aldehydes and ketones in synthesis when strongly basic reagents are being used. Acetal Formation 10. Acetals are stable in basic conditions. • While protonation would make the alkoxy groups into good leaving groups, there is no equivalent for basic conditions. 11. Acetals will hydrolyze in aqueous acid. Acetal Formation 12. Mechanism for acetal hydrolysis in aqueous acid Removing Water During a Reaction with a Dean–Stark Apparatus Removing Water During a Reaction with a Dean–Stark Apparatus