Electrochemical Reactions
advertisement

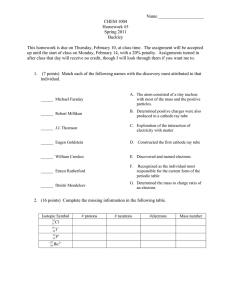
Electrochemical Reactions In electrochemical reactions, electrons are transferred from one species to another. Substance which receives electrons is the oxidising agent or oxidant and is itself reduced. Substance which gives up its electrons is the reducing agent or reductant and is itself oxidised. Oxidation and Reduction Gain of Electrons is Reduction GER LEO Loss of Electrons is Oxidation Balancing Redox Equations (Chem 120) Reduction Potentials Reduction Potentials Voltaic Cells In spontaneous oxidation-reduction (redox) reactions, electrons are transferred and energy is released. Figure 16.2 Voltaic Cells We can use that energy to do work if we make the electrons flow through an external device. We call such a setup a voltaic cell. Figure 16.3 Voltaic Cells A typical cell looks like this. The oxidation occurs at the anode. The reduction occurs at the cathode. Voltaic Cells Figure 16.4 Once even one electron flows from the anode to the cathode, the charges in each beaker would not be balanced and the flow of electrons would stop. Voltaic Cells Figure 16.4 Therefore, we use a salt bridge, usually a U-shaped tube that contains a salt solution, to keep the charges balanced: Cations move toward the cathode. Anions move toward the anode. Voltaic Cells In the cell, electrons leave the anode and flow through the wire to the cathode. As the electrons leave the anode, the cations formed dissolve into the solution in the anode compartment. Voltaic Cells As the electrons reach the cathode, cations in the cathode are attracted to the now negative cathode. The electrons are taken by the cation, and the neutral metal is deposited on the cathode. Electromotive Force (emf) Figure 16.6 Water only spontaneously flows one way in a waterfall. Likewise, electrons only spontaneously flow one way in a redox reaction— from higher to lower potential energy. Electromotive Force (emf) The potential difference between the anode and cathode in a cell is called the electromotive force (emf). It is measured in volts (V): J 1V=1 C It is also called the cell potential, and is designated Ecell. Standard Cell Potentials The cell potential at standard conditions can be found through this equation: = Ered (cathode) − Ered (anode) Ecell Because cell potential is based on the potential energy per unit of charge, it is an intensive property. Standard Hydrogen Electrode Their values are referenced to a standard hydrogen electrode (SHE). By definition, the reduction potential for hydrogen is 0 V. 2 H+ (aq, 1M) + 2 e− H2 (g, 1 atm) Figure 16.7 Standard Reduction (Half-Cell) Potentials Table 16.1 Reduction potentials for many electrodes have been measured and tabulated. Cell Potentials For the oxidation in this cell: = −0.76 V Ered For the reduction: Ered = +0.34 V Figure 16.8 Cell Potentials = Ered (cathode) − Ered (anode) Ecell = +0.34 V − (−0.76 V) = +1.10 V Strengths of Oxidising and Reducing Agents Figure 16.11 The strongest oxidisers have the most positive reduction potentials. The strongest reducers have the most negative reduction potentials. Strengths of Oxidising and Reducing Agents The greater the difference between the two, the greater the voltage of the cell. Figure 16.9 Cell emf and G G for a redox reaction can be found by using the equation: G = −nFE A positive value of E and a negative value of G both indicate that a reaction is spontaneous. Consequently, under standard conditions: G = −nFE n is the number of moles of electrons transferred. F is called Faraday’s constant: 1 F = 96,485 C/mol = 96,485 J/V mol Cell emf Under Non-standard Conditions - The Nernst Equation Remember that: G = G + RT ln Q This means: −nFE = −nFE + RT ln Q Cell emf Under Non-standard Conditions - The Nernst Equation Dividing both sides by −nF, we get the Nernst equation: RT E = E − nF ln Q or, using base-10 logarithms, E = E − 2.303 RT log Q nF Cell emf Under Non-standard Conditions - The Nernst Equation At room temperature (298 K): 2.303 RT = 0.0592 V F Thus the equation simplifies to: E = E − 0.0592 log Q n Concentration Cells Notice that the Nernst equation implies that a cell could be created that has the same substance at both electrodes. would be 0, but Q would not. For such a cell, Ecell Therefore, as long as the concentrations are different, E will not be 0. Figure 16.12 Batteries and Fuel Cells The Hydrogen Fuel Cell Figure 16.16 Corrosion of Iron Figure 16.17 Preventing the Corrosion of Iron Figure 16.18 Electrolysis Figure 16.20 Voltaic cells are based on a spontaneous redox reaction, but it is possible to use electricity to cause non-spontaneous redox reactions to occur. For example, the electrolysis of molten sodium chloride to produce sodium metal and chlorine gas. Electrolysis Electroplating is another instance where elecrolysis is used to deposit a thin layer of one metal on another to improve beauty or resistance to corrosion, for example the nickel-plating of steel. One point worth noting is that in both voltaic and electrolytic cells, the anode is where oxidation occurs (and the cathode is where reduction occurs). Figure 16.21 Quantitative Aspects of Electrolysis Half-reactions show how many electrons are needed to achieve an electrolytic process and so it follows that the amount of a substance oxidised (or reduced) is directly proportional to the number of electrons passed into the cell. Figure 16.22