Ch. 2 (Chemistry of life) review questions; Name: _________________________; Score:... 1. Assuming atomic number is 6 and atomic mass... numbers of proton, neutrons, and electrons, respectively.
advertisement

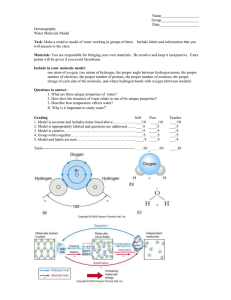
Ch. 2 (Chemistry of life) review questions; Name: _________________________; Score: ______ 1. Assuming atomic number is 6 and atomic mass is 12 for Carbon. Calculate carbon atom’s numbers of proton, neutrons, and electrons, respectively. Answer: Questions 2-4 (Matching)-- Match the following terms to their respective statement below. A. Ionic bond B. Covalent bond C. Hydrogen bond 2. _______ Two hydrogen atoms share valence electrons to form a hydrogen molecule 3. _______ A weak attraction between a slightly positive hydrogen atom in one molecule and a slightly negative oxygen or nitrogen atom in another 4. _______ The attraction of a cation to an anion Questions 5-7 (Matching)-- Match the following terms to their respective statement below. A. Decomposition reaction B. Synthesis reaction C. Exchange reaction 5. _______ Two or more small molecules combine to form a larger one 6. _______ Two molecules exchange atoms or groups of atoms 7. _______ A large molecule breaks down into two or more smaller ones 8. The arrangement of a polypeptide into a fibrous or globular shape is called its _____ structure. (A) primary; (B) secondary; (C) tertiary; (D) quaternary; (E) none of the above 9. When an atom gives up an electron and acquires a positive charge, it is called a/an _____. Answer: 10. Name this functional group (--OH). Answer: l 11. Name this functional group (--NH2). Answer: