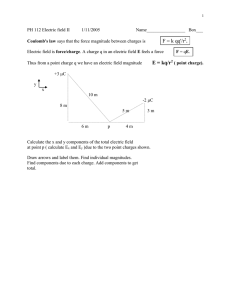
CHAPTER 1: ELECTRICITY Objectives: 1. Explain the role of electron transfer in electrostatic charging. 2. State that there are positive and negative charges, and that charge is measured in Coulombs. 3. Calculate the net electric force on a point charge exerted by a system of point charges. 4. Calculate the electric field due to a system of point charges using Coulomb’s Law. 5. Relate the electric field potential with work, potential energy and electric field. 6. Solve problems involving electric potential energy and electric potentials. Lesson 1.1.1: ELECTROSTATICS The early Greeks knew that amber – a fossilized tree sap used to make jewelry nowadays – have the ability to attract bits of fiber and other small materials after it was rubbed with fur. Later on, however, it was found out that not only amber has this characteristic but other materials as well, such as gems, glass and sealing wax. This is what is now known as electrostatics. This is the process of charging different objects by rubbing them together. Charge from one object can be transferred to another object if they are connected by metal wires and not silk materials. Objects that can transfer charges are known as conductors while those that cannot are nonconductors or insulators. Metals, human bodies, moisture, and a few other objects are conductors. Sparks occur as a result of electrical charges. During dry days, we may feel a shock when we touch a doorknob after walking on a carpet, though this shock does not hurt us. Many charged objects have to be suspended from insulators (silk threads or plastics) or they lose their charge to Earth or the ground. This is called grounding, which is the process to ensure that an object is not charged. Electricity, like gravity, was observed around 1730s to be only attractive. Now, however, it is easy to demonstrate both the attractive and repulsive aspects of electricity. If for example you comb your hair, the plastic comb becomes charged and can be used to attract small bits of paper. Whenever an object is charged by rubbing it with another, both objects become charged. It is now a well-known fact that like charges repel and unlike charges attract. Benjamin Franklin believed that electricity was a single fluid and that an excess of this fluid caused one kind of charged state whereas a deficiency caused the other. Franklin’s use of the fluid model led to his belief that charges are conserved. Meaning, the amount of fluid is just transferred from one object to another but overall remains the same. Later on, the fluid model was discarded as it could not explain later experimental observations. However, Franklin’s idea about the conservation of energy has been verified to a high precision. It is one of the fundamental laws of physics and in generalized form; it can be stated as follows: In an isolated system, the total charge Law of Conservation of Charge is conserved. Lesson 1.1.2: The Electric Force In 1785, French physicist Charles Coulomb measured the changes in the electric force as he varied the distance between two objects and the charges on them. He verified that if the distance two charged objects are doubled, provided that the charges remain constant, the electric force between the objects is reduced. This is known as an inverse–square relationship – inverse because the force gets smaller as the distance gets larger and square because the force changes by the square of the distance. These two effects are combined into a single relationship known as Coulomb’s Law. 𝑭𝑬 = 𝒌 𝒒𝟏 𝒒𝟐 𝒓𝟐 Coulomb’s Law where q1 is the charge of the first object (measured in Coulomb, C) q2 is the charge of the second object (measured in Coulomb, C) d is the distance between the two objects (measured in meters, m) and k = 9 x 109 𝐍𝐦𝟐 𝐂𝟐 Observations of the attraction or repulsion of two charge objects indicates that the magnitude of the electric force depends on the distance between the objects. To summarize, Coulomb Law has the following properties: 1. 2. 3. 4. 5. Like charges repel each other, and unlike charges attract. The attraction or repulsion acts along the line between the two charges. The size of the force varies inversely as the square of the distance between the two charges. The size of the force is proportional to the value of each charge. The unit used to measure charge is the coulomb (C). Electricity and Gravity The mathematical form of Coulomb’s law is the same as that of the universal law of gravitation. The dependence of the inverse–square law on distance also holds for electricity. There are, however, several important differences between gravity and electricity which govern the roles of these two forces. There are two kinds of charges – positive and negative, while there is only one kind of mass. Negative mass does not exist and the gravitational force is never repulsive. Another difference between electricity and gravity lies in the behavior of objects in the respective fields. The gravitational force is proportional to an object’s mass; therefore, objects of all sizes and compositions have the same acceleration in a gravitational field. In contrast, the electric force is proportional to the charge on an object and not to the object’s mass. A consequence of these differences in the different roles these two forces play in man’s lives. The electric force is the dominant force in the atomic world; it determines the properties of atoms and molecules. On the other hand, the gravitational force dominates on the macroscopic scale of people, planets and galaxies. As a numerical example, calculate the magnitude of the electric and gravitational forces between an electron and a proton of a hydrogen atom if the distance between them is 5.29 x 10 – 11 m. G = 6.67 x 10–11 Constants: Nm2 k = 9 x 109 kg2 –31 m1 m2 r2 = 6.67 x 10–11 = 3.63 x 10 FE = k q1 q2 r2 –47 C2 qelectron = –1.6 x 10–19 C qprottton = 1.6 x 10–19 C melectron = 9.11 x 10 kg mprottton = 1.673 x 10–27 kg FG = G Nm2 Nm2 (9.11 x 10−31 kg)(1.673 x 10−27 kg) kg2 (5.29 x 10−11 m)2 N 2 = 9 x 109 Nm2 (1.6 𝑥 10−19𝐶) C2 (5.29 𝑥 10−11𝑚)2 = 8.23 x 10–8 N Therefore, FE > FG. The two forces change in the same way as the distance between the electron and the proton changes, the distance does not matter and the electric force is always much larger. Example 1: By what distance must two charges of +4μC and –7μC be separated so that the force between them is 5.85 x 1010N? k = 9 x 109 q1 = +4μC; Nm2 C2 q2 = –7μC; N = .85 x 1010N Solution: FE = k q1 q2 r2 , deriving the equation for r; r = √ 𝑘𝑞1𝑞2 𝐹𝑒 2 9 𝑁𝑚 (4 𝑥 𝐶2 9 𝑥 10 =√ 10−6𝐶) (−7 𝑥 10−6𝐶) 5.85 𝑥 1010 𝑁 r = 2.075498987 x 10–6 m ≈ 2.08 x 10–6 m Great Scientific Mind! Charles Augustine de Couulomb (1736–1806). Coulomb was a French physicist known for the formulation of Coulomb’s Law, which states that the force between two electrical charges is proportional to the product of the charges and inversely proportional to the square of the distance between them. He likewise established the inverse square law of attraction and repulsion of like an unlike magnetic poles, which became the basis for the mathematical theory of magnetic forces. He also did research on friction on windmills, of machinery, and on the elasticity of metal and silk fibers. Lesson 1.1.3: The Electric Field and Electric Charge Electric field lines are vector representations of electric field in a region of space around a charge. The electric field is defined as the force exerted on a unit charge at a point. It is therefore created by any charged object and exerts forces on other electric charges. Mathematically, it follows the formula: 𝑬= 𝑭 𝒒 Electric Field where E is the electric field (measured in N/C) F is force (measured in Newton’s, N) q is charge (measured in Coulomb, C) Electric Field The diagram above shows the direction of electric field lines – they move away from a positive charge and towards a negative charge. Example 1: What is the electric field at a distance of 2 meters from a point charge of 4μC? Solution: E= 𝐹 𝑞 but F =𝒌 E = 9 x 109 𝒒𝟏 𝒒𝟐 Nm2 C2 𝒓𝟐 ( , therefore, E = 3 𝑥 10−6𝐶 (2𝑚)2 𝑘𝑄 𝑟2 ) = 6750 N/C Electric Charge In 1909, Robert Millikan was able to determine the charge on an electron by studying the behavior of charged oil drops. Through Millikan’s recorded observation, the equation to find the total charge on an object with an excess or deficit number (n) of electrons was calculated to be: Q = ne Electric Charge Example 2: How many electrons have been removed from a positively charge object if it has a charge of 4.5 x 10–14C? Solution: Q = 4.5 x 10–14C e = –1.6 x 10–19 C 𝑞 4.5 𝑥 10−11 𝐶 N = 𝑒 = 1.6 𝑥 10−19 𝐶 N = 2.81 x 108 electrons were removed Fun Fact! #1 Nobel prizes for physics, chemistry, and other fields have been awarded almost annually since 1901, according to the terms of the will of Alfred Bernard Nobel (1833–96), the Swedish industrialist who invented dynamite. The awards are made by the Swedish Royal Academy of Sciences. Each prize has a cash value, which increases from year to year. Fun Fact! #2 Sharks are sensitive to small electric fields surrounding preys such as fish. Experiments have shown that a shark will attack an artificial electric field and ignore food. Artificial electric fields were used at the 2000 Olympics in Australia to keep sharks away from triathlon competitors swimming in the Sydney harbor. Lesson 1.1.4: Electric Potential Work is required to move a charged particle in an electric field; this work changes the electric potential energy of the particle. When the particle is released, the electric potential energy is converted to kinetic energy. Therefore, the electric potential energy of a charge particle is equal to the work one to move the object from a reference position to the object’s final location. Objects with different charges have different electric potential energies at a given point. The electric potential (V) at each point in an electric field is defined as the ratio of the electric potential energy (EPE) and the charge of the object (q). The electric potential is equal to the work required to bring a positive charge of 1 C from the zero reference point to a specific point. The unit for electric potential is joules per coulomb (J/C) or volt (V). For example, a 12-volt battery has an electric potential difference of 12 volts between its two terminals. This means that 1 coulomb of charge moving from one terminal to the other would gain or lose 12 joules of energy = (1C) (12V). The electric potential from a single point charge is given by the equation: 𝒌𝑸 𝑽= 𝒓 where V is electric potential in volts (V), r is the distance travelled by the point charge in meters (m), Q is the charge of the point charge in Coulomb (C), and k (constant) = 8.99 x 109 𝑁𝑚 2 𝐶2 ≈ 9 x 109 𝑁𝑚 2 𝐶2 Example 1: Determine the potential at a point 0.50m from a +20µC point charge. Solution: Q = +20µC = 20 x 10–6C and r = 0.50m 𝑽= 𝒌𝑸 𝒓 (9 x 109 , so V = V = 3.6 x 10 𝑁𝑚2 )(20 x 10–6C 𝐶2 ) 0.50𝑚 5 𝑁𝑚 𝐶 V = 3.6 x 105 V Example 2: Calculate the electric potential at point A due to two point charges. The first point charge is 60cm from Point A and has a charge of –50µC while the second is 30cm way and has a charge of +50 µC. Solution: Q1 = –50µC = –50 x 10–6C and r = 60cm = 0.60m Q2 = +50 µC = +50 x 10–6C and r = 30cm = 0.30m 𝑽𝟏 = 𝑽𝟐 = 𝒌𝑸𝟏 𝒓𝟏 𝒌𝑸𝟐 𝒓𝟐 (9 x 10−9 , so V1 = (9 x 10−9 , so V2= 𝑁𝑚2 )(−50 x 10−6C 𝐶2 0.60𝑚 𝑁𝑚2 )(+50 x 10−6 C ) 𝐶2 0.30𝑚 ) = –0.75 x 106V and = 1.5 x 106V The total potential at point A is equal to the sum of the potential due to the two point charges, mathematically VA = V2 + V1. Therefore, VA = 1.5 x 106V + (–0.75 x 106) V, resulting to VA = 0.75 x 106V. Great Scientific Mind! Alessandro Volta (1745–1827). Volta became a professor of physics at a high school in Como, Italy. He discovered the electrophorus, a device capable of producing and storing a large electrical charge. His most important achievement was the invention of the first electric battery using bowls of salt solution connected by arcs of metal with copper at one end and tin or zinc at the other. Napoleon awarded Volta the medal of the Legion of Honor and made him a Count. NOTES ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________