Cu A chart that organizes the elements by the number... Atomic #
advertisement
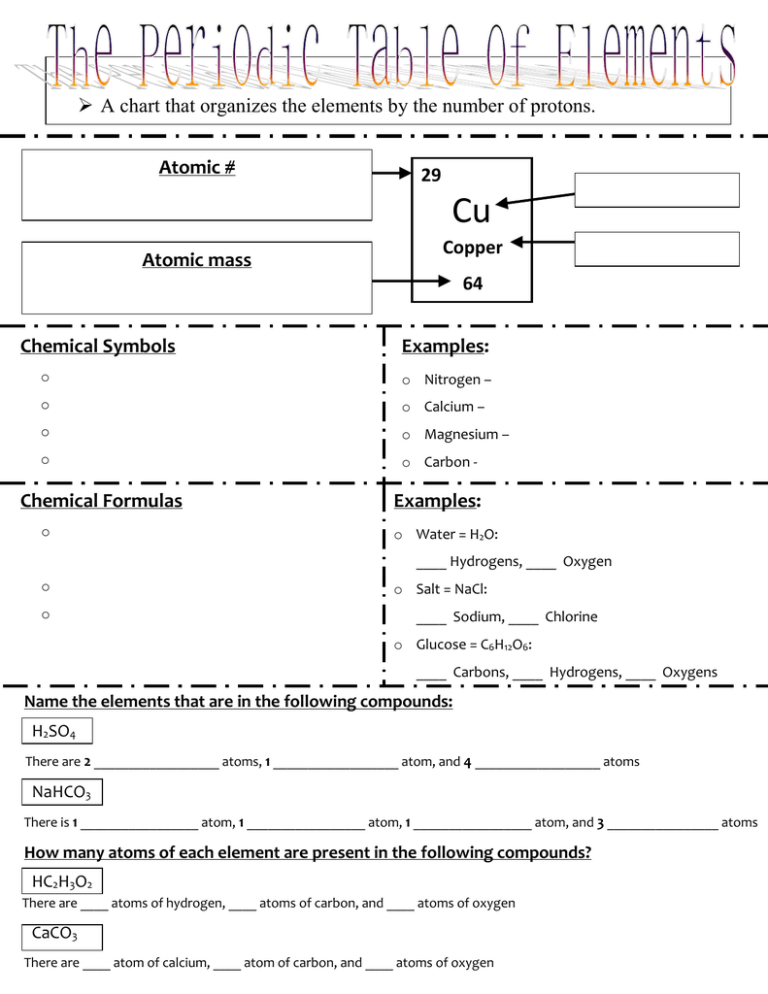
A chart that organizes the elements by the number of protons. Atomic # = number of protons = number of electrons Atomic mass 29 Cu Copper 64 = number of protons = number of electrons Chemical Symbols Examples: o Symbols used to represent the elements. o Nitrogen – o The symbols have 1, 2 or 3 letters o Calcium – o The first letter is ALWAYS CAPATALIZED o Magnesium – o Any other letters are ALWAYS LOWER CASE o Carbon - Chemical Formulas o Chemical symbols put together to represent a compound. o The chemical formula for water is H2O. o The 2 is a subscript. The subscript tells us how many atoms there are of the element IN FRONT OF IT. Examples: o Water = H2O: ____ Hydrogens, ____ Oxygen o Salt = NaCl: ____ Sodium, ____ Chlorine o Glucose = C6H12O6: ____ Carbons, ____ Hydrogens, ____ Oxygens Name the elements that are in the following compounds: H2SO4 There are 2 __________________ atoms, 1 __________________ atom, and 4 __________________ atoms NaHCO3 There is 1 _________________ atom, 1 _________________ atom, 1 _________________ atom, and 3 ________________ atoms How many atoms of each element are present in the following compounds? HC2H3O2 There are ____ atoms of hydrogen, ____ atoms of carbon, and ____ atoms of oxygen CaCO3 There are ____ atom of calcium, ____ atom of carbon, and ____ atoms of oxygen