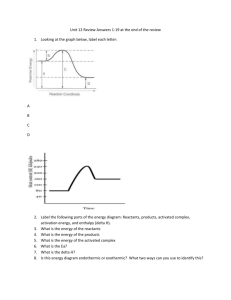
Name__________________________ Chemistry Unit 7 REVIEW Test 4/23/18 SECTION 1: Hess’s Law 1. Calculate the ∆H value for the following reaction using equations 1 and 2: 2NO + O2 N2O4 1. N2O4 2NO2 ∆H = 57.2kJ 2. 2NO + O2 2NO2 ∆H = -114 kJ SECTION 2: Endothermic and Exothermic reactions. Directions: Write down if the following questions (2-6) are endothermic or exothermic reactions. 2. ________ Ethanol is burned, releasing heat. 3. ________ Heat is supplied to water, causing it to boil. 4. ________ A candle is burned so that the wax is melting. 5. ________ When road de-icer reacts with water, it releases energy to help ice melt. Directions: Define the following words or answer the question. 6. Endothermic reaction 7. Exothermic reaction 8. Specific Heat (definition + equation) a. Compare how fast water heats up verses concrete using specific heat. 9. Heat (definition + symbol + equation) 10. Temperature Name__________________________ Chemistry Unit 7 REVIEW Test 4/23/18 11. Enthalpy (definition + symbol) 12. Equilibrium (make sure explain heat flow, see p. 505 in book) SECTION 3: Solving for heat (q) 13. How many Joules of heat are needed to raise the temperature of 17.0 grams of Iron from 25o C to 68o C? The specific heat of Iron is 0.46 J/(g∙oC). 14. In the previous question, how much heat was lost to the surroundings? 15. The specific heat of water is 4.18 J/(g∙oC). 25 grams of water is heated from 12oC to 87oC, how many Joules of heat was required? SECTION 4: Solving for Specific Heat (C) Directions: Use the table pictured below as a reference point to answer the following question. 16. You have a 43.1 gram sample of a metal. However, you don’t know what the metal is so you run a calorimetry experiment to try to figure out what the metal is. You discover that it releases 323 Joules of energy when there was a temperature change of 32oC. Find the specific heat, and then use the table to figure out what metal you have. Name__________________________ Chemistry Unit 7 REVIEW Test 4/23/18 SECTION 5: Thermochemistry Stoichiometry Directions: Answer the following questions. 2 Mg + O2 2MgO + 1204 kJ 17. Is the question above an endothermic or exothermic reaction and how do you know? 18. How many kilojoules of heat would be released if 7.4 moles of Mg was used? 19. How many kilojoules of heat would be released if 96 grams of MgO was produced? 20. How many moles of Mg would be used if 6020 kJ was released? 21. How many grams of Oxygen would be used if 1806 kJ was released?