Biology Slide 1 of 40 End Show
advertisement

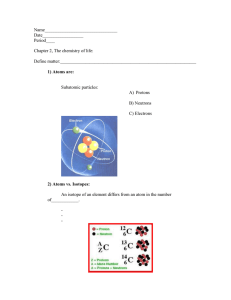
Biology Slide 1 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Slide 2 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Atoms Atoms The study of chemistry begins with the basic unit of matter, the atom. Slide 3 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Atoms Placed side by side, 100 million atoms would make a row only about 1 centimeter long. Atoms contain subatomic particles that are even smaller. Slide 4 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Atoms The subatomic particles that make up atoms are • protons • neutrons • electrons Slide 5 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Atoms The subatomic particles in a helium atom. Slide 6 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Elements and Isotopes Elements and Isotopes A chemical element is a pure substance that consists entirely of one type of atom. • C stands for carbon. • Na stands for sodium. Slide 7 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Elements and Isotopes The number of protons in an atom of an element is the element's atomic number. Commonly found in living organisms: Slide 8 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Elements and Isotopes Isotopes Atoms of the same element that differ in the number of neutrons they contain are known as isotopes. Slide 9 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Elements and Isotopes Because they have the same number of electrons, all isotopes of an element have the same chemical properties. Slide 10 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Elements and Isotopes Isotopes of Carbon 6 electrons 6 protons 8 neutrons Slide 11 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Elements and Isotopes Radioactive Isotopes Some isotopes are radioactive, meaning that their nuclei are unstable and break down at a constant rate over time Slide 12 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Elements and Isotopes Radioactive isotopes can be used: • to determine the ages of rocks and fossils. • to treat cancer. • to kill bacteria that cause food to spoil. • as labels or “tracers” to follow the movement of substances within an organism. Slide 13 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Compounds Chemical Compounds A chemical compound is a substance formed by the chemical combination of two or more elements in definite proportions. Slide 14 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Compounds Chemical Formula Water Table Salt Hydrochloric Acid Glucose Slide 15 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Bonds Chemical Bonds The atoms in compounds are held together by chemical bonds. The electrons that are available to form bonds are called valence electrons. Slide 16 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Bonds The main types of chemical bonds are: • ionic bonds • covalent bonds Slide 17 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Bonds Ionic Bonds An ionic bond is formed when one or more electrons are transferred from one atom to another. These positively and negatively charged atoms are Slide known as ions. 18 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Bonds Sodium ion (Cl-) Sodium atom (Na) Sodium ion (Na+) Sodium atom (Cl) Protons Electrons +11 - 11 10 Protons Electrons +17 18 - 17 Charge +10 Charge -10 Slide 19 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Bonds Covalent Bonds Sometimes electrons are shared by atoms instead of being transferred. Slide 20 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Bonds A covalent bond forms when electrons are shared between atoms. • single covalent bond • double bond • triple bond Slide 21 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Bonds The structure that results when atoms are joined together by covalent bonds is called a molecule. Slide 22 of 40 Copyright Pearson Prentice Hall End Show 2-1 The Nature of Matter Chemical Bonds Van der Waals Forces When molecules are close together, a slight attraction can develop between the oppositely charged regions of nearby molecules. van der Waals forces Slide 23 of 40 Copyright Pearson Prentice Hall End Show 2-1 Click to Launch: Continue to: - or - Slide 24 of 40 End Show Copyright Pearson Prentice Hall 2-1 The particles that move around the nucleus of an atom are called a. neutrons. b. protons. c. electrons. d. isotopes. Slide 25 of 40 End Show Copyright Pearson Prentice Hall 2-1 The atomic number of a carbon atom is 6. How many neutrons does the isotope carbon-14 have? a. 6 b. 8 c. 12 d. 14 Slide 26 of 40 End Show Copyright Pearson Prentice Hall 2-1 Which of the following statements about the three isotopes of carbon is true? a. They are all radioactive. b. They have different numbers of electrons. c. They have the same chemical properties but differ in atomic mass. d. They have the same number of protons and neutrons. Slide 27 of 40 End Show Copyright Pearson Prentice Hall 2-1 A chemical compound consists of a. Electrons mixed with neutrons. b. two or more elements combined in a definite proportion. c. two or more protons combined in any proportion. d. at least three elements combined by ionic or covalent bonds. Slide 28 of 40 End Show Copyright Pearson Prentice Hall 2-1 Van der Waals forces are the result of a. unequal sharing of electrons. b. ionic bonds. c. the bonding of different isotopes. d. the chemical combination of sodium and chlorine. Slide 29 of 40 End Show Copyright Pearson Prentice Hall END OF SECTION