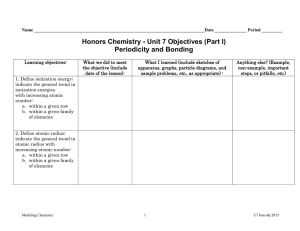
Graph of atomic radius vs. atomic number
advertisement

Graph of atomic radius vs. atomic number 1. Connect the circles from atomic number 1 (hydrogen) to atomic number 54 (xenon) using a pencil 2. Using a different color for each group, fill in the circles that represent the members of the following families: 3. Group 1 (alkali metals) Group 2 (alkaline earth metals) Group 13 (boron group) ▪ Group 14 (carbon group) ▪ Group 17 (halogens) ▪ Group 18 (noble gases) Draw lines between circles of the same color (elements in the same family or group) 1. Does atomic radius appear to be a periodic property? Explain. 2. Which family has the largest atomic radii within any given period? Which family has the smallest radius? 3. In general, what happens to atomic radius as you proceed through a period (row) from the alkali metals to the noble gases? Explain why this might be so in terms of the attractive force between the nucleus and electrons.