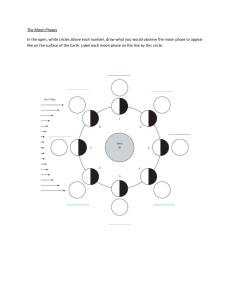
5th Grade Science Tackling the 5th Grade Science Test Review Guide Atom Smallest unit of an element Element Material that cannot be broken down into anything simpler by chemical reactions Molecule 2 or more atoms joined together by sharing electrons Compound Chemical combination of 2 or more different elements Fe₃O₂ (iron oxide, also known as rust) NaCl (sodium chloride, also known as salt) H₂O (water) Anything that has mass and takes up space What is matter? Mass is….. Volume is…. The amount of matter in an object. Measured in grams, kilograms Measured using a balance or triple beam balance The amount of space an object takes up. Measured in milliliters and liters Can be measured by l x w x h, graduated cylinder, water displacement How would you find the volume of an irregular object, such as a rock, using only a graduated cylinder and water? Describe the process. Describe how particles of matter are arranged in a SOLID. Draw a picture showing this arrangement. Read and record the volume of the water. Drop the rock into the water. Read and record the volume of the rock and the water, then subtract the volume of the water from it. Particles are tightly packed together. They can vibrate but cannot move out of position. Describe how particles of matter are arranged in a LIQUID. Draw a picture showing this arrangement. Particles are further apart, and can flow past one another. Describe how particles of matter are arranged in a GAS. Draw a picture showing this arrangement. Particles are much farther apart, rarely coming in contact with one another. What is density? Use this definition to The amount of mass per unit of explain why some object can float and volume. If an object is denser than the some sink. fluid it is in, it will sink. If it is less dense, it will float. What are physical properties? What are examples of physical properties? Properties that can be observed. Color, shape, size, texture, function, density, state of matter, malleability, luster, viscosity, electrical and thermal conductivity, magnetism, solubility, mass, length, volume, density What are conductors and insulators? Conductors: material that easily transmits heat and electrical energy to pass through (ex: most metals) Insulators: Material that is a poor conductor of heat and electricity (ex: rubber, wood, cotton, feathers, Styrofoam) What are chemical properties? When are you able to observe them? Properties that describe how matter interacts with other substances. You are only able to observe them during a chemical change. What happens to matter in a physical change? Matter changes physically, but nothing new is created. It is still the same type of matter. What are some examples of physical changes? Tearing paper, slicing carrots, water changing to water vapor, water melting, making a solution, etc. What happens to the particles of matter when thermal energy is added to it? The particles speed up and start moving faster. Sometimes a phase change can occur. What happens to the particles of matter when thermal energy is taken away from it? The particles slow down and move more slowly. Sometimes a phase change can occur. Describe the phase change during: Melting: Boiling: Condensing: Freezing: In which phase of matter are particles moving fastest? Slowest? Why? What is a mixture? Give some examples of mixtures. Solid to liquid Liquid to gas Gas to liquid Liquid to solid Freezing Point for water: 0⁰ Celsius Boiling Point for water: 100⁰Celsius Particles move fastest in gases. They move slowest in solids. Gases have the most thermal energy and solids have the least. A mixture is made up of two or more types matter that are combined physically, 1. Party mix 2. sand and water 3. Air What is a solution? Give an example of a solution. What is solubility? A type of mixture in which substances are thoroughly dissolved and do not separate with gravity. An example is sea water. Solubility is the maximum amount of a substance that can be dissolved by another substance How are mixtures and solutions alike? Mixtures and solutions are made of How are they different? matter that are physically combined. In solutions, you cannot see the individual particles because they are dissolved in the substance. What is a solute and solvent? A solute is a substance that gets dissolved. A solvent is what dissolves the solute. There is more solvent compared to solute in solutions. Compare concentrations of solutions. 3 ways to increase the rate of dissolving: What happens to the particles of matter during a chemical change? What are some chemical properties? Give some examples of chemical changes. Solutions that have a great amount of solute compared to solvent are considered to be concentrated. Solutions that have a great amount of solvent versus solute is considered to be diluted. 1. Increase temperature 2. Stirring, mixing, or some kind of agitation 3. Using smaller particles so that there is greater surface area exposed to the solvent. In a chemical change, atoms (matter) is not created nor destroyed. Instead, a new substance or substances are created the original substances. Flammability, rusts, tarnishes, reacts with other substances 1.wood burning 2. fireworks exploding 3. metal rusting 4. cake baking 5. food rotting 6. candle burning 7. copper turning What are clues, or evidence, that a chemical reaction has taken place? Bubbles are produced, color changes, an odor is produced, light or heat energy is released, a new solid called a precipitate forms How is a physical change different from a chemical change? In a physical change, the matter changes form, but nothing new is created. In a chemical change, new substances were formed with different properties. Gravity is a force of attraction that exists between any two objects have mass. What is Gravity? What affects the force of gravity? The greater the DISTANCE, the less the force of gravity. The MORE Mass, the stronger the force of gravity. What about gravity on Earth? The earth’s gravity pulls everything to the center of earth which is why what goes up must come down. Heavier objects are harder to lift because the effect of gravity is stronger. The earth has more mass than the moon so therefore its gravity keeps the moon in orbit. The sun has more mass than everything in our solar system, so therefore all the planets orbit around the sun. A force changes the position of an object and puts it into motion. All forces have strength and direction. The strength of a force is measured in Newtons (N). Speed is how fast an object is moving. Acceleration is an increase in speed, and deceleration is a decrease in speed. How role does gravity play in our solar system? A force is a push or a pull. What is speed and acceleration? Some forces result when two objects physically touch each other. These forces are called CONTACT FORCES. Some forces result even when two objects do not physically touch each other. These forces are called NONCONTACT FORCES. Examples of CONTACT FORCES: 1. Applied force (direct push or pull) 2. Friction: resists or opposes motion (rough surfaces have more friction than slippery surfaces) Examples of NONCONTACT FORCES: 1. Gravity 2. Magnetism Newton’s First Law of Inertia An object at rest will remain at rest unless acted on by unbalanced forces. (Why we need seatbelts) Newton’s Second Law of Acceleration An object accelerates when a force acts on an object with mass. The greater the mass of the object being accelerated, the more force needed to accelerate the object. (Force= Mass x Acceleration) Newton’s Third Law For every action, there is an equal and opposite reaction. (Rifle kicks back after bullet is propelled forward). Energy is not Created nor Destroyed. It simply changes forms. For example, from light energy to heat energy. Energy can change from potential energy to kinetic energy. Potential Energy Stored energy in an object due to position Ready to be used, but not being used The higher and greater the mass, the more potential energy Kinetic Energy The energy of motion Formed or caused by motion The faster and greater the mass, the more kinetic energy Characteristics of Inner Planets 1. 2. 3. 4. 5. Smaller in size Closer together Rocky/Terrestrial Few Moons Mercury, Venus, Earth, Mars Characteristics of Outer Planets 1. 2. 3. 4. 5. Larger in size Gaseous Many Moons Farther apart from each other Jupiter, Saturn, Uranus, Neptune The Planets in order from closest to sun to the furthest Mercury, Venus, Earth, Mars, Jupiter, Saturn, Uranus Neptune My Very Excellent Mother Just Served Us Nachos. Why is the Sun the brightest star to Earth? What are the phases of the moon in orders? What phase does a solar eclipse take place? It is the closest star to Earth (9.3 millions miles away) All other starts are LIGHT YEARS aways! 1. New Moon 2. Waxing Crescent 3. First Quarter 4. Waxing Gibbous 5. Full Moon 6. Waning Gibbous 7. Third/Last Quarter 8. Waning Crescent During a NEW MOON: the moon is between the sun and the earth, and the moon’s shadow is cast on a part of Earth. Which phase of the moon does a LUNAR eclipse occur? A Full Moon – The Earth is between the sun and the moon and Earth’s shadow is cast onto the moon (makes it look red in color). Why do the circumpolar constellations (Ursa Major, Ursa Minor) appear to circle in the night sky and other constellations appear to move across the sky? Circumpolar constellations (Big & Little Dipper) are directly north of Earth They appear all year long and APPEAR to circle in the sky because of Earth’s ROTATION. Other constellations also appear to move across the sky because of Earth’s rotations. Why do some constellations only appear during certain times of the year? Orion, Leo the Lion, Scorpio and other seasonal constellations only appear during certain times of the year as Earth passes by their location during its revolution around the sun. 1. One complete rotation of Earth on its axis: 24 Hours (1 Day) 2. One revolution of Earth around the Sun: 365 days, 12 months, 1 year 3. One revolution of the Moon around Earth: 28-31 Days Lengths of Earth and Moon movements: What scientists contributed to the model of the solar system? Aristotle & Ptolemy: Believed the Earth was the center of solar system Copernicus & Galileo: Copernicus believed the sun was the center. Galileo discovered this as a fact by improving the telescope. How are stars used in navigation (finding direction)? The North Star is directly north of the North Pole. It can be used to determine North. Why does Earth experience seasons? It is because of Earth’s revolution around the Sun while being tilted at 23°. When the Northern Hemisphere is tilted AWAY from the Sun, it is winter. When the Northern Hemisphere is tilted TOWARD the Sun, it is Summer. The opposite season occurs in the Southern Hemisphere. The reason is because Earth’s tilt cause the sunlight energy (heat & light) to hit Earth at either more direct or indirect angles. What is photosynthesis? The process through which plants use energy from the sun (LIGHT) to make sugar from carbon dioxide and water. Where does photosynthesis occur? Photosynthesis occurs in the leaves. The roots absorb water, the stem carries the water to the rest of the plant. How do animals depend on plants and plants depend on animals? Plants take in carbon dioxide (CO2) in order to go through photosynthesis. During this process, they release oxygen (O2). Animals take in the oxygen and breathe out carbon dioxide. We need them and they need us! What is the chemical formula for photosynthesis? Light energy + water (H2O) + carbon dioxide (CO2) → chemical energy (sugar/glucose) + Oxygen (O2) Light energy from the sun is converted to chemical energy in the form of sugar which, when eaten by other organisms can provide energy for all cell processes. What if plants live in darkness? Can they go through photosynthesis? Desert Temperate Forest Taiga Tropical Rainforest Tundra Photo means light. Plants that live in darkness find other means to get energy to make their own food. Plants in the deep ocean can get energy from heat in hydrothermal vents from the ocean floor. Or, the can get energy from chemicals in the ocean to make their own food. This is called chemosynthesis. Dry climate, hot during the day, cold at night Animals active at night Plants store water Experience seasons Deciduous trees Deer, squirrel, raccoons Plenty of rain to support many trees Coniferous forest that are evergreen Plenty of precipitation to support trees Most trees have needles for leave Cold, snowy winters Mild Summers Bear, elk, snowy rabbit Lots of Rain Very warm all year long Near equator Tall trees that create a canopy Plants near the rainforest floor have large thick leave to compete for sunlight from the tall trees. Monkeys, jaguars, snakes, birds, insects Cold, harsh weather all year long Permafrost Little rain No trees Plants grow close to the ground and consist of grasses and small shrubs Snowy owl, polar bear, musk ox Grassland Types of Consumers Rains, but not enough to support many trees Fertile soil Lots of grasses with strong roots Cattle, buffalo, prairie dogs Herbivores: eat plants only Carnivores: eats other animals only Omnivores: eats plants and animals Scavengers: eats dead animals What are producers and decomposers? Producers produce their own food (plants). Decomposers break down dead plants and organisms to return their nutrients back into the soil. What happens to energy in a food chain? Energy starts with the sun and is absorbed by producers. The energy available decreases as it moves through the food chain. Producers contain the most energy and there should be more producers in an ecosystem than any other types of organisms. What are food webs? Food webs show multiple pathways of energy flow within an ecosystem. They are used to study how the addition or removal of any organism can affect an entire ecosystem. To prepare for your test, make sure to: Look over your journal notes and reflect on the labs performed during class. 2. Study through all of your notes. 3. If you have internet access, read the lessons and complete the practices and games on studyisland.com. Quizlet.com is also a great tool to help you review. 4. Have someone quiz you over the study guide questions.