Chemical Geology 267 (2009) 175–184
Contents lists available at ScienceDirect
Chemical Geology
j o u r n a l h o m e p a g e : w w w. e l s ev i e r. c o m / l o c a t e / c h e m g e o
Routine isotopic analysis of iron by HR-MC-ICPMS: How precise and how accurate?
Nicolas Dauphas a,b,⁎, Ali Pourmand a,b, Fang-Zhen Teng a,b,1
a
b
Origins Laboratory, Department of the Geophysical Sciences, the University of Chicago, 5734 South Ellis Avenue, Chicago IL 60637, USA
Enrico Fermi Institute, the University of Chicago, 5640 South Ellis Avenue, Chicago IL 60637, USA
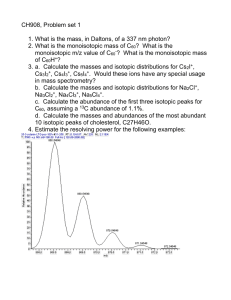
a r t i c l e
i n f o
Article history:
Received 14 June 2008
Received in revised form 16 November 2008
Accepted 5 December 2008
Keywords:
Iron isotopes
HR-MC-ICPMS
Chromatography
Precision
Accuracy
a b s t r a c t
High-temperature isotopic variations documented in natural materials span a narrow range and call for
precise and accurate isotopic analyses. Precisions better than ~ 0.03‰ (95% confidence interval) for δ56Fe
have been obtained by High-Resolution Multi-Collector Inductively Coupled Plasma Mass Spectrometry (HRMC-ICPMS). Whether these uncertainties encompass all sources of error has not been carefully evaluated.
The current study examines all parameters that can affect accuracy and precision of Fe isotopic
measurements by HR-MC-ICPMS. It is demonstrated that accurate δ56Fe measurements can be routinely
achieved within the quoted uncertainties.
© 2008 Elsevier B.V. All rights reserved.
1. Introduction
The field of Fe isotope geochemistry has seen important developments in methodology and scope since the advent of Multi-Collector
Inductively Coupled Plasma Mass Spectrometry (MC-ICPMS) (see recent
reviews by Anbar, 2004; Beard and Johnson, 2004; Johnson et al., 2004;
Dauphas and Rouxel, 2006). There is now increasing evidence that Fe
isotopic variations are not confined to the realm of biology or low
temperature aqueous geochemistry but can also occur at high
temperature by equilibrium fractionation between minerals and melts
(Polyakov and Mineev, 2000; Polyakov et al., 2007; Dauphas et al., 2007;
Schuessler et al., 2007; Shahar et al., 2008), by diffusion in solid metal
(Mullen, 1961; Roskosz et al., 2006; Dauphas, 2007 and references
therein) and silicate melts (Richter et al., 2009), by evaporation/
condensation (Wang et al., 1994; Alexander and Hewins, 2004; Dauphas
et al., 2004; Tachibana et al., 2007; Zipfel and Weyer, 2007), and by Soret
diffusion (Huang et al., in press; Richter et al., 2009). Considering that the
Fe isotopic compositions of most geomaterials formed at high temperature span a narrow range, important unsettled questions require high
precision and accurate analyses. For instance, the Fe isotopic composition
of the silicate Earth is still uncertain. While basalts have almost
homogeneous δ56Fe around +0.1‰ (Beard et al., 2003; Schoenberg and
von Blanckenburg, 2006; Weyer and Ionov, 2007), mantle peridotites
⁎ Corresponding author. Origins Laboratory, Department of the Geophysical Sciences,
the University of Chicago, 5734 South Ellis Avenue, Chicago IL 60637, USA.
E-mail address: dauphas@uchicago.edu (N. Dauphas).
1
Present address: Isotope Laboratory, Department of Geosciences and Arkansas
Center for Space and Planetary Sciences, University of Arkansas, 113 Ozark Hall,
Fayetteville, AR 72701, USA.
0009-2541/$ – see front matter © 2008 Elsevier B.V. All rights reserved.
doi:10.1016/j.chemgeo.2008.12.011
have variable compositions averaging ~0‰ (Williams et al., 2005;
Schoenberg and von Blanckenburg, 2006; Weyer and Ionov, 2007).
Although increasing the number of repeats (n) in High Resolution
(HR-) MC-ICPMS reduces the uncertainty related to instability in
pffiffiffi
instrumental mass bias and counting statistics (~1 = n), many other
parameters can affect the precision and accuracy of Fe isotopic analyses
(Malinovsky et al., 2003; Schoenberg and von Blanckenburg, 2005).
These include mass fractionation during column chromatography,
incomplete separation of Fe from matrix elements that can cause
instrumental mass bias to vary, presence of direct isobars from 54Cr+ on
54 +
Fe and 58Ni+ on 58Fe+ and imperfect matching in acid molarity or Fe
concentration between samples and bracketing standards.
A major challenge in making precise and accurate measurements
of Fe isotopes arises from incomplete mass separation between Fe and
argides, which include interferences of 40Ar14N+, 40Ar16O+, and
40
Ar16O1H+ on 54Fe+, 56Fe+, and 57Fe+, respectively. Several strategies
have been employed to address this analytical difficulty and are briefly
discussed below. (i) The first method employed for isotopic analysis of
Fe by MC-ICPMS relies on desolvating nebulization to increase the
ratio of Fe/argide (Belshaw et al., 2000; Anbar et al., 2000; Zhu et al.,
2002, de Jong et al., 2007). Nevertheless, this brute-force method is
incapable of eliminating all interferences and careful matching
between standard and sample ion intensities during bracketing is
essential. (ii) The GV IsoProble MC-ICPMS is equipped with a hexapole
collision cell, which is primarily used to thermalize the incoming ions
before introduction into the magnetic sector. It also serves the purpose
of breaking down molecules such as argides through interaction with
the collision gas, a mixture of Ar and H2 for Fe isotopes (Beard et al.,
2003; Rouxel et al., 2003; Dauphas et al., 2004). (iii) Operating the MCICPMS at reduced power (cold plasma) can also drastically reduce
176
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
argide interferences (Kehm et al., 2003). The main drawback of this
technique, however, is that it promotes formation of CaOH+, which
can interfere with 57Fe+ peak. (iv) Medium to high mass-resolving
power on the Thermo Scientific Neptune, Nu-Plasma HR, and Nu
Plasma 1700 are used to separate argide interferences from Fe isotopes
(Malinovsky et al., 2003; Weyer and Schwieters, 2003; Arnold et al.,
2004; Poitrasson and Freydier, 2005; Schoenberg and von Blanckenburg, 2005; Williams et al., 2005; Borrok et al., 2007). Precisions of
better than 0.04‰ are achievable with this method.
In the current study, we evaluate the variables that can affect the
accuracy of Fe isotope measurements using HR-MC-ICPMS. Error bars
in Fe isotope geochemistry have been traditionally ascribed based on
either the long-term external reproducibility of geostandards or the
dispersion of replicate analyses of a solution during a session of
measurements. In the following, we present a method, which is also
applicable to other isotope systems, to take into account both sources
of analytical uncertainties. We demonstrate that δ56Fe can be
measured accurately at a precision of ~ 0.03‰ in rocks and minerals.
2. Chemical separation of iron
All chemical protocols used for Fe isotopic analyses rely on the
partition behavior of Fe with anion-exchange resin in HCl medium
(Strelow, 1980). Matrix and interfering elements are eluted with
concentrated HCl while Fe, which forms strong anion chloridecomplexes, is retained on the resin. Iron is subsequently eluted at
lower HCl molarity. Protocols using AG MP-1 anion exchange resin can
be found in Maréchal et al. (1999), Archer and Vance (2004), Brantley et
al. (2004), Borrok et al. (2007) and de Jong et al. (2007). Protocols using
AG1-X3, -X4 or -X8 are described in Rouxel et al. (2003), Dauphas et al.
(2004), Poitrasson and Freydier (2005), Schoenberg and von Blanckenburg (2005), Dideriksen et al. (2006), and Thompson et al. (2007).
Here we used the separation protocol described by Dauphas et al.
(2004, 2007). Typically 2–10 mg of powdered sample is subjected to the
sequence of acid addition and heating and evaporations presented in Table
1. The purification procedure of column chromatography is repeated twice
to ensure complete elimination of the matrix. After elution of Fe from the
second column, the solution is dried down under a heat lamp, taken up in
~100 μL of 15.3 M HNO3 and evaporated to dryness to eliminate chloride
complexes. The dried Fe residue is finally dissolved in 10 mL of 0.3 M HNO3
(an ultrasonic bath is often used at this step to ascertain complete
dissolution). The 0.3 M HNO3 comes from a large batch that also serves for
preparation of IRMM-014 and blanks used in standard bracketing and on
peak zero correction. This ensures perfect matching in acid molarity
between all analyzed solutions, which is critical for accurate isotopic
analyses. At this point, Fe is ready for isotopic measurement by MC-ICPMS.
It must be noted that the chromatographic procedure described herein
does not allow for quantitative separation of Cu and Zn from Fe, which can
Table 1
Sample dissolution and Fe column chemistry protocols.
•
•
•
•
•
•
•
•
•
•
•
•
Addition of 1 mL of 28.8 M HF + 0.5 mL 15.3 M HNO3 + 1 drop 11.3 M HClO4
Heating in close beakers at 100 °C for 5 h
Evaporation, addition of 0.75 mL of 10.2 M HCl + 0.25 mL 15.3 M HNO3 + 1 drop
11.3 M HClO4
Heating and evaporation
Addition of 1.0 mL of 10.2 M HCl + 0.5 mL 15.3 M HNO3 + 1 drop 11.3 M HClO4
Heating and evaporation. Addition of 500 μL 6 M HCl.
Disposable Bio-Rad Poly-Prep polyethylene columns filled with 1 mL of AG1-X8 200400 mesh Cl-form anion exchange resin (resin bed length 2.0 cm, diameter 0.8 cm)
The resin is conditioned with 10 mL of H2O, 5 mL of 1 M HNO3, 10 mL of H2O, 10 mL
of 0.4 M HCl, 5 mL of H2O, and 2 mL of 6 M HCl
The samples in ~ 250 μ L 6 M HCl are loaded on the column
Most matrix and interfering elements are eliminated with 8 mL (0.5+0.5 + 1 + 2 + 4)
of 6 M HCl.
Iron is recovered in a Teflon beaker by passing 9 mL (0.5+0.5+1+3+4) of 0.4 M HCl.
The solution is dried down and the column procedure is repeated a second time with
new resin.
be a problem for certain matrices, such as sulfides. To mitigate this
problem, Schoenberg and von Blanckenburg (2005) introduced an iron
hydroxide precipitation step by basification with NH4(OH).
3. Mass spectrometry
Iron isotopic analyses were performed with a Thermo Scientific
Neptune HR-MC-ICPMS (see Wieser and Schwieters, 2005 for a
description of the instrument). The mass-resolution of the instrument
is controlled with a set of three switchable slits (250 μm, 30 μm, and
16 μm width) located before the electrostatic analyzer. Measurements
were performed in both medium-resolution (MR) and high-resolution
(HR) modes. In these two modes, the Fe+ peaks are resolved from argide
interferences as flat-topped shoulders on the low mass side of argide
peaks (Weyer and Schwieters, 2003). The mass resolution is defined as
m/(m0.95 −m0.05), where m0.95 and m0.05 are the masses measured at
5% and 95% intensity peak heights (see Weyer and Schwieters, 2003,
for details). Mass resolutions are equal to ~ 8500 and ~ 11,000 for MR
and HR, respectively. The ion transmission efficiency of the instrument for Fe is reduced by a factor of ~8 from LR to MR and another factor
of ~2 from MR to HR. The δ notation (in ‰), δiFe= [(iFe/54Fe)sample /
(iFe/54Fe)IRMM-014 − 1] × 103, is used throughout the current study.
IRMM-014 is a pure Fe metal reference material (Taylor et al., 1992) that
has a Fe isotopic composition identical to chondrites (Dauphas and
Rouxel, 2006). Unless otherwise noted, the pair of isotopes reported is
56
Fe/54Fe (i = 56). In the course of this study, the stock solution of IRMM014 used during sample-standard bracketing was renewed. The δ56Fe
and δ57Fe of the new batch of IRMM-014 normalized to the old batch are
−0.004 ± 0.009‰ and 0.001 ± 0.015‰ (95% ci), respectively. This
confirms the conclusion of Dauphas et al. (2004) that IRMM-014 has
homogeneous isotopic composition at the 0.01‰ level.
Except for a series of tests aimed at estimating the influence of acid
molarity on isotopic ratios, all analyzed solutions were in 0.3 M HNO3.
An enclosed SC-2 sample changer installation (autosampler)
equipped with an ULPA filter unit was used throughout this study.
Most analyses were performed with the Thermo Scientific Stable
Introduction System (SIS), which combines quartz cyclonic and Scotttype spray chambers. A few tests were also performed with the ApexQ + Spiro TMD desolvating inlet system from Elemental Scientific Inc.
operated with addition of N2 (~10 mL/min). Introduction of N2
increases the sensitivity of the Apex + Spiro inlet system but it also
increases the level of interferences on 54Fe+ from 40Ar14N+. The
temperatures of cooler (multipass condenser) and heater (cyclonic
spray chamber) were set at 2 °C and 100 °C, respectively. A 50–100 μL/
min PFA Teflon self-aspirating micronebulizer was used with both SIS
and Apex-Q. The virtue of the Apex-Q + Spiro relative to SIS is that it
drastically reduces oxides and increases Fe sensitivity. However,
instrumental mass fractionation is less stable and achieving optimal
tuning conditions is more daunting with the Apex-Q + Spiro than with
the SIS. Therefore, using the SIS is recommended unless low quantities
of Fe are analyzed. The sampler and skimmer cones are made of Ni, yet
the contribution of 58Ni from the cones on 58Fe is negligible (Dauphas
et al., 2004). Two cone geometries are available for the Neptune; the
standard (H) and high-sensitivity (X). X-cones can increase transmission by a factor of 3 relative to H-cones (Weyer and Schwieters, 2003),
which can be important for samples containing little Fe. H-cones were
exclusively used in this study.
Instrumental mass fractionation is large in MC-ICPMS (between
3.5 and 5.5%/amu for Fe, see Fig. 2 of Dauphas and Rouxel, 2006).
Excluding double spike (Johnson and Beard, 1999; Balci et al., 2006;
Dideriksen et al., 2006; Fantle and Bullen, 2009), three strategies have
been developed for correcting instrumental mass fractionation during
MC-ICPMS analysis: (i) Ni-doping, (ii) Cu-doping, and (iii) standard
bracketing (Belshaw et al., 2000, Malinovsky et al., 2003; Arnold et al.,
2004, Dauphas et al., 2004; Schoenberg and von Blanckenburg, 2005;
also see Albarède and Beard, 2004, for a detailed review of this topic).
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
Instrumental mass bias in MC-ICPMS is adequately described by the
exponential law (Maréchal et al., 1999), which relates the measured
isotopic ratio (r) to the true ratio (R) and the relative mass difference
of the isotopes (△m/m):
β
r = Rð1 + Δm=mÞ ;
ð1Þ
where β represents the instrumental mass bias and is a free
parameter. In standard-bracketing with element doping, a fixed
amount of Ni or Cu of known isotopic composition is added to the
sample and the standard solutions. β is calculated for Ni or Cu using
Eq. (1). The fractionation corrected ratio for Fe is subsequently
computed from the measured ratio assuming that βFe = βNi,Cu. In
reality βFe ≠ βNi,Cu (e.g., Maréchal et al., 1999; Woodhead, 2002;
Archer and Vance, 2004) and the exponential law may not be
completely adequate in describing instrumental mass fractionation
(e.g., Vance and Thirlwall, 2002; Wombacher and Rehkämper, 2003).
It is therefore critical to bracket sample analyses by measurements of
the IRMM-014 Fe reference material doped with the same amount of
Ni or Cu and reduced in the same manner. In standard bracketing
without element doping, the instrumental mass bias is computed for
the two reference standard 56Fe/54Fe analyses (βSTD1 and βSTD2 using
R = 15.6986 in Eq. (1); Taylor et al., 1992) that bracket the sample. The
average of these two values is used to correct the sample analysis for
instrumental mass fractionation (βSAMPLE = 0.5βSTD1 + 0.5βSTD2). In
both cases, the isotopic ratios are then reported in δ-notation relative
to IRMM-014.
The advantage of the Ni-doping method is that both Fe and Ni (60
and 61) isotopes can be measured at the same time, i.e., no peak
jumping is involved. In addition, Ni is easily separated from Fe during
the column chemistry. Poitrasson and Freydier (2005) pointed out
that keeping the 61Ni+ signal above 500 mV is essential for precise
analyses because uncertainty in the measurement of 61Ni (1.13% of
total Ni) propagates unfavorably to the corrected Fe isotope ratios.
The main drawback of this method is that it generates a major
interference on 58Fe (0.2819 atom % of total iron) from 58Ni
(68.27 atom % of total Ni). This is not a problem if the laws of massdependent fractionation solely govern variations of isotopic ratios.
However, non-mass-dependent isotopic variations may arise from
photochemistry in aqueous systems or incomplete mixing of the
products of stellar nucleosynthesis in meteorites (Völkening and
Papanastassiou, 1989; Dauphas et al., 2004, 2008). Another difficulty
is that while Fe and Ni isotopes can be measured at the same time, our
collector setting does not allow 53Cr+ to be measured simultaneously
with 61Ni+ and the contribution of 54Cr+ on 54Fe+ cannot be
controlled. For these reasons, this scheme was not utilized (Malinovsky et al., 2003, Schoenberg and von Blanckenburg, 2005;
Poitrasson and Freydier, 2005, provide ample details on how to
implement this correction scheme).
On the other hand, Sharma et al. (2001), Kehm et al. (2003), and
Arnold et al. (2004) advocated the use of Cu-doping. Nevertheless, the
mass-dispersion in the collector array of the Neptune is not wide
enough to allow analysis of the 4 stable isotopes of Fe and 2 isotopes of
Cu (63 anf 65) at the same time. Two sequences must therefore be
used, making peak jumping unavoidable. As is the case with the Ni
doping scheme, isotopic analyses with Cu-doping are potentially less
sensitive to matrix effects, which can improve accuracy. However, the
duration of the measurements is significantly extended, more Fe is
consumed, and in Cu-rich samples, one must ascertain complete
removal of Cu during the chemistry. Although tests using Cu-doping
were performed on selected samples to validate the quality of the
measurements, the standard-bracketing method was preferred for
most tests and measurements (also see Schoenberg and von
Blanckenburg, 2005).
The Neptune MC-ICPMS at the University of Chicago is equipped
with 9 Faraday collectors connected to an array of one 1012 Ω, seven
177
1011 Ω, and one 1010 Ω amplifiers. With the standard-bracketing
method, 54Fe, 56Fe, 57Fe, and 58Fe were measured on L2, C, H1, and H2
Faraday collectors. Possible isobaric interferences from Cr and Ni were
monitored at masses 53Cr and 60Ni on L4 and H4 Faraday collectors,
respectively. All measurements were performed in medium and high
mass resolution. Isobaric interferences of 40Ar14N, 40Ar16O, 40Ar16O1H,
and 40Ar18O on 54Fe, 56Fe, 57Fe, and 58Fe, were resolved by measuring
iron isotopes as flat-topped peak shoulders. The ion intensities of 53Cr
and 60Ni were measured at the center of the peaks. High-intensity
measurements of minor isotopes became possible by measuring the
high-abundance 56Fe+ with a 1010 or 1011 Ω amplifier. The low
intensity 60Ni+ signal, on the other hand, was measured with a 1012 Ω
amplifier (settling time ~3 s) to reduce the Johnson noise/signal ratio
(Wieser and Schwieters, 2005). The washout time with 0.45 M HNO3
between samples and standards was 90 s. The take-up time was 60 s.
The mass spectrometer baseline was measured with the beam
defocused for 30 cycles of 1.05 s each. Before a sequence of analysis,
a clean 0.3 M HNO3 solution was measured under conditions identical
to sample measurements (e.g., same number of cycles and integration
time). The ion intensities of this solution were subtracted from all
sample/standard measurements (On Peak Zero, OPZ). The idle time
was set to 3 s. Iron isotopes were acquired in a single block consisting
of 25 cycles of 8.389 s each. The Fe concentrations of samples and
standards ranged between 2 to 5 ppm at a sensitivity of 5 to 13 V/ppm
in MR on 56Fe with a 50–100 μL/m nebulizer.
All measurements for the Cu-dopping tests were made in HR. The
same amounts of pure Cu were added to the sample and standard
solutions to reach a constant concentration of ~ 5 ppm, with a
sensitivity of 2 V/ppm on 63Cu. The sample δFe values were computed
relative to bracketing IRMM-014 also doped with the same level of Cu.
It is well documented that βFe ≠ βCu so sample-standard bracketing is
critical. The measurements were performed in 25 cycles of 2
sequences. The first sequence was identical to the standard-bracketing
method and a 3 s idle time was used between each sequence. In the
second sequence, 63Cu and 65Cu were measured on C and H2 Faraday
collectors. The integration times were 8.389 s for both Fe and Cu
sequences. The relative mass dispersion between 65Cu and 63Cu was
different from that of 58Fe and 56Fe, which was corrected by adjusting
the dispersion lens to ~ − 75 V. To correct aberrations in the Cu peak
shape, the focus lens was also adjusted to ~10 V, which resulted in
square peak shapes appropriate for Cu isotopic measurement. The
lenses switched between Fe and Cu mode on a timescale of ~ 10 s. No
peak centering was required during the analysis because hysteresis
was not observed at any time. In other words, the center of peak
measurement consistently returned to the appropriate position on the
flat-topped iron peak shoulder.
The standard-sample sequences were repeated n times (typically
5 ≤ n ≤ 9). Data processing was performed offline. In a sequence
consisting of standard (i− 1), sample (i− 1), standard (i), sample (i),
standard (i+1), sample (i+1), the isotopic composition of the ith sample
repeat is δFesample(i) =[Rsample (i)/(0.5⁎Rstandard (i) +0.5⁎Rstandard (i + 1))−
1]×103. The reported isotopic compositions are the averages of n repeat
analyses,
δFesample =
n
X
δFesampleðiÞ = n:
ð2Þ
i=1
δFe values for standards were also computed by considering each
standard i as a sample bracketed by 2 standards (i − 1 and i + 1).
Because sample-standard bracketing is equivalent to a linear time
interpolation, inaccurate δFe (≠ 0 for standards bracketed by
standards) could arise from curvature in instrument drift with time,
which was not observed. The dispersion of the standard-bracketed
standard δFe was used to estimate the instrumental uncertainty (σ) of
the sample measurements by assuming that σsample =σstandard. The
main virtue of using standards rather than samples to quantify the
178
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
the number of standard brackets (m) is 44. The uncertainty on the
sample δFe analysis is therefore:
2
σ MassSpec =
m h
i2
1X
δFestandardðiÞ = m;
n i=1
ð3Þ
where n represents the number of sample measurements, as we are
interested in the uncertainty of the average of n. For n = 9 replicate
analyses of the sample solution, σMassSpec is the standard deviation of all
the standard δFe values measured in that session divided by 3
pffiffiffi
( n). Instrumental uncertainty only represents a part of the total
uncertainty, which must be assessed by examining the external
reproducibility of the analyses.
4. Optimization of isotopic analyses
Several parameters such as the peak position, acid molarity, Fe
concentration, presence of matrix elements or isobaric interferences
can influence the quality of isotopic analyses. We explored these
factors in order to establish a robust and optimized method that can
be used routinely for high-precision Fe isotope measurements.
4.1. Shoulder peak plateau in medium-resolution (MR) mode
Fig. 1. Test of the peak flatness and interference tailing on Fe isotope measurements in
MR using the SIS and 5 ppm Fe solutions in HNO3 0.3 M. The top panel (A) shows peak
scans at masses 54, 56, 57, and 58 (on L2, C, H1, and H2). The voltages of some curves
were multiplied by an arbitrary factor for clarity purposes (provided in the legend). The
vertical lines represent the masses where isotopic measurements were made. All
measurements were normalized by bracketing with measurements taken at mass
55.730. The bottom panels (B) show the Fe isotopic compositions at all masses relative
to the composition measured at mass 55.730.
instrumental uncertainty is that there are many more standard
analyses than samples and the standard deviation is better known. For
instance, in a session of 5 sample analyses repeated 9 times each,
Malinovsky et al. (2003) tested how the flatness of the peak
shoulder in HR mode can affect the precision and accuracy of isotopic
analyses. They showed that highly reproducible results can be
obtained over a mass range of ~0.01 provided the Faraday cups are
properly aligned. Iron isotopic analyses can degrade the HR slit very
rapidly by ion milling and thereby reduce the mass resolution.
Because the MR slit blocks a smaller fraction of the incoming ions, it
has a longer lifetime in terms of the number of analyses that can be
performed. Weyer and Schwieters (2003) looked at the same issue in
MR and showed that consistent δ56Fe within 0.06‰ (2σ) ratios can be
obtained over a mass range of 0.005 (δm/m = 100 ppm). In the
following we improve the results reported by Weyer and Schwieters
(2003) to higher precision δ56Fe (0.02 to 0.03‰, 2σ). A 5 ppm solution
of Fe was introduced into the mass spectrometer using the SIS. A
sequence was established where the Fe isotopic composition was
measured alternatively at the center and off-center of the Fe shoulder
peak. The On Peak Zero (OPZ) used for all analyses was taken at the
center (during routine analyses only one OPZ is measured at the
beginning of a sequence). The center was selected visually, as would
be done during a routine analysis. Six off-center measurements were
performed, spanning a mass interval of 0.006 amu. We computed the
δ56Fe, δ57Fe, and δ58Fe values for off-center analyses relative to the
bracketing center measurements. The results are shown in Fig. 1.
Expectedly, as the mass positions approach the argide peaks, tailing
leads to erroneous δ56Fe and δ57Fe values. Uncertainties also tend to
increase, as the position gets further away from the center, due to
fluctuations in argide/Fe ratio and/or peak position. Overall, consistent measurements were achieved with minimum uncertainties of
~0.023‰ for δ56Fe, 0.043‰ for δ57Fe, and 0.187‰ for δ58Fe (2σ) over a
mass interval of 0.003 amu. These observations indicate that it is
preferable to position the mass slightly to the left (~ 0.001 amu) of
what visually appears to be the center of the Fe shoulder peak plateau.
4.2. Concentration matching between samples and standards
With the first generation of MC-ICPMS, where Fe isotopic analyses
depended on the use of a desolvating nebulizer to reduce argide
interferences, it was absolutely crucial to match the concentration of
samples and standards (Belshaw et al., 2000; Anbar et al., 2000; Zhu
et al., 2002). In the Neptune, three strategies help eliminate the effect
of argide interferences. Clearly the most important factor is the use of
MR or HR modes. Subtraction of OPZ ion intensities can also help
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
179
the concentration of the matrix element (this is easily converted to a
ratio to iron by dividing by 1 to 5 ppm). We do not report δ56Fe for the
Cr-doping test because there is a direct isobaric interference of 54Cr on
Fig. 2. Effect of concentration matching on the isotopic composition of Fe. Standard
solutions with mismatched concentrations were analyzed by bracketing with the same
standard at 5 × 10− 11 A (filled circles), 25× 10− 11 A (filled squares), and 38 × 10− 11 A (open
circles) on 56Fe. The solutions were measured using the SIS and Apex-Q in both MR and HR.
No clear correlation is observed between the Fe isotopic composition and concentration
mismatch. Ion intensities of samples and standards are typically matched to within ±5%.
reduce the influence of tailing from argide peaks, if present. Finally,
matching the concentration between samples and standards can also
play a role in improving accuracy. Schoenberg and von Blanckenburg
(2005) studied this aspect by bracketing standard measurements by
analyses of the same standard but with a Fe concentration that was
deliberately mismatched. These authors concluded that δ56Fe was not
affected by mismatch between the concentration of Fe in the standard
and the sample as long as the difference was less than a factor of 2.
Examination of Fig. 4 from Schoenberg and von Blanckenburg (2005)
however reveals a statistically significant positive correlation between
δ56Fe and Csample/Cstandard. We performed similar tests in both MR and
HR modes using the SIS and Apex-Q at different ion intensities of the
reference solution (5, 25 and 38× 10− 11 A on 56Fe). The measurements
reveal that indeed, individual sample analyses do not depart from the
standard isotopic compositions when 0.5 b C/Cstandard b 2 (Fig. 2).
Nevertheless, there seems to be a negative correlation between
δ56Fe and the degree of mismatch between the sample and standard.
In MC-ICPMS, concentration matching within 5% is easily achieved,
which is sufficient to obtain accurate analyses within ~0.03‰ on
δ56Fe. Under these conditions, only one OPZ analysis needs to be made
per session.
4.3. Influence of matrix elements on instrumental mass fractionation
Although our chemistry is highly efficient at eliminating most
common rock forming elements, certain transition elements that are
present in appreciable quantities in Fe-bearing rocks can persist after
chromatography. This is true in particular for Cu, which follows Fe
throughout the chemistry. Such elements can affect the accuracy of
the isotopic analyses by modifying the value and the stability of
instrumental mass fractionation. Schoenberg and von Blanckenburg
(2006) examined the influence of Cu, Zn, Cd, and Co on Fe isotopic
analyses on a Neptune MC-ICPMS. They found that even when the
concentrations of these elements reach 50% of Fe concentration, no
significant deviation of δ56Fe is observed within ~ 0.06‰.
We have extended the range to higher matrix concentrations.
Solutions of 1 to 5 ppm Fe were mixed with up to ~ 100 ppm Co, Ni, Cr,
and Cu. These solutions were then analyzed by bracketing with pure
Fe solutions. The resulting δ56Fe are plotted in Fig. 3A as a function of
Fig. 3. Influence of matrix elements and isobars (Cr and Ni) on the isotopic composition
of Fe. The measurements were made using the SIS in both MR and HR at Fe
concentrations ranging from 1 to 5 ppm in HNO3 0.45 M. The top panel (A) shows the
isobar-free Fe isotopic composition (because of an interference of 54Cr on 54Fe, the
isotopic compositions for Cr-doping experiments are reported as 2 × δ57/56Fe).
Instrumental mass bias for Fe is not affected by the presence of Co, Ni, Cr, and Cu up
to concentrations of ~ 10 ppm. The bottom panels (B) show the effect of the presence of
direct isobars on the isotopic composition of Fe (54Cr and 58Ni interfere on 54Fe and 58Fe,
respectively). 54Cr and 58Ni interferences were monitored and corrected for using 53Cr
and 60Ni (see text for details). The corrections break down for 54Cr/54Fe and 58Ni/58Fe
ratios above ~ 0.01 to 0.1 (corrections of 10 to 100‰, measured with precisions of 0.1‰
for δ56Fe and ~ 0.5‰ for δ58Fe).
180
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
anomalies on δ56/54Fe and δ58/54Fe, which is a practical way of
accounting for the fact that βFe ≠βCr,Ni. It is important to note that the
threshold 54Cr/54Fe and 58Ni/58Fe are well above those obtained in
natural samples after Fe separation chemistry. These results agree with
the study of Schoenberg and von Blanckenburg (2005) who showed that
Fe isotopic analyses are unaffected for 54Cr/54Fe and 58Ni/58Fe ratios up
to 0.001 and 0.3, respectively. Dauphas et al. (2008) reported normal
58
Fe/54Fe analyses in Ni-rich meteoritic materials with a precision of
±0.04‰ after correcting for 58Ni isobaric interference (mainly from
memory in the Apex-Q+ Spiro desolvating nebulizer) and internal
normalization using the 57Fe/54Fe ratio.
4.5. Acid molarity
Fig. 4. Effect of acid concentration matching on the accuracy of Fe isotopic analyses. All
measurements were bracketed by standard measurements in 0.3 mol/L HNO3 and were
done in MR. The filled circles were measured using the Apex-Q/Spiro introduction
system with 1 ppm Fe (~ 50 × 10− 11 A on 56Fe). The open circles were measured using
the SIS with 5 ppm Fe (also ~ 50 × 10− 11 A on 56Fe). As shown, during simple samplestandard bracketing, it is crucial to precisely match the acid concentrations of the
sample and the standard (within ~1%), which can be achieved easily.
54
Fe. Instead, 2 × δ57/56Fe is reported, which should be similar to δ56Fe
for mass-dependent fractionation. All doped solutions have normal
δ56Fe within 0.1‰ up to matrix concentrations of ~10 ppm but error
bars seem to increase with increasing matrix concentrations, reflecting greater instability of instrumental mass bias. Because Cu is not
easily separated during the chemistry, particular attention was paid to
this element. Iron has normal isotopic composition within 0.035‰
(2σ) even when the Cu concentration reaches 10 ppm. These results,
together with the study of Schoenberg and von Blanckenburg (2005)
indicate that conservatively, matrix elements should not affect the
accuracy of isotopic analyses, as long as their concentration is kept
below ~10 ppm (Element/Fe b 2).
4.4. Correction of isobaric interferences from
54
Cr and
68
Ni
Separation of Cr and Ni from Fe is straightforward with the chemistry
presented in Section 2. However, trace amounts of these elements may
remain after the chemistry, which can affect measurements of 54Fe and
58
Fe. In particular, isotopic analyses are very sensitive to the presence of
Ni because 58Fe is the least abundant isotope of Fe (0.2819 atom %, Taylor
et al., 1992) while 58Ni is the most abundant isotope of Ni (68.0769 atom
%, Gramlich et al., 1989). Even in HR mode, Cr and Ni interferences cannot
be resolved from Fe. However, one can correct these interferences by
analyzing the ion intensities of 53Cr and 60Ni. The data reduction proceeds
as follows. At first, β is calculated for the sample or standard based on the
57
Fe/56Fe ratio. This β is then used to compute fractionated 54Cr/53Cr and
58
Ni/60Ni ratios assuming that βCr =βNi =βFe and using unfractionated
ratios 54Cr/53Cr=0.24890 (Shields et al., 1966) and 58Ni/60Ni=2.59607
(Gramlich et al., 1989). The fractionated 54Cr/53Cr and 58Ni/60Ni ratios are
then combined with measured 53Cr and 60Ni intensities to estimate and
subtract isobaric interferences on 54Fe and 58Fe. In order to test this
correction scheme and evaluate its potential limitations, we have analyzed
the isotopic composition of Fe mixed with various levels of Cr and Ni
(Fig. 3B). The δ56/54Fe value is normal within 0.08‰ up to 54Cr/
54
Fe=0.04. This ratio corresponds to a correction of 40‰. When the
54
Cr/54Fe ratio reaches 0.7 (700‰), the correction begins to break down
(δ56/54Fe≈+0.6‰), most likely because the mass bias obtained from
57
Fe/56Fe is not appropriate for Cr (and Ni). The δ58/54Fe value remains
normal within uncertainties (±0.5‰) for Ni, as long as the 58Ni/58Fe ratio
is kept below ~0.05 (50‰). Erroneous negative δ58/54Fe are observed for
higher 58Ni/58Fe ratios. Better corrections can be obtained by artificially
modifying the 54Cr/53Cr and 58Ni/60Ni ratios to eliminate isotopic
Malinovsky et al. (2003) showed that the molarity of the acid used to
dilute the samples can affect instrumental mass fractionation for Fe.
They observed an increase of δ56Fe with increasing HNO3 concentration.
Correcting mass fractionation by doping with Ni, however, eliminates
the observed trend. The potential effect of acid molarity on the quality of
Fe isotopic analyses was studied as follows. Standards in 0.15 to 0.9 M
HNO3 were bracketed with standards in 0.3 M HNO3. Tests were
performed with both the SIS (5 ppm Fe) and the Apex-Q+ Spiro (1 ppm
Fe). In both cases, very large isotopic variations with changing acid
molarity were measured (Fig. 4). Note that the uncertainty also seems to
increase with increasing acid molarity. The use of a desolvating nebulizer
does not seem to considerably improve this problem. It is therefore
critical to match the molarity of the sample and standard whenever the
standard bracketing method is applied. The best results are achieved by
using the same batch of acid to dilute all solutions, including standards
(IRMM-014) and blanks. Effort must be also directed towards eliminating evaporative loss during the course of sample preparation and
analyses, which can change the molarity of the remaining solution (e.g.,
use an ultrasonic bath rather than a hot plate to dissolve the iron
residue).
4.6. Cu-doping
Details on how to implement Cu-doping method for instrumental
mass fractionation correction are given by Sharma et al. (2001), Kehm
Fig. 5. Comparison between δ56Fe analyses by sample-standard bracketing and Cudoping methods for basalts from Kilauea Iki lava lake (Teng et al., 2008). The error bars
take into account the long-term external reproducibility (see Section 5 for details).
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
Fig. 6. Long-term (a year) external reproducibility of BHVO-1 δ56Fe measurements
(n = 23). Each data point represents an independent measurement (from weighing the
powder to isotopic analysis). The small error bars represent 95% confidence intervals
based on the reproducibility of IRMM-014 measurements during the session when
BHVO-1 was analyzed (σ56
MassSpec, Eq. (3)). The large error bars include an additional
component of uncertainty to yield a MSWD of ~1 (σ56
Unknown= 0.011, Eq. (4)). The
56
weighted average δ Fe of BHVO-1 is 0.111 ± 0.006‰.
et al. (2003), Arnold et al. (2004), and Schoenberg and von
Blanckenburg (2005). The advantage of this method is that it reduces
the uncertainty arising from instability in instrumental mass bias and
it can also mitigate matrix effects that could be a source of inaccuracy.
However the duration of each analysis conducted on the Neptune is
increased by a factor of ~2 (Cr, Fe, Ni, and Cu cannot be analyzed
simultaneously and multi-dynamic mode must be used) and one must
ascertain that Cu from the sample has been completely eliminated
during the chemical separation of Fe or that it is present at a
sufficiently low level in the original sample not to affect Cu-doping.
Considering the pros and cons, sample-standard bracketing was
adopted for routine analyses. Cu-doping is still useful to evaluate the
accuracy of the measurements on selected samples. To test the Cudoping method, we analyzed basalt samples from Kilauea Iki lava lake
(Hawaii) measured previously by sample-standard bracketing (Teng
et al., 2008). The Fe/Cu ratio in these basalts is ~ 700 (~12 wt.% FeOtotal
and ~130 ppm Cu; Helz and Wright, 1992). Although Cu was not
separated from Fe, the contribution of Cu already in the sample to total
Cu after doping is negligible (~0.0014) and uncertainty on the isotopic
composition of indigenous Cu does not affect δ56Fe measurements
during mass discrimination correction. Unlike most sample-standard
bracketing measurements, which were done in MR mode, the Cudoping tests were performed in HR mode because isotopic ratios
should be less sensitive to the peak position. As shown in Fig. 5, there
is perfect agreement between sample-standard bracketing and Cudoping δ56Fe analyses. Although this does not unambiguously
demonstrate accuracy, it shows that the measurements do not suffer
from matrix effects.
5. Precision and accuracy
The uncertainties calculated using Eq. (3) only represent one
component of the total uncertainty, i.e., instrumental instability.
Indeed, other sources of uncertainty that were discussed in Sections
2 and 4 could influence the reproducibility of Fe isotopic analyses.
Given that one can arbitrarily reduce the uncertainty associated
with the instrument by increasing the number of analyses (e.g.,
precisions as low as 0.01‰ on δ56Fe have been reported in the
literature), special care must be given to other sources of error.
There are two aspects that need to be addressed regarding the
overall uncertainty of the measurements: (i) How can the
uncertainty arising from the whole procedure be quantified,
considering that only the instrumental uncertainty is known for
most samples? (ii) Are the measurements accurate within the
uncertainties that are quoted?
The proper way to address the first question is to run replicate
analyses of the same sample, from loading on the column or
181
dissolution (assuming that the powder is homogeneous) to
isotopic measurement. This will help better estimate the external
reproducibility. However, it is also desirable to incorporate the
instrumental uncertainty when reporting results. This can be easily
achieved by writing the standard deviation of the data as the
quadratic sum of the standard deviation associated with instability
of the mass spectrometer (σMassSpec, Eq. (3)), the standard
deviation corresponding to the distribution in δFe of the material
being analyzed (σNatural), and a standard deviation (σUnknown) that
accounts for all other sources of variability associated with the
analytical procedure (Humayun and Clayton, 1995; Rehkämper and
Halliday, 1999):
2
2
2
2
σ Data = σ MassSpec + σ Natural + σ Unknown :
ð4Þ
In order to estimate σUnknown, the Fe isotopic compositions of
geostandard powders were analyzed. The starting materials have
homogeneous δ56Fe with σNatural = 0. For a set of k replicate analyses,
one can compute the Mean Squared Weighted Deviates (MSWD or
reduced χ2),
MSWD =
2
xi −X
1 X
;
k − 1 i σ 2MassSpec; i + σ 2Unknown
ð5Þ
―
where X is the weighted average. Note that in this equation, σMassSpec
depends on the sample while σUnknown is fixed. If the analytical
uncertainty has been properly estimated, the dispersion of the data
around the average should be explained by σMassSpec and the MSWD
should be around unity. Otherwise, the measurements would call for a
positive σUnknown. The isotopic compositions of 23 replicates of the
BHVO-1 geostandard were measured over a period of a year using the
SIS (Fig. 6). The MSWD associated with these 23 replicates is 2.52. This
is outside the 95% confidence interval of 0.5–1.7 given by χ2 statistics.
The MSWD can be brought down to unity by adjusting σ56
Unknown to
0.011. Similarly, for δ57Fe, one can compute σ57
Unknown= 0.012. For
Table 2
Iron isotope results.
δ56Fe
σMassSpec
95% ci
δ57Fe
σMassSpec
95% ci
Geostandard
matrix+IRMM014
Allende a
Allende b
AC-E a
AC-E b
BCR-2 a
BCR-2 b
DTS-2 a
DTS-2 b
IF-G a
IF-G b
0.004
– 0.016
− 0.004
− 0.010
− 0.003
− 0.011
0.015
− 0.002
− 0.017
− 0.007
0.011
0.015
0.008
0.011
0.009
0.008
0.009
0.009
0.011
0.009
0.031
0.038
0.028
0.031
0.028
0.028
0.028
0.028
0.031
0.028
0.004
− 0.025
− 0.004
− 0.014
− 0.008
− 0.017
0.017
− 0.007
− 0.016
− 0.018
0.017
0.028
0.015
0.017
0.013
0.015
0.013
0.013
0.018
0.014
0.042
0.062
0.039
0.042
0.035
0.039
0.036
0.036
0.044
0.037
Geostandards
Allende
AC-E
BCR-2
DTS-2
IF-G
BHVO-1 (n = 23)
0.002
0.322
0.091
0.017
0.647
0.111
0.006
0.006
0.006
0.006
0.006
0.025
0.025
0.025
0.025
0.025
0.006
0.008
0.461
0.133
0.023
0.939
0.164
0.010
0.010
0.010
0.010
0.010
0.031
0.031
0.031
0.031
0.031
0.009
Geostandard matrix+IRMM014 correspond to sample matrices mixed with IRMM-014
iron. These samples have δFe = 0, demonstrating that the measurements are accurate.
The geostandards agree with recommended values (Dauphas and Rouxel, 2006).
σMassSpec corresponds to the standard deviation from the mass spectrometer (typically,
replicate analyses obtained on a single day). The 95% ci (confidence interval) integrates
our knowledge of the long-term reproducibility of BHVO-1 (Section 5) a and b represent
2 aliquots of IRMM-014 mixed with sample matrices processed independently through
the chemistry.
182
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
Fig. 7. Accuracy and precision of Fe isotopic analyses. A. Measurements of 50 μg IRMM014 mixed with sample matrices (a and b represent two aliquots processed
independently through the chemistry; see text for details). IRMM-014 purified by
column chromatography from these mixtures was analyzed by bracketing with pure
IRMM-014 not passed through column. Each measurement was repeated a large
number of times to reduce the uncertainty arising from mass spectrometer instability
and thus test the accuracy. Those uncertainties were compounded with the long-term
external reproducibility characterized based on BHVO-1 replicate analyses (Fig. 6). All
measurements are equal to 0 within uncertainties. The MSWD (reduced χ2) is 0.39,
within the 2-sided 95% confidence interval of 0.30 to 2.11 for n − 1 = 9 degrees of
freedom. The weighted mean δ56Fe is −0.0045 ± 0.0094‰ (95% confidence interval),
demonstrating that the measurements are accurate within ~ 0.01‰ for δ56Fe.
B. Comparison of geostandard measurements with recommended values (Dauphas
and Rouxel, 2006). All data points fall on the line with the slope of unity within
uncertainties. Note that a solid residue was left in the beaker after dissolution of DTS-2.
δ58Fe, the precision does not allow resolving other possible sources of
uncertainty than those from the mass spectrometer. So for this
isotope, σUnknown for δ56Fe was simply scaled by a factor of 2,
corresponding to the slope between δ58Fe and δ56Fe for mass
dependent fractionation (σ58
Unknown= 0.022). The values of σUnknown
for all isotopes based on BHVO-1 external reproducibility will be used
in all future studies and will be updated when more BHVO-1
measurements become available. All error bars quoted hereafter
include σUnknown. It is worth mentioning that σUnknown may differ
depending on the sample matrix, the chemistry used for Fe separation,
the inlet system (SIS or desolvating nebulizer) and the instrument
type. Therefore, similar tests (long-term external reproducibility of
high-precision measurements) should be performed in each laboratory. If the long-term external reproducibility can be explained by the
instrumental instability (MSWD ≈ 1 in Eq. (5)), σUnknown can be set to
0. The same approach can also be used for other isotopic systems.
Over the past several years, important developments have been
made in terms of analytical precision. Therefore, systematic biases
that could not be detected before may be revealed as the precision
improves. In order to test whether the measurements are accurate,
we have proceeded in a similar manner as that described in Bermin
et al. (2006) and Dauphas et al. (2007). Powdered geostandards
(Allende CV3 chondrite, AC-E granite, BCR-2 basalt, DTS-2 dunite,
and IF-G banded iron formation) with previously documented Fe
isotopic compositions (see the compilation in Dauphas and Rouxel,
2006) were passed through a first column of anion exchange. Both
Fe and the matrix were recovered. The matrix was passed through a
second column to completely eliminate any Fe that may have
persisted through the first column. The Fe-free matrix was doped
with 50 μg of IRMM-014 and then split into 2 aliquots (a and b in
Table 2 and Fig. 7). Each fraction was treated by 2 columns
following the procedure described in Section 2. The sample Fe
eluted from the first column was passed through a second column.
To summarize, three Fe solutions were generated from the original
geostandard solution. The first solution is Fe from the sample
treated using our usual procedure. The remaining two solutions
contained matrix elements doped with IRMM-014 and purified
using the procedure described in Section 2.
If the measurements are accurate and the analytical uncertainties
properly estimated, the isotopic composition of the solutions of
IRMM-014 + matrix should have δ56Fe = 0 within uncertainties. As
shown in Fig. 7 (Table 2), this is indeed the case. The weighted average
δ56Fe of all 10 measurements is −0.0045 ± 0.0094‰ and the MSWD is
0.39, which falls within the 2-sided 95% confidence interval of 0.30–
2.11. The geostandard matrix + IRMM-014 tests indicate that the
measurements are highly accurate.
The results obtained on geostandards were also compared with
those published in the literature (Fig. 7B). δ56Fe values of 0.647 ±
0.027, 0.091 ± 0.027, 0.017 ± 0.027, 0.002 ± 0.027, and 0.322 ±
0.027‰ were measured for IF-G, BCR-2, DTS-2, Allende, and AC-E,
respectively. This compares very well with published values of
0.631 ± 0.019, 0.064 ±0.034, 0.022 ± 0.041, − 0.011 ± 0.047,
0.289 ± 0.024‰ (see compilation in Dauphas and Rouxel, 2006).
Overall, the results from these tests demonstrate that that δ56Fe can be
measured accurately with a precision of less than ± 0.03‰ (95%
confidence interval).
6. Double-spike vs. sample-standard bracketing
Double-spike (58Fe–54Fe or 58Fe–57Fe have been used) is an
approach that was not investigated in this study but has the
potential to yield precise and accurate Fe isotopic analyses by
TIMS (Johnson and Beard, 1999; Fantle and Bullen, 2009) or MCICPMS (Balci et al., 2006; Dideriksen et al., 2006, 2007, 2008)
with a high sample throuhput. The main advantage of the double
spike is that the instrumental mass fractionation can be efficiently
corrected. Dideriksen et al. (2006, 2007, 2008) reported a
dispersion (2SD) of standard δ56Fe values of ± 0.06‰. This is
similar to what can be achieved by standard-sample bracketing
but the measurements of Dideriksen et al. were acquired with a
Thermoelemental Axiom instrument, which provides no intrinsic
capability to resolve argide interferences. Balci et al. (2006) reported
external reproducibilities (2SD) of ±0.07‰ on a Neptune HR-MC-ICPMS.
Whether better precisions can be achieved with such instruments
remains to be explored. One major drawback of the double-spike
technique is that the search for 58Fe anomalies in meteorites may be
hindered (Völkening and Papanastassiou, 1989; Dauphas et al., 2008).
Iron-58 was produced in massive stars together with 60Fe (t1/2 =1.49 My)
by neutron-capture reactions and as such, it represents a useful proxy to
probe the distribution of 60Fe in the protoplanetary disk (Dauphas et al.,
2008). An additional limitation is that the double-spike method
does not allow one to study the form of the mass-fractionation
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
law, which may provide clues on the mechanisms underlying
isotopic fractionation (Young et al., 2002).
7. Conclusion
A protocol was developed for routine analyses of the Fe isotopic
compositions of common natural materials. Iron is purified using a
two-step anion exchange column scheme. Iron isotopes are then
analyzed by HR-MC-ICPMS. A number of parameters that can affect
the accuracy and precision of δ56Fe measurements were identified and
examined:
1. The samples were introduced into the mass spectrometer using the
Standard Introduction System (a 50 μL/min Teflon nebulizer with a
cyclonic + Scott spray chamber). For samples with high Fe content,
utilizing an Apex-Q + Spiro inlet system did not offer any
advantage over a conventional wet-introduction system. Medium
resolution and standard Ni H-cones provided adequate intensities
for high-precision measurements with ~ 2 ppm Fe solutions.
2. The analyte concentrations of the sample and the reference
material (IRMM-014) were matched within 5%. Tests reveal that
concentration was not critical to acquiring accurate analyses. This
disagrees with the results of Malinovsky et al. (2003) but is in
agreement with Schoenberg and von Blanckenburg (2005).
3. Matching in acid molarity between samples and standards is
critical to achieving accurate measurements. Batches of blank and
reference material were prepared together with samples with the
exact same molarity (HNO3 0.3 M).
4. The measurements were not sensitive to the presence of other
transition elements in the solution, as long as their concentrations
remained below ~10 ppm (Element/Fe b 2).
5. Measurement of each solution was repeated 5 to 9 times. This
allowed the uncertainty associated with instability in instrumental
mass fractionation to be reduced to less than 0.02‰ for δ56Fe.
However, precision may not be limited by instrument instability per
se, but rather by the overall external reproducibility. Taking that into
account, a precision of ~0.03‰ (95% confidence interval) can be
routinely obtained.
6. The accuracy of Fe isotopic analyses was tested by measuring
IRMM-014 mixed with Fe-free matrix from geostandards. The
average δ56Fe is −0.0045 ± 0.0094‰, demonstrating that δ56Fe
measurements are accurate within the quoted uncertainties.
This study complements the earlier efforts of Weyer and
Schwieters (2003), Malinovsky et al. (2003), Arnold et al. (2004),
Poitrasson and Freydier (2005) and Schoenberg and von Blanckenburg (2005) by demonstrating that accurate isotopic analyses with
precisions of less than 0.03‰ on δ56Fe can be achieved routinely on
HR MC-ICPMS. This opens up exciting avenues of research in high
temperature Fe isotope geochemistry.
Acknowledgments
We thank Brian Lynch, Frank M. Richter, and Olivier J. Rouxel for
support and discussions. Careful reviews by two anonymous referees
were greatly appreciated. This work was supported by a fellowship
from the Packard Foundation, the France Chicago Center, and NASA
grant NNG06GG75G to N.D.
References
Albarède, F., Beard, B., 2004. Analytical methods for non-traditional isotopes. In:
Johnson, C.M., Beard, B.L., Albarède, F. (Eds.), Geochemistry of Non-Traditional
Isotopes, Reviews in Mineralogy and Geochemistry, vol. 55. Mineralogical Society of
America and Geochemical Society, Washington DC, pp. 113–152.
Alexander, C.M.O′D., Hewins, R.H., 2004. Mass fractionation of Fe and Ni isotopes in
metal in Hammadah Al Hamrah 237. Meteoritics Planet. Sci. 39, A13.
Anbar, A.D., 2004. Iron stable isotopes: beyond biosignatures. Earth Planet. Sci. Lett. 217,
223–236.
183
Anbar, A.D., Roe, J.E., Barling, J., Nealson, K.H., 2000. Nonbiological fractionation of iron
isotopes. Science 288, 126–128.
Archer, C., Vance, D., 2004. Mass discrimination correction in plasma source mass spectrometry: an example using Cu and Zn isotopes. J. Anal. At. Spectrom. 19, 656–665.
Arnold, G.L., Weyer, S., Anbar, A.D., 2004. Fe isotope variations in natural materials
measured using high mass resolution multiple collector ICPMS. Anal. Chem. 76,
322–327.
Balci, N., Bullen, T.D., Witte-Lien, K., Shanks, W.C., Motelica, M., Mandernack, K.W., 2006.
Iron isotope fractionation during microbially stimulated Fe(II) oxidation and Fe(III)
precipitation. Geochim. Cosmochim. Acta 70, 622–639.
Beard, B.L., Johnson, C.M., 2004. Fe isotope variations in the modern and ancient earth
and other planetary bodies. Rev. Mineral. Geochem. 55, 319–357.
Beard, B.L., Johnson, C.M., Skulan, J.L., Nealson, K.H., Cox, L., Sun, H., 2003. Application of
Fe isotopes to tracing the geochemical and biological cycling of Fe. Chem. Geol. 195,
87–117.
Belshaw, N.S., Zhu, X.K., Guo, Y., O′Nions, R.K., 2000. High precision measurement of
iron isotopes by plasma source mass spectrometry. Int. J. Mass Spectrom. 197,
191–195.
Bermin, J., Vance, D., Archer, C., Statham, P.J., 2006. The determination of the isotopic
composition of Cu and Zn in seawater. Chem. Geol. 226, 280–297.
Borrok, D.M., Wanty, R.B., Ridley, W.I., Wolf, R., Lamothe, P.J., Adams, M., 2007.
Separation of copper, iron, and zinc from complex aqueous solutions for isotopic
measurement. Chem. Geol. 242, 400–414.
Brantley, S.L., Liermann, L.J., Guynn, R.L., Anbar, A., Icopini, G.A., Barling, J., 2004. Fe
isotopic fractionation during mineral dissolution with and without bacteria.
Geochim. Cosmochim. Acta 68, 3189–3204.
Dauphas, N., Janney, P.E., Mendybaev, R.A., Wadhwa, M., Richter, F.M., Davis, A.M., van
Zuilen, M., Hines, R., Foley, C.N., 2004. Chromatographic separation and multicollection-ICPMS analysis of iron. Investigating mass-dependent and independent
isotope effects. Anal. Chem. 76, 5855–5863.
Dauphas, N., 2007. Diffusion-driven kinetic isotope effect of Fe and Ni during formation
of the Widmanstätten pattern, Meteoritics Planet. Sci. 42, 1597–1614.
Dauphas, N., Rouxel, O., 2006. Mass spectrometry and natural variations of iron
isotopes. Mass Spectrom. Rev. 25, 515–550. Erratum 25, 831–832.
Dauphas, N., van Zuilen, M., Busigny, V., Lepland, A., Wadhwa, M., Janney, P.E., 2007. Iron
isotope, major and trace element characterization of early Archean supracrsutal
rocks from SW Greenland: protolith identification and metamorphic overprint.
Geochim. Cosmochim. Acta 71, 4745–4770.
Dauphas, N., Cook, D.L., Sacarabany, A., Fröhlich, C., Davis, A.M., Wadhwa, M., Pourmand, A.,
Rauscher, T., Gallino, R., 2008. Iron-60 evidence for early injection and efficient mixing
of stellar debris in the protosolar nebula. Astrophys. J. 686, 560–569.
de Jong, J., Schoemann, V., Tison, J.-L., Becquevort, S., Masson, F., Lannuzel, D., Petit, J.,
Chou, L., Weis, D., Mattielli, N., 2007. Precise measurement of Fe isotopes in marine
samples by multi-collector inductively coupled plasma mass spectrometry (MCICP-MS). Anal. Chim. Acta 589, 105–119.
Dideriksen, K., Baker, J.A., Stipp, S.L.S., 2006. Iron isotopes in natural carbonate minerals
determined by MC-ICP-MS with a 58Fe-54Fe double spike. Geochim. Cosmochim.
Acta 70, 118–132.
Dideriksen, K., Christiansen, B.C., Baker, J.A., Frandsen, C., Balic-Zunic, T., Tullborg, E.,
Mørup, S., Stipp, S.L.S., 2007. Fe-oxide fracture fillings as a paleo-redox indicator:
Structure, crystal form and Fe isotope composition. Chem. Geol. 244, 330–343.
Dideriksen, K., Baker, J.A., Stipp, S.L.S., 2008. Equilibrium Fe isotope fractionation
between inorganic aqueous Fe(III) and the siderophore complex, Fe(III)-desferrioxamine B. Earth Planet. Sci. Lett. 269, 280–290.
Fantle, M.S., Bullen, T.D., 2009. Essentials of iron, chromium, and calcium isotope
analysis of natural materials by thermal ionization mass spectrometry. Chem. Geol.
258, 50–64.
Gramlich, J.W., Machlan, L.A., Barnes, I.L., Paulsen, P.J., 1989. The absolute abundance
ratios and atomic weight of a reference sample of nickel. J. Res. Natl. Inst. Stan. 94,
347–356.
Helz, R.T., Wright, T.L., 1992. Differentiation and magma mixing on Kilaueas east rift
zone—A further look at the eruptions of 1955 and 1960. Part I. The late 1955 lavas.
Bull. Volcanol. 54, 361–384.
Huang, F., Lundstrom, C.C., Glessner, J., Ianno, A., Boudreau, A., Li, J., Ferré, E.C., Marshak,
S., DeFrates, J., 2009. Chemical and isotopic fractionation of wet andesite in a
temperature gradient: experiments and models suggesting a new mechanism of
magma differentiation. Geochim. Cosmochim. Acta 73, 729–749.
Humayun, M., Clayton, R.N., 1995. Precise determination of the isotopic composition of
potassium: application to terrestrial rocks and lunar soils. Geochim. Cosmochim.
Acta 59, 2115–2130.
Johnson, C.M., Beard, B.L., 1999. Correction of instrumentally produced mass fractionation during isotopic analysis of Fe by thermal ionization mass spectrometry. Int. J.
Mass Spectrom. 193, 87–99.
Johnson, C.M., Beard, B.L., Roden, E.E., Newman, D.K., Nealson, K.H., 2004. Isotopic
constraints on biogeochemical cycling of Fe. Rev. Mineral. Geochem. 55, 359–408.
Kehm, K., Hauri, E.H., Alexander, C.M.O′D., Carlson, R.W., 2003. High precision iron
isotope measurements of meteoritic material by cold plasma ICP-MS. Geochim.
Cosmochim. Acta 67, 2879–2891.
Malinovsky, D., Stenberg, A., Rodushkin, I., Andren, H., Ingri, J., Öhlander, N., Baxter, D.C.,
2003. Performance of high resolution MC-ICP-MS for Fe isotope ratio measurements in sedimentary geological materials. J. Anal. At. Spectrom. 18, 687–695.
Maréchal, C.N., Télouk, P., Albarède, F., 1999. Precise analysis of copper and zinc isotopic
compositions by plasma-source mass spectrometry. Chem. Geol. 156, 251–273.
Mullen, J.G., 1961. Isotope effect in intermetallic diffusion. Phys. Rev. 121, 1649–1658.
Poitrasson, F., Freydier, R., 2005. Heavy iron isotope composition of granites determined
by high resolution MC-ICP-MS. Chem. Geol. 222, 132–147.
184
N. Dauphas et al. / Chemical Geology 267 (2009) 175–184
Polyakov, V.B., Mineev, S.D., 2000. The use of Mössbauer spectroscopy in stable isotope
geochemistry. Geochim. Cosmochim. Acta 64, 849–865.
Polyakov, V.B., Clayton, R.N., Horita, J., Mineev, S.D., 2007. Equilibrium iron isotope
fractionation factors of minerals: reevaluation from the data of nuclear inelastic
resonant X-ray scattering and Mössbauer spectroscopy. Geochim. Cosmochim. Acta
71, 3833–3846.
Rehkämper, M., Halliday, A.N., 1999. The precise measurement of Tl isotopic
compositions by MC-ICPMS: application to the analysis of geological materials
and meteorites. Geochim. Cosmochim. Acta 63, 935–944.
Richter, F.M., Dauphas, N., Teng, F.-Z., 2009. Non-traditional fractionation of nontraditional isotopes: Evaporation, chemical diffusion and Soret diffusion. Chem.
Geol. 258, 92–103.
Roskosz, M., Luais, B., Watson, H.C., Toplis, M.J., Alexander, C.M.O′D., Mysen, B.O., 2006.
Experimental quantification of the fractionation of Fe isotopes during metal
segregation from a silicate melt. Earth Planet. Sci. Lett. 248, 851–867.
Rouxel, O., Dobbek, N., Ludden, J., Fouquet, Y., 2003. Iron isotope fractionation during
oceanic crust alteration. Chem. Geol. 202, 155–182.
Schoenberg, R., von Blanckenburg, F., 2005. An assessment of the accuracy of stable Fe
isotope ratio measurements on samples with organic and inorganic matrices by
high-resolution multicollector ICP-MS. Int. J. Mass Spectrom. 242, 257–272.
Schoenberg, R., von Blanckenburg, F., 2006. Modes of planetary-scale Fe isotope
fractionation. Earth Planet. Sci. Lett. 252, 342–359.
Schuessler, J.A., Schoenberg, R., Behrens, H., von Blanckenburg, F., 2007. The
experimental calibration of the iron isotope fractionation factor between pyrrhotite
and peralkaline rhyolitic melt. Geochim. Cosmochim. Acta 71, 417–433.
Shahar, A., Young, E.D., Manning, C.E., 2008. Equilibrium high-temperature Fe isotope
fractionation between fayalite and magnetite: an experimental calibration. Earth
Planet. Sci. Lett. 268, 330–338.
Sharma, M., Polizzotto, M., Anbar, A.D., 2001. Iron isotopes in hot springs along the Juan
de Fuca Ridge. Earth Planet. Sci. Lett. 194, 39–51.
Shields, W.R., Murphy, T.J., Catanzaro, E.J., Garner, E.L., 1966. Absolute isotopic abundance
ratios and the atomic weight of a reference sample of chromium. J. Res. Natl. Bur. Stand.
70A, 193–197.
Strelow, F.W.E., 1980. Improved separation of iron from copper and other elements by
anion-exchange chromatography on a 4% cross-linked resin with high concentrations of hydrochloric acid. Talanta 27, 727–732.
Tachibana, S., Nagahara, H., Ozawa, K., Yamada, M., 2007. Isotopic fractionation of iron
during kinetic evaporation of metallic iron. Meteoritics Planet. Sci. 42, A146.
Taylor, P.D.P., Maeck, R., De Bièvre, P., 1992. Determination of the absolute isotopic
composition and atomic weight of a reference sample of natural iron. Int. J. Mass
Spectrom. Ion Processes. 121, 111–125.
Teng, F.-Z., Dauphas, N., Helz, R.T., 2008. Iron isotope fractionation during magmatic
differentiation in Kilauea Iki lava lake. Science 320, 1620–1622.
Thompson, A., Ruiz, J., Chadwick, O.A., Titus, M., Chorover, J., 2007. Rayleigh
fractionation of iron isotopes during pedogenesis along a climate sequence of
Hawaiian basalt. Chem. Geol. 238, 72–83.
Vance, D., Thirlwall, M., 2002. An assessment of mass discrimination in MC-ICPMS using
Nd isotopes. Chem. Geol. 185, 227–240.
Völkening, J., Papanastassiou, D.A., 1989. Iron isotope anomalies. Astrophys. J. 347,
L43–L46.
Wang, J., Davis, A.M., Clayton, R.N., Mayeda, T., 1994. Kinetic isotopic fractionation
during the evaporation of the iron oxide from liquid state. Lunar Planet. Sci. XXV,
1459–1460.
Weyer, S., Schwieters, J.B., 2003. High precision Fe isotope measurements with high
mass resolution MC-ICPMS. Int. J. Mass Spectrom. 226, 355–368.
Weyer, S., Ionov, D.A., 2007. Partial melting and melt percolation in the mantle: the
message from Fe isotopes. Earth Planet. Sci. Lett. 259, 119–133.
Wieser, M.E., Schwieters, J.B., 2005. The development of multiple collector mass
spectrometry for isotope ratio measurements. Int. J. Mass Spectrom. 242, 97–115.
Williams, H.M., Peslier, A.H., McCammon, C., Halliday, A.N., Levasseur, S., Teutsch, N.,
Burg, J.-P., 2005. Systematic iron isotope variations in mantle rocks and minerals:
the effects of partial melting and oxygen fugacity. Earth Planet. Sci. Lett. 235,
435–452.
Wombacher, F., Rehkämper, M., 2003. Investigation of the mass discrimination of
multiple collector ICP-MS using neodymium isotopes and the generalized power
law. J. Anal. At. Spectrom. 18, 1371–1375.
Woodhead, J., 2002. A simple method for obtaining highly accurate Pb isotope data by
MC-ICP-MS. J. Anal. At. Spectrom. 17, 1381–1385.
Young, E.D., Galy, A., Nagahara, H., 2002. Kinetic and equilibrium mass-dependent
isotope fractionation laws in nature and their geochemical and cosmochemical
significance. Geochim. Cosmochim. Acta 66, 1095–1104.
Zipfel, J., Weyer, S., 2007. In situ analyses of Fe isotopes in zoned metal grains of
Hammadad Al Hamra 237. Lunar Planet. Sci. XXXVIII #1927.
Zhu, X.K., Guo, Y., Williams, R.J.P., O′Nions, R.K., Matthews, A., Belshaw, N.S., Canters, G.W.,
de Waal, E.C., Weser, U., Burgess, B.K., Salvato, B., 2002. Mass fractionation processes of
transition metal isotopes. Earth Planet. Sci Lett. 200, 47–62.