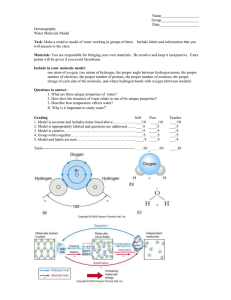
Intermolecular forces Dispersio n This is the weakest of Dipoledipole attraction These forces occur Hydrogen bonds This is a special kind the intermolecular forces when the partially of dipole-dipole and exist between all positively charged interaction that occurs types of molecules, part of a molecule specifically between a whether ionic or interacts with the hydrogen atom covalent—polar or partially negatively bonded to either an nonpolar. The more charged part of the oxygen, nitrogen, or electrons a molecule neighboring has, the stronger the molecule. fluorine atom. The partially positive end London dispersion of hydrogen is forces are. attracted to the partially negative end of the oxygen, nitrogen, or fluorine of Done by: Maryam Taha Grade: 10A another molecule.