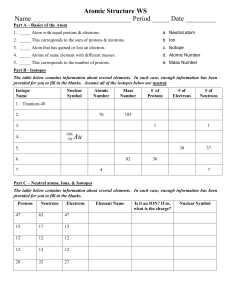
Ions and Isotopes Practice Worksheet
Isotopes:
1.
Define an isotope.
Name:
2.
What would happen if the number of protons were to change in an atom?
3.
Another way to write isotope
6
14 C is carbon-14. What is the number of neutrons for this isotope?
4.
Here ae three isotopes of an element:
6
12 C
6
13 C
6
14 C a.
The element is: b.
The number 6 refers to the: c.
The numbers 12, 13, and 14 refer to the: d.
How many neutrons are in the first isotope: e.
How many neutrons are in the second isotope: f.
How many neutrons are in the third isotope:
# of protons
# of neutrons
# of electrons
Ions:
Determine the charges of the scenarios below:
1.
An atom having lost two electrons
Chromium-58 Chromium-63
2.
An atom having lost six electrons
3.
An atom having gained one electron
4.
An atom having gained three electrons
5.
An atom having lost 5 electrons
Extension: For each of the following ions listed below use the periodic table to determine the total number of electrons that element contains.
1.
Al +3
2.
Fe +3
3.
Mg +2
4.
Sn +2
5.
Co +2
6.
Li +1