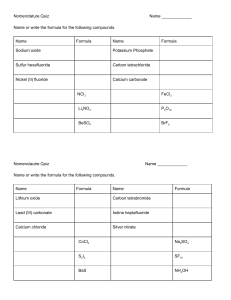
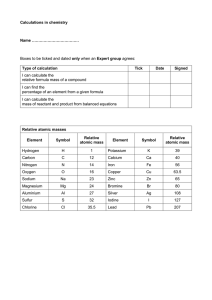
THE EXTRACTION OF IRON • Three substances are needed to enable to extraction of iron from its ore. –Iron ore, haematite - often contains sand with iron oxide, Fe2O3. –Limestone (calcium carbonate). –Coke - mainly carbon. • The combined mixture is placed a giant chimney called a blast furnace • Hot air is blasted into the furnace causing coke to burn rapidly and o raise the temperature to 2000 C. – C + O2 → CO2 • The carbon dioxide then reacts with hot carbon to form carbon monoxide. – CO2 + C → CO • Carbon monoxide then reduces iron(III) oxide to iron metal. –3CO + Fe2O3 → 2Fe + 3CO2 • The molten iron is collected at the bottom of the furnace. • It is drained off periodically into moulds and it allowed to cool. • Limestone is calcium carbonate and it is added to the blast furnace to remove the impurities in the iron ore. • Calcium carbonate is decomposed by heat in the furnace to give calcium oxide and carbon dioxide. – CaCO3 → HEAT CaO + CO2 • The main impurity is silica which is silicon dioxide. • Silicon dioxide is solid at the furnace temperature and the furnace would become blocked if it was not removed. • Silicon dioxide reacts with calcium oxide to form calcium silicate or slag which is liquid. – CaO + SiO2 → CaSiO3 • Slag flows to the bottom of the furnace where it floats on the liquid iron and is easily removed. • The slag is allowed to cool until becomes a solid and is used for road construction.