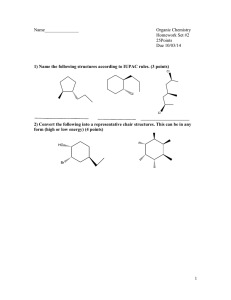
UNIVERSITY OF ZAMBIA NATURAL SCIENCES CHEMISTRY DEPARTMENT CHE 2511 BASIC ORGANIC CHEMSTRY 31st March 2021 TUTORIAL SHEET 2 QUESTION ONE (a) Draw the two chair conformations for each of the following di-substituted cyclohexanes. Circle the more stable one. For convenience, you may abbreviate the substituents (Me, Et, Pr, Bu, iPr, tBu, or the like rather than drawing out methyl, ethyl, propyl, butyl, isopropyl, t-butyl….) Assume that a halogen, OH, or NH2 is smaller than a CH3 or any other alkyl group. • Remember to draw in the hydrogens on each of the “substituted” carbons, also show all the hydrogen atoms for the first three questions, from a-c. a) b) c) d) e) Cis-2-bromo-1-methylcyclohexane Cis-3-isopropyl-1-methyl cyclohexane Trans-2-butyl-1-isopropylcyclohexane Cis-4-ethyl-1-hydroxylcyclohexane Which of the following correctly lists the conformations of cyclohexane in order of increasing energy? (i) chair < boat < twist < half chair (ii) half chair < boat < twist < chair (iii) chair < twist < half chair < boat (iv) chair < twist < boat < half chair (v) half chair < twist <boat < chair (f) What is the energy difference between axial and equatorial conformers of methylcyclohexane? (h) From the perspective of viewing the C-2-C3 bond, draw the Newman projection of the most stable conformation of 2,3-dimethylbutane. 1 QUESTION TWO (a) Consider the chair conformers shown above. The energy costs for each one is given: (i) Which one is more stable? Briefly explain your answer. (ii) Use a suitable formula to estimate the amount of conformer (in percent) at equilibrium at a temperature of 298o K. (iii) What is the meaning of your calculated percentage of this particular conformer? QUESTION THREE (a) Define the term conformation. (b) Draw a Newman projection of the most stable conformation of 2-methylpropane. (c) Describe the source of angle strain and torsional strain present in cyclopropane. (d) View a butane molecule along the C2-C3 bond and carry out a conformational analysis of the molecule: (i) (ii) (iii) Plot a graph of potential energy (PE) versus the dihedral angle. Calculate the energy costs for all the projections you drew in (a). Give the Newman projection of the lowest energy conformer. (e) Consider the molecule 2,3,4-tribromohexane. (i) Draw a fisher projection (C-1 on top, C-4,5 on bottom) of the 2(S),3(R), 4(R) stereoisomer and label as A. (ii) Name (B) and draw (C) molecules, show an pertinent stereochemistry: B Br CH2CH2CH=CH2 CH2CH CH2 C (S)-4-cyclopropyl-4-methyl-2-hexyne CH2CH3 2 (f) Give the relationship between the following structures given below as; conformational isomers, constitutional isomers, enantiomers, diastereomers, cis-trans isomers, meso compounds or not related. (i) CH3 H H H H H H CH3 H CH3 H (ii) CH3 CH3 CH3 H Cl Cl H H Cl H Cl CH3 CH3 (iii) CH3 H Br Br H H3C F H F H CH3 H3C (iv) HO H H3C CH3 Cl CH3 Cl NH2 H NH2 OH CH3 3 QUESTION FOUR (a) What does a polarimeter measure? (b) A 0.5 g of a chiral stereoisomer was dissolved in 20 mL solution. The observed rotation was measured using a polarimeter was found to be -3.0 degrees at 25 oC using a wavelength of 589 nm. The sample tube was 20 cm long. What is the specific rotation of the stereoisomer? (c) If a solution of a compound (30.0 g/100 mL of solution) has a measured rotation of +15° in a 2 dm tube, what is the specific rotation? (d) What is the percent composition of a mixture of (S)-(+)-2-butanol, α= +13.52°, and (R)-(-)-2butanol, α = -13.52°, with a specific rotation α = +6.76°? END OF QUESTIONS 4