HW Solution
advertisement

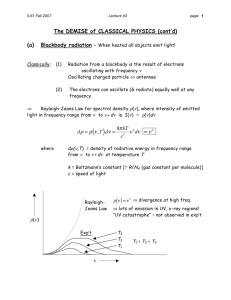
Solution set 1: Blackbody radiation and photoelectric effect Blackbody radiation and photoelectric effect Some background, especially for Q No. 2 The total energy radiated per unit time from a surface S is given by, ∫ P = IdA, s where I is the measure of intensity and dA is the differential element of the surface through which the energy is passing. For example, for a sphere centered at a point source radiating equally in all directions with power P , the above equation becomes, P = |I| Asurface P , |I| = 4πr2 where I is the intensity at the surface of sphere, r is the radius of the sphere and 4πr2 is the surface area. ∆Ε Α ∆x=v∆t The energy density is defined as the wave energy per unit volume. Suppose that a wave of velocity v is transferring energy ∆E into a volume ∆V in a time ∆t, i.e., ∆V = A∆x = Av∆t. The energy density therefore is, ρ = = = = I = ∆E ∆V ∆E Av∆t P Av I v ρv, 1 Solution set 1: Blackbody radiation and photoelectric effect which means that the intensity is energy density times wave velocity. Now, we have two definitions of intensity, P , A I = ρv, in units of W/m2 = J m−2 s−1 I = in units of (J/m3 ) (m/s) = J m−2 s−1 , (1) which depicts that both these definitions are consistent and dimensionally correct. 1. A student claims that she is going to eject electrons from a piece of metal by placing a radio transmitter antenna adjacent to the metal and sending a strong AM radio signal into the antenna. The work function of a metal is typically a few electron volts. Will this work? Answer The highest frequency in commercial AM radio is 1.6 MHz and the energy corresponding to this wave is, E = hf = 6.63 × 10−34 J s × 1.6 × 106 Hz = 6.7 neV. It is nine orders of magnitude too small to eject electrons from a metal surface. Therefore, it would be impossible to accomplish this task with this frequency. One has to broadcast at a minimum of ≈ 250 THz frequency, which is in the infrared region, to perform a similar task. 2. A black body at 7500 K consists of an opening of diameter 0.0500 mm, looking into an oven. Find the number of photons per second escaping the hole and having wavelengths between 500 nm and 501 nm (Use Planck’s law, ρT (λ) = 8πhc2 1 )· λ5 exp (hf /λkT )−1 Answer To find number of photons in the given wavelength range, we may take the average, i.e., λ= λ1 + λ2 , 2 as λ could be any wavelength greater than 500 nm and less than 501 nm. 2 Solution set 1: Blackbody radiation and photoelectric effect Now, if we multiply the intensity distribution with surface area and use the given wavelength range, we will get the energy leaving the hole. Therefore, from equation (1), the power total power radiated is, P = IA = ρcA. For a given wavelength range, between λ and dλ, the energy leaving the hole is, Pλ dλ = ρcAdλ. 8πhc dλ = c A dλ 5 λ exp( λkhc T ) − 1 B = (λ 1 +λ2 )5 2 = 8πhc2 ( 2hc exp( (λ1 +λ )−1 2 )kT )π ( d )2 (λ2 − λ1 ) 2 64π 2 hc2 d2 (λ2 − λ1 ) ), ( 2hc 5 (λ1 + λ2 ) exp( (λ1 +λ2 )kT ) − 1 but the energy of emitted photons should be equal to nhf . Therefore, Pλ dλ = nhf Pλ dλ n = , hf where hf is the energy of a single photon. 2 hc2 d2 (λ −λ ) 64π( 2 1 (λ1 +λ2 )5 2hc exp( (λ +λ 1 2 )kT = ) )−1 hf 2 hc2 d2 (λ −λ ) 64π( 2 1 (λ1 +λ2 )5 = exp( (λ 2hc 1 +λ2 )kT ) )−1 2hc (λ1 +λ2 ) 2 2 = 32π cd (λ2 − λ1 ) ( ) 2hc (λ1 + λ2 )4 exp( (λ1 +λ2 )kT ) − 1 = 32π 2 (3 × 108 m/s)(5 × 10−5 m)2 (1 × 10−9 m) ( ) 2(6.63×10−34 J/s)(3×108 ) m/s −9 4 (1001 × 10 m) exp( (1001×10−9 m)(1.38×10−23 J/K)(7.5×103 K) − 1) = 1.3 × 1015 , which is the number of photons emitted per second. 3. The radius of our sun is 6.96 × 108 m, and its total power output is 3.77 × 1026 W. (a) Assuming that the sun’s surface emits as a black body, calculate 3 Solution set 1: Blackbody radiation and photoelectric effect its surface temperature. (b) Using the results of part (a), find λmax for the sun. Answer Stefan’s law states that the total power of the emitted radiation increases with fourth power of temperature and is given by, P = σAεT 4 , where P is the power radiated from the surface of an object, σ = 5.67 × 10−8 W/(m2 K4 ) is the Stefan-Boltzmann constant, A is the surface area of an object and ε is the emmissitivity of the surface. Assuming the surface of the sun to be a blackbody, ε = 1, the surface temperature is, ( )1/4 P T = σA ( )1/4 3.77 × 1026 Watt = 4π(6.96 × 108 m)(5.67 × 10−8 W m−2 K4 ) = 5.75 × 103 K, which is in agreement with the actual average value of the solar surface temperature. (b) The maximum wavelength corresponding to this temperature is given by Wien’s displacement law. λmax × T = 2.898 × 10−3 m K 2.898 × 10−3 m K λmax = 5.75 × 103 K = 504 nm, which is blueish green light. 4. (a) Show that the expression for the number of standing waves can also be expressed in terms of wavelength and written as, N(λ) dλ = 8πV dλ , λ4 (2) where V = L3 is the volume of the cubic cavity. (b) How many independent standing waves with wavelengths between 9.5 and 10.5 mm are present in a cubical cavity 1 m on a side? 4 Solution set 1: Blackbody radiation and photoelectric effect Answer (a) The number of standing waves in the cavity (in terms of frequency) is given by, N (f ) df = 8πV 3 f df, c3 where V = a3 . Since, c = cλ−1 λ −c df = 2 dλ. λ f = The negative sign shows that λ decreases when f increases. Since we are concerned with just the scalars, we consider only the magnitude, 8πV ( c2 )( c ) N (λ)dλ = dλ c 3 λ2 λ2 8πV = dλ. λ4 This is the required expression for the number of standing waves in terms of wavelength. (b) The number of standing waves are, 8πV dλ λ4 8π(1 m)3 (10−3 m) = (10 × 10−3 m)4 8π10−3 m4 = 10−8 m4 = 8π108−3 = 8π105 N (λ)dλ = = 2.5 × 106 . 5. A simple pendulum has a length of 1.00 m and a mass of 1.00 kg. The amplitude of oscillations of the pendulum is 3.0 cm. Estimate the quantum number for the pendulum. Answer The energy of the pendulum is, E = mgh = mg(L − L cos θ) = (1 Kg)(9.8 m/s2 )(1 − cos(0.0300)) = 4.4 × 10−3 J. 5 Solution set 1: Blackbody radiation and photoelectric effect θ Lcosθ L x m mg The frequency is, √ ω 1 g f = = 2π 2π l √ 1 9.8 m/s2 = 2π 1.0 m = 0.5 Hz. Therefore, the quantum number for this pendulum is, n = E hf 4.4 × 10−3 J (6.63 × 10−34 J s)(0.5 Hz) = 1.34 × 1031 . = One readily sees verify that due to the large value of quantum number n, it is impossible to detect any quantum mechanical effects in this macroscopic pendulum (We will learn that this is called Bohr’s correspondence principle.) 6. Find the energy density of black body radiation at T = 6000 K in the range from 450 to 460 nm, assuming that this range is so narrow that the energy density function ρT (f ) does not vary much over it. (Use Planck’s law, ρT (f ) = 8πhf 3 1 )· c3 exp (hf /kT )−1 6 Solution set 1: Blackbody radiation and photoelectric effect Answer 3 × 108 m/s = 6.52 × 1014 Hz λmax 460 × 10−9 m c 3 × 108 m/s fmax = = = 6.66 × 1014 Hz λmin 450 × 10−9 m df = 1.45 × 1013 Hz fmin = c = favg = 6.60 × 1014 Hz. To use Plank’s formula, we first evaluate hfavg , kT assuming the frequency in the interval is the average value, hfavg (6.63 × 10−34 J s)(6.60 × 1014 Hz) = = 5.28 kT (1.38 × 10−23 J/K)6000 K hfavg exp( ) ≈ exp(5.28) = 197. kT Therefore, 8πhf f 2 df ( ) c3 exp hfavg − 1 kT (8π)(6.63 × 10−34 J s)(6.60 × 1014 Hz)3 (1.45 × 1013 Hz) = (3 × 108 )2 (197 − 1) = 8.2 × 10−7 J/m3 , ρT (f )df = which is the required result. 7. Why does the photoelectric current not rise vertically to its maximum value when the applied potential difference is slightly more positive than −V0 ? Answer The electric field is not strong enough to ensure that all the ejected electrons reach the anode, therefore the current does not immediately rise to its saturation value. Second, there is a cloud of freshly ejected electrons surrounding the cathode, called the space charge, which prevents the transport of electrons due to repulsive forces. As the voltage is increased, the space charge density decreases allowing more and more electrons to reach the cathode. 8. A student studying the photoelectric effect from two different metals records the following information: (a) the stopping potential for photoelectrons released from metal 1 is 1.48 V larger than that for metal 2, and 7 Solution set 1: Blackbody radiation and photoelectric effect (b) the threshold frequency for metal 1 is 40.0 % smaller than that for metal 2. Determine the work functions for each metal. Answer hf = e(∆Vs1 ) + ϕ1 hf = e(∆Vs2 ) + ϕ2 It is given that, ∆Vs1 = ∆Vs2 + 1.48 V ⇒ ϕ2 − ϕ1 = 1.48 V. (3) Similarly, hfc1 = ϕ1 = 0.600 hfc2 = 0.6 ϕ2 , substituting this in (1) yields, ϕ2 − 0.60ϕ2 = 1.48 ⇒ ϕ2 = 3.7 eV ϕ1 = 2.2 eV. 9. The work function for tungsten is 4.50 eV. Calculate the speed of the fastest electrons ejected from a tungsten surface when light whose photon energy is 5.80 eV shines on the surface. Answer 1 2 = Ephoton − ϕ Kmax = mvmax 2 ( )2 1 vmax 2 (me c ) = Ephoton − ϕ 2 c √ 2(Ephoton − ϕ) vmax = c me c2 √ 2(5.8 eV − 4.50 eV) = (3 × 108 m/s) 0.511 × 106 eV = 6.8 × 105 m/s. 8 Solution set 1: Blackbody radiation and photoelectric effect 10. At what rate does the sun emit photons? For simplicity, assume that the Sun’s entire emission at the rate of 3.9 × 1026 W is at the single wavelength of 550 nm. Answer Since we know that, P = IE, where P is the power output of sun and E is the energy of a single photon. Therefore, P = IE = I = Ihc λ Pλ hc (550 nm)(3.9 × 1026 W) (6.63 × 10−34 J s)(3 × 108 m/s) = 1 × 1045 photons/sec. = 11. Suppose the fractional efficiency of a cesium surface (with work function 1.80 eV) is 1.0 × 1016 ; that is, on average one electron is ejected for every 1016 photons that reach the surface. What would be the current of electrons ejected from such a surface if it were illuminated with 600 nm light from a 2.00 mW laser and all the ejected electrons took part in the charge flow? Answer The number of photons emitted from the laser surface per unit time is, I = P Ephoton =( 2 × 10−3 W ) 1240 eV.nm (1.6 × 10−19 J/eV) 600 nm = 6.05 × 105 /s, of which (1.0 × 10−16 )(6.05 × 1015 /s) = 0.605/s actually cause photoelectric emission. Therefore, the current is, i = (0.605/s)(1.6 × 10−19 C) = 9.7 × 10−20 A. 9