Name: _______________________________________ Block: _________ Chapter 5 activity: Periodic Table
advertisement

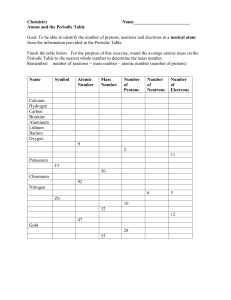
Name: _______________________________________ Block: _________ Chapter 5 activity: Periodic Table Objective: To identify information obtained from the periodic table. Materials: Periodic table, writing instrument and calculator. PART 1 Procedure: Answer the following questions by referring to the periodic table. 1. Name the five (5) family members included with beryllium. What do they have in common? 2. Are potassium, iron, and zinc in the same family or the same period? What do they have in common? 3. Are lithium, magnesium, scandium, zirconium, and tantalum in the same family or period? What do they have in common? 4. Which element of the following is not a member of this family? Oxygen, polonium, sulfur, antimony, tellurium, selenium. PART 2 Procedure: It is possible by using information about each element supplied by a periodic table to determine the number of electrons, neutrons, and protons of a given atom. To review, here are a few simple rules: 1. Number of protons is the same as the atomic number. 2. Number of electrons equals the number of protons. 3. Atomic mass is the number of protons plus the number of neutrons. Given the information above, fill in the blanks below. Atomic Atomic Element Number Protons Neutrons Electrons Mass _______ _______ _______ _______ 8 16 Iron _______ 26 _______ _______ 56 Uranium _______ _______ 146 92 _______ Silicon _______ _______ 14 _______ 28 _______ 20 _______ _______ _______ 40 _______ _______ _______ _______ _______ 4 _______ 11 _______ _______ _______ _______ Hydrogen _______ _______ _______ _______ 1 _______ _______ 7 _______ _______ _______ Potassium _______ _______ _______ _______ _______ Copper _______ _______ _______ _______ 64 _______ 82 _______ _______ _______ _______ _______ _______ _______ _______ 30 _______