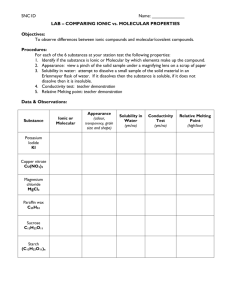
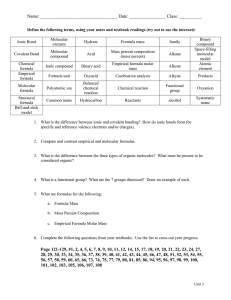
Name: ________________________________________ Date: ___________________________ Period: ____ Lab Partner(s): ______________________________________________________________________________ IONIC VS. COVALENT PROPERTIES LAB OBJECTIVE: 1. To observe properties of one ionic compound and one molecular compound and compare experimental data to generalized data on ionic and molecular properties. 2. Investigate differences in properties based upon bonding type and structure. SAFETY: Safety goggles and aprons MUST be worn at all times! Notify your instructor of any spills or broken glass. MATERIALS: Sodium chloride (NaCl) Aluminum weigh plates Paraffin wax (C36H74) Large forceps Deionized water Conductivity meter Pipette Hot plate Spot plate PROCEDURE: 1. Gather all necessary materials. 2. Record the name, molecular formula, and appearance of each compound in Table 1. 3. Conduct tests for volatility (smell) and hardness. Record observations. 4. Place a small amount (fewer than 10 crystals of NaCl and about a pea-sized piece of paraffin wax) of each sample into individual wells in the well plate. Use the conductivity meter to test for conductivity of the solids. Record observations. 5. Add a few drops of deionized water to each sample in the well plate and stir with a stirring rod to test for solubility in water. Record observations. 6. Use the conductivity meter again to test for conductivity of the dissolved compounds. Record observations. 7. Look at the test tubes in the fume hood to determine the solubility in hexane. 8. Add a small sample of NaCl to the aluminum weigh plate. 9. Turn on the hot plate and place the sample on the hot plate. Wait 2 minutes to see if the sample melts. Record your observations in Table 1. 10. Use large forceps to remove the weigh plate from the hot plate surface. Throw away NaCl from step 11. Add a small sample (pea-sized) of paraffin wax to the aluminum weigh plate and place it on the hot plate. Record observations. 11. Pour all liquid waste into the labeled waste container. 12. Throw away all solid waste. DATA: Table 1: Properties of Ionic and Molecular Compounds Property Ionic Compound Molecular Compound Observations Chemical Name and Formula Appearance (at RT) Volatility (High or Low) Hardness (Hard or Soft) Melting Point (High or Low) Solubility in Water (Soluble or Insoluble) Solubility in Hexane (Soluble or Insoluble) Conductivity in the Solid State Conductivity in the Dissolved State ANALYSIS: Directions: Answer each question in complete sentences in your lab notebook. 1. Compare the data obtained in the lab to expected properties of ionic and molecular compounds. Does the data support the expected resultant properties? 2. Look at the models of paraffin wax and sodium chloride at the front of the room. Explain the differences in conductivity and melting point between the two substances in terms of bonding and structure. 3. How would one determine if an unknown compound was ionic or molecular based upon data from this lab? What evidence would indicate that the compound was ionic or molecular?