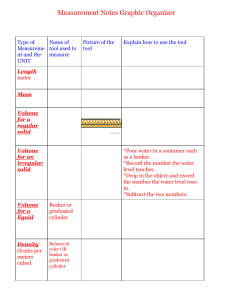
Pre-Lab Assignment Name: Divya Paul _____ Exp # 1 Title: “Introduction to Measuring Techniques” Brief description of tasks In this experiment, we will be utilizing a variety of common laboratory methods to measure solids and liquids including the balance, graduated cylinder, pipet, and buret. We will also become familiar with dispensing liquids and understanding how to read measurements. Outline of the experiment (major steps), and an example of Calculations Balance 1. Turn on balance and place weighing paper on pan once display is at 0 2. Record the mass 3. Place 1/3 tsp of the solid onto the pan and record the mass 4. Calculate how much solid you have (mass of paper and solid – mass of weighing paper) and record the mass 5. Repeat the first 3 steps and spoon out about 1 g of the solid to weigh out predetermined amount of solid Graduated Cylinder 6. To get mass of 6mL of liquid from graduated cylinder, first add water up to 6 mL line 7. Dry your beaker and measure its weight 8. Pour the water into the beaker and reweigh it Pipet 9. Fill a beaker halfway with water 10. Attach the pump to the pipet’s top and draw water in by placing the spout under water and turning the pump wheel in a clockwise motion 11. Remove spout from under the water after the water reaches past the last graduation 12. Weigh another dry beaker and record mass 13. Place 6 mL of water into beaker and record its mass Buret 14. Clamp a buret by attaching the buret clamp to a stand 15. Rinse the buret by adding 6 mL of water using a funnel and letting the water run into a waste container 16. Then, fill the buret to a satisfactory level and let the liquid run into the waste container until reaching the desirable amount 17. Dry a beaker and measure its mass 18. Add 6 mL of water into the beaker from the buret and reweigh Calculations Mass of weighing paper = 0.3670 g Mass of solid and weighing paper = 0.5881 g Mass of substance = mass of solid and weighing paper – mass of weighing paper = 0.5881 g – 0.3670 g = 0.2211 g Turn over Pre-Lab Assignment Table of the Reagents Chemical name Formula Water H2O Unknown Solid N/A Molecular wt 18.015 g/mL N/A Physical State Amount to use Liquid 18 mL total (used 6 mL three times) Solid 1/3 of tsp and 1 g