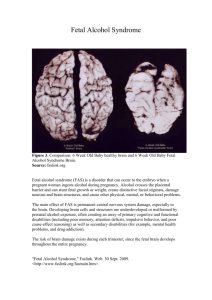
as a PDF
advertisement

INFANT BEHAVIOR AND DEVELOPMENT 17, 159-l 64 (1994) Fetal Reactions to Recurrent Maternal Speech ANTHONY J. DECASPER University oj’Nor_th Carolina at Greensboro JEAN-PIERRE LECANUET EPHE, CNRS U.R.A. 315 MARIE-CLAIRE BUSNEL Unil,ersite Paris V CAROLYN GRANIER-DEFERRE EPHE, CNRS U.R.A. 315 ROSELYNE MAUGEAIS Universitc~ Paris V Pregnant women recited a short child’s rhyme, “the target,” aloud each day between the thirty third and thirty seventh weeks of their fetuses’ gestation. Then their fetuses were stimulated with tape recordings of the target and a control rhyme. The target elicited a decrease in fetal heartrate whereas the control did not. Thus, fetuses’ exposure to specific speech sounds can affect their subsequent reactions to those sounds. More generally, the result suggests that third trimester fetuses become familiar with recurrent, maternal speech sounds. fetal experience fetal speech perception fetal memory fetal-auditory perception maternal speech prenatal sensory experience The hypothesis that normal prenatal exposure to maternal speech influences the early development of speech perception is consistent with research showing that pregnant women’s speech sounds are present in utero, that lateterm fetuses possess relevant perceptual abilities, and that postnatal perception of speech sounds can be influenced by prenatal experience. One implication of the hypothesis is that exposure to maternal speech alters fetal perception of speech sounds. A test of this implication is reported here. Recordings from within a pregnant human uterus have revealed the presence of the woman’s speech sounds (Abrams, Gerhardt, & Peters, 1992; Benzaquen, Gagnon, Hunse, & supported by grant CIES This work was 34410.360.80604 from the French Ministry of Exterior Relations to A.J.D.. who was sponsored by P. Roubertoux, Universite Paris V, URA 1294 CNRS, Genetique, Neurogenetique et Comportement. There was also support from the Fulhright Program and the Research Council of the University of North Carolina at Greensboro to AJD and from grant 3006 I, 1300.5 INSERM and 63X CNAMTS to M.C.B. and C. Sureau. The authors wish to thank C. Sureau (Baudelocquc Maternity CHU Cochin. Paris, France) and his staff for their valuable help. We are especially grateful to the mothers and their infants for making this research possible. Correspondence to Anthony J. DeCasper, Department of Psychology. UNC-Greensboro, Greensboro. NC 27412. Foreman, 1990; Querleu, Renard, Versyp, Paris-Delrue, & Crepin, 1988; Richards, Frentzen, Gerhardt, McCann, & Abrams, 1992; Versyp, 1985). The intensity of her speech in utero is near its intensity ex utero; some studies report intrauterine intensity is slightly augmented and some report it is slightly attenuated. All reports indicate that higher speech frequencies are attenuated more (augmented less) than lower ones. Otherwise, the spectral properties of maternal speech are similar in and ex utero. It follows that some acoustical properties important for speech perception are preserved in utero. Indeed, adults can readily perceive suprasegmental features of a pregnant woman’s speech that had been recorded in utero, for example, intonation contours or meter, and can identify about 30% of its individual phonemes (Querleu et al., 1988; F. Versyp, personal com- munication, May 5, 1985). That late-term fetuses respond to sound has been known for some time (e.g., Busnel & Granier-Deferre, 1983). Now, studies of stimulus-elicited heartrate change are being used to assess the character of fetal auditory perception. For example, when fetuses are in a behaviorally active state and are stimulated with octave-band stimuli centered on 500, 1000, and 2000 Hz at intensities of 10.5, 110, and 120 dB SPL, ex utero, the stimuli elicit heartrate accel159 160 DeCasper et al. erations with a probability and magnitude that increase along with intensity and frequency. However, if the fetus is in a behaviorally quiescent state, cardiac accelerations are dampened and the 500 Hz stimulus elicits many more cardiac decelerations at 105 dB and only decelerdtions at 100 dB (M = -10 bpm; Lecanuet, Granier-Deferre, & Busnel, 1988). If a quiescent fetus is repeatedly stimulated with the bisyllable /hi/w/ which is then changed to /hahi/, both presented at 95 dB ex utero, cardiac decelerations occur to the initial stimulation and then again at the stimulus change (M = -2.6 bpm; Lecanuet, Granier-Deferre, & Busnel, 1989). Quiescent fetuses’ heartrate also slows with the onset of brief sentences presented at 87 to 90 dB ex utero (M = -3.4 bpm), but reactions to less intense stimuli have not been discerned (Lecanuet, Granier-Deferre. Jacquet, & Busnel. 1992). Practically speaking. demonstrations of speech discrimination require that the fetus be in a behaviorally quiet state because that is when heartrate variability is low: Detecting the small decelerations elicited by moderate to low intensity acoustically-complex speech sounds is easier to do when variability is low. Research with newborns indicates that prenatal experience with maternal speech can influence their perception of speech cues important for discriminating among speakers (e.g., DeCasper & Fifer, 1980) as well as their perception of linguistically-important speech sounds (DeCasper & Spence. 1986). In the DeCasper and Spence (1986) study, pregnant women recited a specific prose passage throughout their last 6 weeks of pregnancy. After birth. their newborns were tested in an operant choice task where one pattern of sucking was rewarded with a recording of the recited passage and a second pattern was rewarded with a recording of a control passage. Importantly, some infants’ test passages had been recorded by their own mothers whereas others’ had been recorded by another woman. The recited passage was the more potent reinforcer. independent of who spoke it during the test, presumably because of the infants’ prenatal exposure to its acoustic properties. The data and the hypothesis imply that fetal perception, too, not just that of young infants, should be affected by exposure to maternal speech. This implication was evaluated by hav- ing pregnant women recite a specific child’s rhyme, “the target,” each day for a month and then testing fetal reactions to recordings of the target rhyme and a control rhyme. METHOD Recitation Phase P~~,~~~nt Suhjrcts. Twenty-eight healthy women in their thirty fifth week of pregnancy awe recruited Itom the birth training program at the Cllniquc Universitaire Baudelocque. Paris, France. They volunteered to participate atier being mformed about the study. t’roced~wes und hfatc~kdr. Hall’ the women were taught to recite a rhyme cntitlcd Ltr Poulerrr and hall’ here taught a rhyme entitled 1,~ l’clir C‘/u/xrrrt/.’ They wrrc told to recite their rhyme, the target, aloud three time\ in uccession each day I’M 4 weeks. They recorded the date and time of each recitation m a log as proof that they followed instructions. After 4 week\, their letuses were stimulated with the recordings of both rhyme\-one the tarset and the other the control. Testing Phase The mtrautcrine Intenalty of the te\t stimuli was c&ulated to be 5X to 60 dB uhtch approximate\ the level of it pregnant woman’\ qxech. Although exi\tinp data suggest that the control stmIuIu\ dclwered at X0 dB would bc too weak to elicit drtcctahlc cartllac dcccleratwn~ (I.cc;rnuct et al., IYXY). a large hod!< 01 rewarch indicates that cardiac reacttonk to the target fiould hc enhanced by prior cxpw 5urc to it (e.g.. Cooptxmith Xr Leon. IYX4: Demher, IYhO: Jacohy K: Dallas. lOXI: Jacohy & Wlther?poon. IYX?_: Solomon HL Po~tm;rn, I Y52: Warm-en. IYX3-). Rcwnrch show\. l’or cxamplc. that Inmiliar stimuli can he detected at lower inten\itwa or 5hortcr p~wwntation durations than unfamiliar stimuli. Enhanced perceptual procc\ring 01 familiar stimuli also wem\ to occur ,ju\t after hirth (e.g.. DeC’a\per Kr Pifcr. 19x1: DeCn\pcr NC Spcnce. IYXh). Therefore. the hpecific expectation u:l\ that target \timuli would elicit a decrcow in letal heartratt‘, hut control \timull would have no dtxrrnahlr cflect. The women uorc earphones (Ku\\ Pro4/AAAI and II\tcned to continuous melodic guitar music w,how tntcnsity wa\ gradually incrawl until It ma\kcd their perccptltrn 01 the speech stimuli hut was not uncomfortable. The masking music prevented the women 1‘rom influencing MaI reaction\ through their dll’l’ercntlnl wxtion% to the two poa\apr\. 161 Fetal Speech Perception Beat-to-beat intervals of the fetal heart were recorded, in ms. with a doppler cardiotocograph (Hewlett-Packard, Model 8030) and stored in an Apple Be computer. Cardiac activity was monitored via the real-time audio and video displays and a hardcopy tracing (in bpm). Procedures. The women sat in a comfortable semireclined position and were made to feel at ease while the transducer was positioned to record fetal heartbeats. The two or three researchers who were present monitored cardiac activity for indications that the fetus had entered a quiet state. Behavioral states of 37.week-old fetuses can be defined by the same systematic covariations among eye movements, body movements, respiration movements, and cardiac activity that are used to define behavioral states in neonates (e.g., Nijhuis, Prechtl, Martin, & Bots, 1982; Prechtl & O’Brien, 1982). During periods of behavioral activity, fetal state 2F according to Nijhuis et al. (1982) there is high variability (HV) in ongoing heartrate (range > f4 bpm) and there are occasional abrupt movement-induced accelerations. During quiescent periods, fetal state IF, vatiability in ongoing heartrate is low (range < k3 bpm) and there are no abrupt movement-induced accelerations. Stimuli were presented only when the researchers decided that fetal heartrate was in a low variability (LV) period. A maximum of six stimulations were planned. Half the fetuses were assigned to one stimulus order (TCTCCT) and half to the reverse order (CTCTTC). Half the fetuses within each order had been exposed to one of the rhymes and half to the other. Each stimulation began at the beginning of a rhyme and lasted 30 s. Successive stimulations were delivered when at least 30 s had elapsed since the end of the last stimulation, and the fetus was judged to be in a LV period. Datu Screening. After all fetuses were tested, three observers (A.J.D., J.P.L., and R.M.) independently inspected each hardcopy record in its entirety. They partitioned the entire record into LV periods, HV periods. or neither (e.g., the hardcopy tracing was unreadable) without regard to stimulus presentation and without knowledge of the subject’s target or order of stimulation. Heartrate data were accepted for analysis only if all three independent observers had judged that the fetus was in a LV period during the 15-s prestimulus period and subsequent 30-s stimulus period. If a target (or control) period was eliminated for any reason, then the paired control (or target) period was also eliminated. For example, if the record corresponding to the second control stimulation was rejected, then the portion corresponding to the second target stimulation was automatically rejected. These screening procedures maximized the likelihood of analyzing data only from LV periods, minimized the likelihood of biasing the selection of data, and maintained within-subject equality in the number and ordinal posttion of target and control stimulations for statistical purposes. These procedures are similar to those used in other fetal studies (e.g., Lecanuet, Granier-Deferre, Cohen, Le HoueTec, & Busnel, 1986; Lecanuet et al., 1987, 1988. 1992). accepted for analysis. According to the logs, the 17 mothers had recited their targets an average of 78 times over the preceding 26 days. The number of acceptable stimulations obtained from each of the 17 fetuses varied. Six fetuses provided data from all three pairs of target/control stimulations, 2 fetuses provided data from two pairs of stimulation, and 9 fe-tuses provided data from one pair of stimulations. Ten of the 17 had encountered the TCTCCT order, and 6 of them had La Poulette as the target. Seven fetuses had encountered the CTCTTC order, and 4 of them had La Poulette as the target. Successive stimulations were separated, on average, by 7.5 s (range = 30-150 s). The interbeat intervals were passed through an algorithm which filtered out artifacts and converted the interbeat intervals within successive l-s periods to bpm. Thus, the data consisted of artifact-free heartrate in bpm for each second of the 15-s prestimulus period and 30-s stimulus period. Prestimulus Conditions We first examined the initial prestimulus periods. The mean pre-target heartrates of the 17 fetuses ranged from 124 to 155 bpm (M = 136.5 bpm, SD = 7.75), and the standard deviations ranged from 0.93 to 3.77 bpm (M = 2.20 bpm, SD = 0.79). The mean pre-control heartrates ranged from 125 to 153 bpm (M = 135.9 bpm, SD = 7.67), and the standard deviations ranged from 0.90 to 3.77 bpm (M = 2.30 bpm, SD = 0.78). The pre-target and pre-control heartrate levels did not differ, t( 16) = 0.9 1, nor did the standard deviations, t(16) = -0.28. Pre-target and pre-control heartrate levels were correlated, r(16) = .94, p < .005, but the standard deviations were not, r( 16) = .34, p > .05. Importantly, heartrate levels and standard deviations were not correlated during the pre-target period, r( 16) = .23, p > .05, nor during the precontrol period, r.( 16) = .17, p > .05. Thus, any systematic difference in reactions to targets and controls cannot be attributed to differences in heartrate that existed during baseline. RESULTS Stimulus Effects Data For each subject, the mean heartrate of the 15-s prestimulus period was subtracted from the mean heartrate of successive 5-s intervals of the stimulus period. These difference scores were analyzed with a mixed analysis of variance (ANOVA) program (BMDP2V, version Eleven fetuses were eliminated from consideration. Two fetuses could not be recorded, 1 mother failed to return her log, and 8 fetuses were judged to have been in HV periods during all stimulations. Data from 17 fetuses were 162 DeCasper pc90) which provides conventional and adjusted probability estimates of F statistics for repeated measures (Hertzog & Rovine, 1985; Kisilevsky & Muir, 1991). The first betweensubjects factor, acceptable stimulations (2 vs. 4/6), assessed whether fetuses who had the minimum number of acceptable stimulations reacted differently from fetuses who had more. The second, acoustic structure of the target (La Paulette vs. Le Petit Ctupaud), assessed whether reactions differed as a function of the acoustic properties of the targets. The first within-subjects factor, seconds of stimulation (l-5 vs. 6-10 vs. I I-IS vs. 26-30), assessed whether difference scores changed across the 30-s stimulus period. The second, prior exposure (target vs. control), assessed whether reactivity was influenced by maternal recitation of a rhyme. Acceptable stimulations had no effect, I;( I, 13) = 0.18, nor did it interact with any other factor, 17 values of all interaction terms > IS. Acoustical structure of the target had no effect. F( I, 13) = 0.43, nor did it interact with any other factor, 11values of all interaction terms > .lS. Seconds of stimulation had no effect, F(S, 65) = 1.30, p > .2S. nor did it interact with any other factor, all interaction /> values > 38. Only the main effect of prior exposure was statistically reliable: The average difference score elicited by the targets (M = -1.25 bpm, S1> = I .70) differed from the average elicited by the controls (M = 0.54 bpm. SD = I .92), F( I, 13) = 7.20, /I < .02. Prior exposure did not interact with any other Factor, all interaction /I values > .lS (see Figure I).? In addition. the target-clicited decrease was reliably below its baseline, t( 16) = -3.03, p < .()I; Wilcoxon’s T = 23, p < .OIS, whereas heartrate during control stimulation did not differ from its baseline, t( 16) = l.s7,/~>.os;T=ss./~>.os. Some Post-Hoc Considerations A. Evidence for some effect of repeated stimulation was sought by analyzing difference scores of the 6 fetuses who yielded data from et al. -13 -e --3 3 TIME E’ROM STIMULUS 8 13 ONSET 18 (5 23 SEC 28 INTERVALS) Figure 1. Average difference scores (5-s M - 15-s prestimulus M) at consecutive 5-s intervals b innin 15 s before stimulus onset and ending at stimu 7 us o P set for targets (filled) and controls (open). The vertical lines represent f one standard error. all three pairs of target/control stimulations. Factors of the repeated measures ANOVA were stimulation number (1st vs. 2nd vs. 3rd). prior exposure. and seconds of stimulation. Here, too, the only statistically reliable effect was that of prior exposure, F‘( I. 5) = 5.44, 1~< .07. Other attempts to find some effect of‘ stimulus repetition also failed to do so. H. Data from IO fetuses who were judged to have been in a HV period during testing with a target and its paired control were analyzed. First, we detcrmincd that pre-target and precontrol heartrates were equivalent, and then we conducted a repeated measures ANOVA of their difference scores. There was no effect of prior exposure. E‘(I, 9) = 1.33, 17 > .OS: The average difference score occurring with the tarb<yet (M = 0.44 bpm. .SII = I .2X) did not difl’ci from that occurring with the control (M = -0. I9 bpm. Sl> = 1.36). There was no effect 01‘ seconds of stimulation. F(S. 35) = 2.04. 17 > IS. Thus, no familiarity effects were observed during HV periods. C. Reactions during HV and LV periods were compared after determining that the prestimulus heartratc means did not differ. t(X) = <I .O. but that the standard deviations did, t(X) = 4.50, 1~ < .0002. Indeed, the prestimulation variance of HV periods wa\ almost thrco times that of LV periods which corroborates the obcrservers’ judgments of whether a fetus WBS in a LV or HV period. The mixed ANOVA had hcartrate variability (LV vs. HV) as the 163 Fetal Speech Perception between-subjects factor. Only the heartrate variability x prior exposure interaction was statistically reliable, F( 1, 26) = 5.51, p < .03. Target-elicited reactions during LV were more negative than during HV, and control reactions during LV and HV did not differ. DISCUSSION An important implication of the prenatal experience hypothesis was confirmed: Fetal heartrates decreased in response to stimulation with target rhymes their mothers had recited during the previous 4 weeks but did not change in response to stimulation by control rhymes their mothers had never recited. The different reactions occurred with important variables held constant or counterbalanced, and the direction and magnitude of the target-elicited change were consistent with existing research. Therefore, we conclude that fetal reactions to the target were influenced by previous exposure to the target. replicates This result systematically DeCasper and Spence’s (1986) finding with newborns that target stimuli were more potent reinforcers than control stimuli. In that study, mothers had recited the target throughout the last 6 weeks of pregnancy. Here, mothers began reciting about 6 weeks before term but stopped after 4 weeks. Thus, this study extends our knowledge of the amount and timing of prenatal speech experience that is sufficient to influence the fetus’s/infant’s subsequent perception of speech sounds. Although neither this study nor that of DeCasper and Spence (1986) allows determining which acoustic characteristic(s) of the targets were relevant for the familiarity effects, they do permit deciding which characteristics were not necessary. First, familiarity with maternal voice cues cannot have been necessary for the target effects because test stimuli were recorded by other women. Second, because two French targets and three English targets have been used, any acoustic characteristics specific to either French or English cannot have been necessary for the familiarity effects. Third, because the five passages serving as targets also served as controls, the unique acoustic characteristics of any one passage cannot have been necessary for the familiarity effects. Rather, it seems likely that some set(s) of characteristics that can acoustically define any well-spoken passage from any lan- guage is capable of supporting prenatal familiarity effects like those observed to date. Such characteristics might include intonation contours and/or patterns of syllabic beats and/or patterns of syllables (e.g., Cooper & Aslin, 1990; Femald & Kuhl, 1987; Lecanuet et al., 1987; Mehler, Bertoncini, Barriere, & JassikGershenfeld, 1978; R.K. Panneton, personal communication, November 15, 1985; Querleu et al., 1988). Differential reactivity to the target and control was seen during LV periods but not during HV periods. This interaction between state and prior experience is reminiscent of the interactive effects of state and stimulus characteristics (Lecanuet et al., 1988). Here, too, the interaction may indicate that the small heartrate decrease elicited by a low-intensity familiar stimulus was easier to detect during LV periods than during HV periods. This study shows that prenatal exposure to the “mother’s tongue” can enhance fetal reactions to linguistically important speech sounds and, thus, could promote language-relevant perceptual tuning before birth. More analytical studies of how prenatal exposure to speech and nonspeech sounds affects subsequent perception will substantially influence our conception of perceptual development, language development, and language. REFERENCES Abrams, R., Gerhardt, Transmission K., & Peters, A. (1992, September). of sound and \ihrczrion to the fetus. Paper presented at the Conference on Fetal Development: A Psychobiological Perspective, Paris, France. Benzaquen, S., Gagnon, R., Hunse, C., & Foreman, J. (1990). The intrauterine sound environment of the human fetus during labor. Amrrkan Jourrwl of Olntetrics and Gyner~olqqy, 163, 484489. Busnel, M.-C., & Granier-Deferre. C. (1983). And what of fetal audition? In A. Oliveirio & M. Zappella (Eds.), The hrhaior ofhuman infunrs. New York: Plenum. Cooper, R.P.. & Aslin, R.N. (1990). Preference for infant directed speech in the first month after birth. Child Development. 61, 1584-1595. Coopersmith, R., & Leon, M. (1984). Enhanced neural responses to familiar olfactory cues. Sc~icvre. 225. 849-85 I. DeCasper. A.J., & Fifer. W.P. (1980). Of human bonding: Newborns prefer their mothers’ voices. %ienw. 208. 1174-l 176. DeCasper, A.J.. & Spence, M.J. (1986). Prenatal maternal speech influences newborns’ perception of speech sounds. lnfunt Behoviw und Development. 9. 133-150. DeCasper et al. 164 Dember, W.N. ( 1960). The ps~chulo,q~ r$/~c~(‘cpfio~z. New York: Holt, Rmehart, & Winston. Femald, A., & Kuhl, P. (1087). Acoustrc determinants ot infant preference for motherese speech. In~urzf Behavior and Devrlopmenr, IO, 279-293. Hertrog, C.. & Ravine, M. (1985). Repeated-measures analysis of variance in developmental research: Selected issues. Child Dewlopmrnf. 56, 787-X09. Jacoby. L.L.. & Dallas, M. (198 I ). On the relation between autobiographical memory and perceptual learning. .IOUI-nal of E.xperimental P.\whology Ge~wrol.I10. X06-340. Jacoby, L.L., & Witherspoon, D. (19X2). Remembering without awareness. Canadim .loumul of Pvyc~holo,sy, 36, 3OG324. Kisilevsky. B.S., & Muir, D.W. (1991). Human fetal and subsequent newborn responses to sound and vibration. Infant Behavior und De~&pmenr. 14, l-26. Lecanuet, J.-P., Granier-Deferre, C., & Busnel, M.-C. (198X). Fetal cardiac and motor responses to octave-band noises as a function of central frequency. intensity, and heartrate variability. Early Humcm Dcwlopmmt, Lecanuet, IX, 81-93. J.-P., Granier-Deferre. C., & Busnel, M.-C. (I 98’)). Differential fetal auditory reactiveness as a function of stimulus characteristic and state. Srminu~ rn PrGzaroln~ypv. /3(S), 421-429. Lecanuet, J.-P., Granier-Deferre. C.. Cohen, H.. Le Houerec, R., & Busnel, M.-C. (1986). Fetal responses to acoustic stimulation depend on heart rate variability pattern, stimulus intensity and repetition. Ear& Human Der~cs/opmc~nr.13. 269%2X3. Lecanuet, J.-P., Granicr-Defcrre, C., DeCasper. A.J.. Maugeais, R.. Andrieu, A.-J.. & Buynel, M.-C. (1987). Perception et discrimination foetale de <timuli langagierx mise en evidence a partir de In rcactivite cardiaque. Resultats preliminairc\ [Fe- tubes’ perception and dihcrrmination of language stimuli as indexed by cardiac reactivity: Prclimnary results]. Comptc-Rc~nduv LIP I’Ac~trdrmic~ t/es S?ienc~es dc furis. Srria 111(t.305)16 I- 164. Lecanuet. J.-P.. Granier-Deferrc. C., Jacquet, A.-Y., & Busnel, M.-C. (1902). Dccelerative cardiac respon\ivcness to acoustical stimulation in the near term foetuc. fJuuuw!\ ./ourm~l of’E.tpc~~imentu/ P.vwhr& “,$?‘, ‘Mb, 279-303. Mehler. J., Bertoncini. J., Barriere, M.. M Ja\aikGershenfeld. D. (1978). Infant recognition of mother’\ voice. PcrwpGon. 7, 4Yl497. Nijhuis. J.G., Prechtl, H.F.R., Martm. C.B.. & Bats, R.S.G.M. (1982). Arc there behavioral states In the human fetus? .I%~/~ /f~mrm Lkdqmcnt. 6. 177-19s. Prcchtl, H.F.R.. & O’Brien. M.J. (1982). Behavioral states of. the full-term newborn: The emergence of a ~011~ cept. In P. Stratton (Ed.), Psvc~hohiolqq of Ihe hrtmcrn rwwhorn. Chichester: Wiley. Querleu. D.. Renard, X.. Versyp. F., ParIs-Delrue, Ia., Kr Crepin. G. (I YXX). Fetal hearing. Eur-open ./ortr-ncrl of Ohstr,tric,.s & G~r~ec~o/o,q~ crnd Repr-otluc~tiw Bio/o,cg, X, 191-212. Richard\, D., Frcntzen, B.. Gerhardt, K., McCann. M.. & Abram\. R. (lYY2). Sound lcvelr in the human uterus. Ohsrc~tric~s ud G~rwolo,q,v. HO.I X6-l 90. Solomon. R.L., & Postman, 1.. (1052). The frequency of usage as a determinant of recognition threshold\ for words. ./ofr0xr/ 0f /?\/x~rinlentcr/ Ps~~+o/r>,q~. -l.i, 195-20 I Warren. R.M. ( I YX2). Autlitot:v pw~~q~fion. A mw s!mlusis. NW York: Pergamon. 2 October 1992; Revised 3 February 1993 n