Water Properties: Structure, Bonding, and Unique Characteristics
advertisement

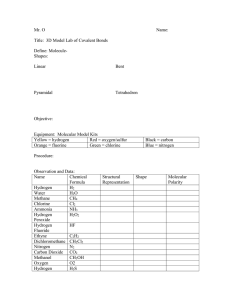
WATER *HYDROGEN AND OXYGEN bond covalently to form the small V-shaped water molecule. * Partial charges develop on the water molecule due to the oxygen being more electronegative than hydrogen and pulling the covalently shared electron pair to itself. Oxygen becomes partially negative and hydrogen partially positive. * Hydrogen bonds develop between the partial positively charge hydrogen of one water molecule and the partial negatively charged oxygen of another water molecule. These hydrogen bonds are intermolecular bonds and are long and strong and they contribute to the relatively high melting and boiling point and other unique properties of water. Draw a diagram of the intra-molecular ( covalent bond) and intermolecular (H- bonds) in water. UNIQUE PROPERTIES OF WATER PURE WATER IS: 1COLOURLESS, ODOURLESS, TASTELESS LQUID WITH MELTING POINT 00 C and boiling point 1000C at standard temperature. 2 Water expands on freezing. Water is unusual, at 40C its density starts to decrease and when it freezes at 0 0 C there is a sudden decrease in density, so ice EXPANDS on cooling unlike most liquids which contract on cooling. Ice will also floats on water as solid ice is less dense than liquid water. In lakes , ponds etc ice forms on the surface trapping more dense warmer water below and thus, aquatic life can survive under the ice in winter. 3 Water has a high specific heat capacity(shc) Shc of a substance is the amount of heat needed to raise the temperature of unit mass 1g of the substance by 10C Shc of water is 4.2 KJ/kg/0C. This keeps water at a relatively constant temperature as large amounts of energy is needed to change the temperature of water Hence ,living things body temperature is maintained and they can survive in extremes of temperature especially in aquatic environments 4 Water has a high heat of vaporization. Heat of vapourisation is the amount of heat energy needed to change water into gas. This is high for water because of the strong hydrogen bonds (intermolecular bonds) between water molecules. Hence water is not very volatile. Explain. Volatile – water is easily changed from liquid water to gas Large amounts of heat can be removed from an organism during sweating and transpiration cooling the organism. 5 Water is a universal , polar covalent solvent. Water dissolves both ionic and polar covalent substances. Many chemical reactions occur in the cells of living things in aqueous solution. The compounds in our bodies which are polar eg amino acids and ionic substances like salt can dissolve in the water in cells . dissolved ions play an important role in nerve conduction and allowing some ions to pass through cell membranes while preventing others from passing. Water plays an important role in many hydrolysis and other reactions in the cell. Question : Property of water 1Odorless, colourless, high mpt, high bpt 2 Expands on freezing 3 High specific heat capacity 4 High heat of vapourisation 5 Water is a universal solvent Effect on living system Water exists as a liquid, water can drinkable as it is odorless Ponds & Lakes freeze at surface and insulate warmer water below and sustains aquatic life in the winter Maintains a constant body temperature. Causes high heat loss during sweating and transpiration, keeps body cool Water dissolves both ionic and polar covalent substances and allows for biochemical reactions to occur in cells of living things.