practical considerations
advertisement

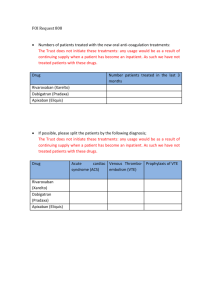
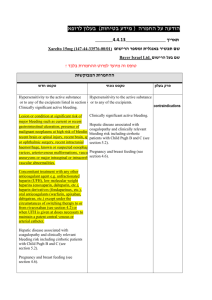
P R A C T I C A L C O N S I D E R AT I O N S W H E N U S I N G X A R E LT O ® Contents Patient Types •Patients With Nonvalvular Atrial Fibrillation . . . . . . . . . . . . . . . . . . .. . . . . . . . . . 2 •Patients With Deep Vein Thrombosis or Pulmonary Embolism .. . . . . . . 4 •Knee or Hip Replacement Surgery Patients.. . . . . . . . . . . . . . . . . . . .. . . . . . . . . . 6 Other Considerations With XARELTO® •Bleeding Risk. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . •Bleed Management. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .. . . . . . . . . . •Considerations. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .. . . . . . . . . . •Management of Overdose. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .. . . . . . . . . . •Increased Risk of Stroke After Discontinuation in Nonvalvular AF .. . . •Transitioning to and From XARELTO®. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . •Important Food Effect Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .. . . . . . . . . . •Dose Adjustments .. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . •Missed Dose.. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .. . . . . . . . . . •Administration Options.. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .. . . . . . . . . . •Surgery and Other Medical Interventions. . . . . . . . . . . . . . . . . . . . . . .. . . . . . . . . . •Assessment of Coagulation Status. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7 7 8 8 8 9 9 10 10 10 11 11 Additional Resources . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .. . . . . . . . . . 12 Prescribing Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14 IMPORTANT SAFETY INFORMATION WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO® INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA A. PREMATURE DISCONTINUATION OF XARELTO® INCREASES THE RISK OF THROMBOTIC EVENTS Premature discontinuation of any oral anticoagulant, including XARELTO®, increases the risk of thrombotic events. If anticoagulation with XARELTO® is discontinued for a reason other than Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. (continued on page 2) APPROVED FOR MULTIPLE INDICATIONS Patient Types IMPORTANT SAFETY INFORMATION (cont’d) Patients With Nonvalvular Atrial Fibrillation Indication XARELTO® (rivaroxaban) is indicated to reduce the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation (AF). There are limited data on the relative effectiveness of XARELTO® and warfarin in reducing the risk of stroke and systemic embolism when warfarin therapy is well controlled. Patients Included in Clinical Trial ROCKET AF was the pivotal phase 3 trial that determined noninferiority of XARELTO® to dose-adjusted warfarin for reducing the risk of stroke and non–central nervous system (CNS) systemic embolism in patients with nonvalvular AF. Patients with documented nonvalvular AF were at a moderate to high risk of stroke.1 In ROCKET AF, XARELTO® was studied in patients with multiple comorbidities reflecting an elevated risk for stroke. Patients included in the trial had a history of stroke, transient ischemic attack (TIA), or non–CNS systemic embolism, or ≥2 of the following additional risk factors1: Age ≥75 years Hypertension Heart failure or left ventricular ejection fraction ≤35% Diabetes mellitus The mean CHADS2 score of patients in the trial was 3.5.1 Patient CHADS2 Scores in ROCKET AF 2 44 28.7% 28.7% 5 12.7% 3 2 6 2.0% 13.0% 43.6% A. PREMATURE DISCONTINUATION OF XARELTO® INCREASES THE RISK OF THROMBOTIC EVENTS (cont’d) pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant. B. S PINAL/EPIDURAL HEMATOMA Epidural or spinal hematomas have occurred in patients treated with XARELTO® who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. Consider these risks when scheduling patients for spinal procedures. Factors that can increase the risk of developing epidural or spinal hematomas in these patients include: u se of indwelling epidural catheters U C u oncomitant use of other drugs that affect hemostasis, such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet inhibitors, other anticoagulants, see Drug Interactions u A history of traumatic or repeated epidural or spinal punctures u A history of spinal deformity or spinal surgery u Optimal timing between the administration of XARELTO® and neuraxial procedures is not known Monitor patients frequently for signs and symptoms of neurological impairment. If neurological compromise is noted, urgent treatment is necessary. Consider the benefits and risks before neuraxial intervention in patients anticoagulated or to be anticoagulated for thromboprophylaxis. (continued on page 3) Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 2 Patients With Nonvalvular Atrial Fibrillation (cont’d) IMPORTANT SAFETY INFORMATION (cont’d) Patients With Renal Impairment CONTRAINDICATIONS In the ROCKET AF trial, patients with creatinine clearance (CrCl) >50 mL/min received XARELTO® 20 mg once daily with the evening meal. Patients with CrCl 30 to 50 mL/min were administered XARELTO® 15 mg once daily with the evening meal, resulting in serum concentrations of rivaroxaban and clinical outcomes similar to those in patients with better renal function administered XARELTO® 20 mg once daily atients with CrCl 15 to 30 mL/min were not studied, but administration of XARELTO® P 15 mg once daily is also expected to result in serum concentrations of rivaroxaban similar to those in patients with normal renal function void use of XARELTO® in nonvalvular AF patients with CrCl <15 mL/min since drug A exposure is increased eriodically assess renal function as clinically indicated (ie, more frequently in situations in P which renal function may decline) and adjust therapy accordingly. Discontinue XARELTO® in patients who develop acute renal failure while on XARELTO® Additional Important Considerations remature discontinuation of any oral anticoagulant, including XARELTO®, in the absence P of adequate alternative anticoagulation increases the risk of thrombotic events. If XARELTO® is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant. Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. u Active pathological bleeding u S evere hypersensitivity reaction to XARELTO® (eg, anaphylactic reactions) WARNINGS AND PRECAUTIONS u I ncreased Risk of Thrombotic Events After Premature Discontinuation: Premature discontinuation of any oral anticoagulant, including XARELTO®, in the absence of adequate alternative anticoagulation increases the risk of thrombotic events. An increased rate of stroke was observed during the transition from XARELTO® to warfarin in clinical trials in atrial fibrillation patients. If XARELTO® is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant. (continued on page 4) 3 Patients With Deep Vein Thrombosis or Pulmonary Embolism IMPORTANT SAFETY INFORMATION (cont’d) Indications WARNINGS AND PRECAUTIONS (cont’d) XARELTO (rivaroxaban) is indicated for the treatment of deep vein thrombosis (DVT). u Risk of Bleeding: XARELTO® increases the risk of bleeding and can cause serious or fatal bleeding. Promptly evaluate any signs or symptoms of blood loss and consider the need for blood replacement. Discontinue XARELTO® in patients with active pathological hemorrhage. • A specific antidote for rivaroxaban is not available. Because of high plasma protein binding, rivaroxaban is not expected to be dialyzable. • Concomitant use of other drugs affecting hemostasis increases the risk of bleeding. These include aspirin, P2Y12 platelet inhibitors, other antithrombotic agents, fibrinolytic therapy, and NSAIDs. ® XARELTO® is indicated for the treatment of pulmonary embolism (PE). XARELTO® is indicated for the reduction in the risk of recurrence of deep vein thrombosis and of pulmonary embolism following initial 6 months treatment for DVT and/or PE. Patients Included in Clinical Trials EINSTEIN DVT and EINSTEIN PE Trials EINSTEIN DVT and EINSTEIN PE were the multinational, open-label, noninferiority clinical trials comparing XARELTO® (at an initial dose of 15 mg twice daily with food for the first 3 weeks, followed by XARELTO® 20 mg once daily with food) to enoxaparin (1 mg/kg twice daily for at least 5 days with warfarin and then continued with warfarin only after the target international normalized ratio [2.0–3.0] was reached). Patients were eligible for the EINSTEIN DVT trial if they had acute, symptomatic, objectively confirmed proximal DVT, without symptomatic pulmonary embolism.3 Patients were eligible for the EINSTEIN PE trial if they had acute symptomatic PE with objective confirmation, with or without symptomatic DVT.4 (continued on page 5) In the EINSTEIN DVT and EINSTEIN PE trials, 49% of patients had an idiopathic DVT/PE at baseline. Other risk factors are shown below. Patient Risk Factors in EINSTEIN DVT and EINSTEIN PE Trials 50 40 49% 30 20 19% 10 0 Idiopathic DVT/PE Previous DVT/PE 18% Recent Surgery or Trauma 16% 8% 6% Immobilization Use of EstrogenKnown Containing Thrombophilic Drug Conditions 5% Active Cancer More than 8200 patients in both trials were randomized and followed on study treatment for a mean of 208 days in the XARELTO® group and 204 days in the enoxaparin + warfarin group. Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 4 Patients With Deep Vein Thrombosis or Pulmonary Embolism (cont’d) IMPORTANT SAFETY INFORMATION (cont’d) Patients Included in Clinical Trials (cont’d) WARNINGS AND PRECAUTIONS (cont’d) EINSTEIN Extension Trial The EINSTEIN Extension trial was a multinational, double-blind, superiority clinical study comparing XARELTO® (20 mg once daily with food) with placebo in patients who had completed 6 to 14 months of treatment for DVT and/or PE following the acute event. More than 1100 patients were randomized and followed on study treatment for a mean of 190 days in both the XARELTO® and placebo treatment groups. The mean age was approximately 58 years. The population was 58% male, 78% Caucasian, 8% Asian, and 2% Black. In the EINSTEIN Extension trial, approximately 60% of patients had a history of proximal index DVT without a PE event, and 29% of patients had a PE without a symptomatic DVT event. Patients With Renal Impairment In the EINSTEIN trials, patients with CrCl values <30 mL/min at screening were excluded from the studies void the use of XARELTO® in patients with CrCl <30 mL/min due to an expected increase A in rivaroxaban exposure and pharmacodynamic effects in this patient population u Spinal/Epidural Anesthesia or Puncture: When neuraxial anesthesia (spinal/epidural anesthesia) or spinal puncture is employed, patients treated with anticoagulant agents for prevention of thromboembolic complications are at risk of developing an epidural or spinal hematoma, which can result in long-term or permanent paralysis. To reduce the potential risk of bleeding associated with the concurrent use of rivaroxaban and epidural or spinal anesthesia/analgesia or spinal puncture, consider the pharmacokinetic profile of rivaroxaban. Placement or removal of an epidural catheter or lumbar puncture is best performed when the anticoagulant effect of rivaroxaban is low; however, the exact timing to reach a sufficiently low anticoagulant effect in each patient is not known. An epidural catheter should not be removed earlier than 18 hours after the last administration of XARELTO®. The next XARELTO® dose is not to be administered earlier than 6 hours after the removal of the catheter. If traumatic puncture occurs, the administration of XARELTO® is to be delayed for 24 hours. Should the physician decide to administer anticoagulation in the context of epidural or spinal anesthesia/analgesia or lumbar puncture, monitor frequently to detect any signs or symptoms of neurological impairment, such as midline back pain, sensory and motor deficits (numbness, tingling, or weakness in lower limbs), or bowel and/or bladder dysfunction. Instruct patients to immediately report if they experience any of the above signs or symptoms. (continued on page 6) Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 5 Knee or Hip Replacement Surgery Patients IMPORTANT SAFETY INFORMATION (cont’d) Indications WARNINGS AND PRECAUTIONS (cont’d) XARELTO (rivaroxaban) is indicated for the prophylaxis of DVT, which may lead to PE in patients undergoing knee replacement surgery. Spinal/Epidural Anesthesia or Puncture: (cont’d) If signs or symptoms of spinal hematoma are suspected, initiate urgent diagnosis and treatment including consideration for spinal cord decompression even though such treatment may not prevent or reverse neurological sequelae. ® Patients Included/Excluded in Clinical Trials XARELTO® was studied in the RECORD clinical trials with more than 9000 orthopedic patients undergoing knee or hip replacement surgery. HIP REPLACEMENT TRIALS RECORD1 AND RECORD2 KNEE REPLACEMENT TRIAL RECORD3 Elective total hip/ knee replacement ean age = 63, with 49% of M patients aged ≥65 years Female = 55% ean age = 68, with 66% M of patients aged ≥65 years Female = 68% Patients with these conditions were excluded from the trials ndergoing staged bilateral U total hip replacement Severe renal impairment (creatinine clearance [CrCl] <30 mL/min) Significant liver disease (hepatitis or cirrhosis) S evere renal impairment (CrCl <30 mL/min) Significant liver disease (hepatitis or cirrhosis) u Use in Patients With Renal Impairment: • Nonvalvular Atrial Fibrillation: Avoid the use of XARELTO® in patients with creatinine clearance (CrCl) <15 mL/min, since drug exposure is increased. Discontinue XARELTO® in patients who develop acute renal failure while on XARELTO®. • Treatment of Deep Vein Thrombosis (DVT), Pulmonary Embolism (PE), and Reduction in the Risk of Recurrence of DVT and of PE: Avoid the use of XARELTO® in patients with CrCl <30 mL/min due to an expected increase in rivaroxaban exposure and pharmacodynamic effects in this patient population. (continued on page 7) Patients With Renal Impairment void the use of XARELTO® for the prophylaxis of DVT in patients with severe renal A impairment (CrCl <30 mL/min) due to an expected increase in rivaroxaban exposure and pharmacodynamic effects in this patient population. Use with caution in patients with moderate impairment (CrCl 30 to 50 mL/min) ombined analysis of the RECORD1-3 trials did not show an increase in bleeding risk for C patients with moderate renal impairment, and reported a possible increase in total venous thromboembolism in this population. Observe closely and promptly evaluate any signs or symptoms of blood loss in patients with moderate renal impairment (CrCl 30 to 50 mL/min) Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 6 Other Considerations With XARELTO® IMPORTANT SAFETY INFORMATION (cont’d) Bleeding Risk WARNINGS AND PRECAUTIONS (cont’d) XARELTO® increases the risk of bleeding and can cause serious or fatal bleeding. In clinical trials, the most common adverse reactions with XARELTO® were bleeding complications. XARELTO® is contraindicated in patients with active pathological bleeding. In deciding whether to prescribe XARELTO® to patients at increased risk of bleeding, the risk of thrombotic events should be weighed against the risk of bleeding. Promptly evaluate any signs or symptoms of blood loss. Discontinue XARELTO® in patients with active pathological hemorrhage Concomitant use of drugs affecting hemostasis increases the risk of bleeding. These include: Drugs affecting hemostasis Aspirin, P2Y12 platelet inhibitors, other antithrombotic agents, fibrinolytic therapy, non-steroidal anti-inflammatory drugs (NSAIDs) Concomitant use of drugs that are combined P-gp and CYP3A4 inhibitors increases rivaroxaban exposure and may increase bleeding risk. These include: Drugs that are combined P-gp and CYP3A4 inhibitors For example, ketoconazole and ritonavir Bleed Management* • Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery: Avoid the use of XARELTO® in patients with CrCl <30 mL/min due to an expected increase in rivaroxaban exposure and pharmacodynamic effects in this patient population. Observe closely and promptly evaluate any signs or symptoms of blood loss in patients with CrCl 30 to 50 mL/min. Patients who develop acute renal failure while on XARELTO® should discontinue the treatment. u Use in Patients With Hepatic Impairment: No clinical data are available for patients with severe hepatic impairment. Avoid use of XARELTO® in patients with moderate (Child-Pugh B) and severe (Child-Pugh C hepatic impairment or with any hepatic disease associated with coagulopathy, since drug exposure and bleeding risk may be increased. (continued on page 8) Promptly evaluate any signs and symptoms of blood loss and consider the need for blood replacement Discontinue XARELTO® in patients with active pathological hemorrhage P artial reversal of prothrombin time prolongation has been seen after administration of prothrombin complex concentrates (PCCs) in healthy volunteers T he use of other procoagulant reversal agents like activated prothrombin complex concentrates (APCCs) or recombinant factor VIIa (rFVIIa) has not been evaluated *This is not intended to replace clinical judgment or determine individual patient care. Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 7 Considerations IMPORTANT SAFETY INFORMATION (cont’d) A specific antidote for XARELTO is not available ® The terminal elimination half-life of XARELTO® is 5 to 9 hours in healthy subjects aged 20 to 45 years and 11 to 13 hours in the elderly XARELTO® is not expected to be dialyzable due to high plasma protein binding Protamine sulfate and vitamin K are not expected to affect the anticoagulant activity of XARELTO® Partial reversal of prothrombin time prolongation has been seen after administration of PCCs in healthy volunteers The use of other procoagulant reversal agents like APCC or recombinant factor VIIa (rFVIIa) has not been evaluated Management of Overdose Overdose of XARELTO® may lead to hemorrhage. A specific antidote for rivaroxaban is not available (see information above). Discontinue XARELTO® and initiate appropriate therapy if bleeding complications associated with overdosage occur. The use of activated charcoal to reduce absorption in case of XARELTO® overdose may be considered Due to high plasma protein binding, rivaroxaban is not expected to be dialyzable Increased Risk of Stroke After Discontinuation in Nonvalvular AF Discontinuing XARELTO® in the absence of adequate alternative anticoagulation increases the risk of thrombotic events. An increased rate of stroke was observed during the transition from XARELTO® to warfarin in clinical trials in atrial fibrillation patients. WARNINGS AND PRECAUTIONS (cont’d) u Use With P-gp and Strong CYP3A4 Inhibitors or Inducers: Avoid concomitant use of XARELTO® with combined P-gp and strong CYP3A4 inhibitors (eg, ketoconazole, itraconazole, lopinavir/ritonavir, ritonavir, indinavir/ritonavir, and conivaptan). Avoid concomitant use of XARELTO® with drugs that are P-gp and strong CYP3A4 inducers (eg, carbamazepine, phenytoin, rifampin, St. John’s wort). u R isk of Pregnancy-Related Hemorrhage: In pregnant women, XARELTO® should be used only if the potential benefit justifies the potential risk to the mother and fetus XARELTO® dosing in pregnancy has not been studied. The anticoagulant effect of XARELTO® cannot be monitored with standard laboratory testing and is not readily reversed. Promptly evaluate any signs or symptoms suggesting blood loss (eg, a drop in hemoglobin and/or hematocrit, hypotension, or fetal distress). (continued on page 9) If XARELTO® must be discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider administering another anticoagulant. The Boxed WARNINGS for XARELTO® are based on the risk associated with permanently discontinuing XARELTO® therapy without proper transition or management. Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 8 Transitioning to and From XARELTO® IMPORTANT SAFETY INFORMATION (cont’d) WARNINGS AND PRECAUTIONS (cont’d) SWITCHING TO XARELTO® From warfarin Stop warfarin and start XARELTO® when INR is <3.0 From unfractionated heparin Stop the infusion and start XARELTO at the same time From other anticoagulants Start XARELTO® 0 to 2 hours prior to the next scheduled evening administration of the other anticoagulant ® SWITCHING FROM XARELTO® To warfarin* One approach is to stop XARELTO® and start parenteral anticoagulant and warfarin at time of next scheduled XARELTO® dose To other anticoagulants† Stop XARELTO® and start other anticoagulant when the next dose of XARELTO® would have been given * No clinical trial data are available to guide converting patients from XARELTO® to warfarin. XARELTO® affects INR, so INR measurements made during coadministration with warfarin may not be useful for determining the appropriate dose of warfarin. † Oral or parenteral rapid-onset anticoagulants. ARELTO® eliminates the need for bridging with a parenteral heparin or X low-molecular-weight heparin to oral therapy Important Food Effect Information XARELTO® 15-mg and 20-mg tablets should be taken with food, while the 10-mg tablet can be taken with or without food. It is recommended that patients with nonvalvular AF take XARELTO® with the evening meal, as specified in the ROCKET AF trial protocol. The absolute bioavailability of XARELTO® is dose dependent. For the 10-mg dose, it is estimated to be 80% to 100% and is not affected by food. The absolute bioavailability of XARELTO® at a dose of 20 mg in the fasted state is approximately 66%. Coadministration of XARELTO® with food increases the bioavailability of the 20-mg dose (mean area under the curve [AUC] and maximum concentration [Cmax] increasing by 39% and 76%, respectively, with food). Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. u Patients With Prosthetic Heart Valves: The safety and efficacy of XARELTO® have not been studied in patients with prosthetic heart valves. Therefore, use of XARELTO® is not recommended in these patients. u Acute PE in Hemodynamically Unstable Patients/Patients Who Require Thrombolysis or Pulmonary Embolectomy: Initiation of XARELTO® is not recommended acutely as an alternative to unfractionated heparin in patients with pulmonary embolism who present with hemodynamic instability or who may receive thrombolysis or pulmonary embolectomy. DRUG INTERACTIONS u Avoid concomitant use of XARELTO® with other anticoagulants due to increased bleeding risk, unless benefit outweighs risk. Promptly evaluate any signs or symptoms of blood loss if patients are treated concomitantly with aspirin, other platelet aggregation inhibitors, or NSAIDs. u X ARELTO® should not be used in patients with CrCl 15 to 80 mL/min who are receiving concomitant combined P-gp and moderate CYP3A4 inhibitors unless the potential benefit justifies the potential risk. (continued on page 10) 9 Dose Adjustments IMPORTANT SAFETY INFORMATION (cont’d) No dose adjustments based on age, weight, or gender are required for XARELTO . ® In clinical studies, elderly subjects exhibited higher rivaroxaban plasma concentrations than younger subjects with mean AUC values being approximately 50% higher, mainly due to reduced (apparent) total body and renal clearance. Age-related changes in renal function may play a role in this age effect Extremes in body weight (<50 kg or >120 kg) did not influence (less than 25%) rivaroxaban exposure Gender did not influence the pharmacokinetics or pharmacodynamics of XARELTO® Missed Dose If a dose of XARELTO® is not taken at the scheduled time, the dose should be administered as soon as possible on the same day as follows: atients receiving 20, 15, or 10 mg once daily should take the missed dose of P XARELTO® immediately atients receiving 15 mg twice daily should take XARELTO® immediately to ensure intake P of 30 mg of XARELTO® per day. In this particular instance, two 15-mg tablets may be taken at once. Patients should continue with the regular 15-mg twice-daily intake as recommended on the following day USE IN SPECIFIC POPULATIONS u P regnancy Category C: XARELTO® should be used during pregnancy only if the potential benefit justifies the potential risk to mother and fetus. There are no adequate or wellcontrolled studies of XARELTO® in pregnant women, and dosing for pregnant women has not been established. Use XARELTO® with caution in pregnant patients because of the potential for pregnancy-related hemorrhage and/or emergent delivery with an anticoagulant that is not readily reversible. The anticoagulant effect of XARELTO® cannot be reliably monitored with standard laboratory testing. u L abor and Delivery: Safety and effectiveness of XARELTO® during labor and delivery have not been studied in clinical trials. (continued on page 11) Administration Options F or patients who are unable to swallow whole tablets, XARELTO® 10-mg, 15-mg, or 20-mg tablets may be crushed and mixed with applesauce prior to use and administered orally fter the administration of a crushed XARELTO® 15-mg or 20-mg tablet, the dose should be A immediately followed by food Administration via nasogastric (NG) tube or gastric feeding tube: After confirming gastric placement of the tube, XARELTO® 10-mg, 15-mg, or 20-mg tablets may be crushed and suspended in 50 mL of water, and administered via an NG tube or gastric feeding tube. Since rivaroxaban absorption is dependent on the site of drug release, avoid administration of XARELTO® distal to the stomach, which can result in reduced absorption and, thereby, reduced drug exposure. After the administration of a crushed XARELTO® 15-mg or 20-mg tablet, the dose should then be immediately followed by enteral feeding rushed XARELTO® 10-mg, 15-mg, or 20-mg tablets are stable in water and in applesauce for up C to 4 hours. An in vitro compatibility study indicated that there is no adsorption of rivaroxaban from a water suspension of a crushed XARELTO® tablet to PVC or silicone NG tubing Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 10 Surgery and Other Medical Interventions If anticoagulation must be discontinued to reduce the risk of bleeding with surgical or other procedures, XARELTO® should be stopped at least 24 hours before the procedure In deciding whether a procedure should be delayed until 24 hours after the last dose of XARELTO®, the increased risk of bleeding should be weighed against the urgency of intervention XARELTO® should be restarted after the surgical or other procedures as soon as adequate hemostasis has been established. If oral medication cannot be taken after surgical intervention, consider administering a parenteral anticoagulant Assessment of Coagulation Status No routine monitoring of INR or other coagulation parameters is required for patients taking XARELTO®3-5 •INR should not be used to monitor XARELTO®, as INR is only calibrated and validated for vitamin K antagonists (eg, warfarin) •Dose-dependent inhibition of Factor Xa activity was observed in humans and the Neoplastin® prothrombin time (PT), activated partial thromboplastin time (aPTT), and HepTest® are prolonged dose-dependently •Anti-Factor Xa activity is also influenced by rivaroxaban IMPORTANT SAFETY INFORMATION (cont’d) USE IN SPECIFIC POPULATIONS (con’t) u N ursing Mothers: It is not known if rivaroxaban is excreted in human milk. u Pediatric Use: Safety and effectiveness in pediatric patients have not been established. u Females of Reproductive Potential: Females of reproductive potential requiring anticoagulation should discuss pregnancy planning with their physician. OVERDOSAGE u D iscontinue XARELTO® and initiate appropriate therapy if bleeding complications associated with overdosage occur. A specific antidote for rivaroxaban is not available. The use of activated charcoal to reduce absorption in case of XARELTO® overdose may be considered. Due to the high plasma protein binding, rivaroxaban is not expected to be dialyzable. (continued on page 12) Neoplastin® and HepTest® are trademarks of their respective owners. Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 11 IMPORTANT SAFETY INFORMATION (cont’d) ADVERSE REACTIONS IN CLINICAL STUDIES Visit www.XareltoHCP.com for detailed information about XARELTO and resources to address access and affordability questions. u The most common adverse reactions with XARELTO® were bleeding complications. If you have questions about XARELTO®, call Janssen Medical Information at 1-800-JANSSEN (1-800-526-7736), Monday - Friday, 9:00 am - 8:00 pm ET, Saturday - Sunday, 9:00 am - 5:00 pm ET. Please see accompanying full Prescribing Information, including Boxed WARNINGS. ® 011416-140307 Additional Resources Direct patients and their caregivers to www.XareltoCarePath.com for additional information, including educational videos, tools, and access to the XARELTO® CarePath™ Support Program. A comprehensive support program to help patients start and stay on therapy Help patients start on therapy by making treatment accessible and affordable to eligible patients Help keep patients informed about their disease and the importance of staying on treatment to help support their success Help patients stay on therapy through useful tools and reminders Please see Important Safety Information throughout and accompanying full Prescribing Information, including Boxed WARNINGS. 12 References 1. P atel MR, Mahaffey KW, Garg J, et al; and the ROCKET AF Steering Committee, for the ROCKET AF Investigators. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365(10):883-891. 2. Data on file. Janssen Pharmaceuticals, Inc. 3. The EINSTEIN Investigators. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363(26):2499-2510. 4. EINSTEIN–PE Investigators. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med. 2012;366(14):1287-1297. 5. Mueck W, Eriksson BI, Bauer KA, et al. Population pharmacokinetics and pharmacodynamics of rivaroxaban—an oral, direct Factor Xa inhibitor—in patients undergoing major orthopaedic surgery. Clin Pharmacokinet. 2008;47(3):203-216. XARELTO® is licensed from Bayer HealthCare AG, 51368 Leverkusen, Germany. © Janssen Pharmaceuticals, Inc. 2015 March 2015 015447-150128 Janssen Pharmaceuticals, Inc.