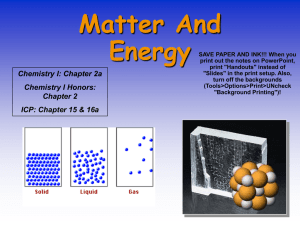
ANYTHING WITH MASS AND VOLUME
advertisement

ANYTHING WITH MASS AND VOLUME Elements Draw this chart!, leave space to define the terms Pure Substances Compounds Matter Homogeneous Mixtures Heterogeneous Pure Substances vs. Mixtures • A pure substance is made of only one kind of material and has definite properties. • Matter that consists of two or more substances mixed together but not chemically combined is called a mixture. Pure Substances: • Elements are the simplest pure substance. – Examples: hydrogen, carbon, and oxygen. • The smallest particle of an element that has the properties of that element is called an atom. Pure substances: Compounds are pure substances that are made of more than one element bound together. – Examples: water (H2O), and carbon dioxide. A molecule is formed when two or more atoms chemically combine. Example: water (H2O), O2 MIXTURES Heterogeneous vs. Homogeneous Homogeneous matter (solutions): matter that has identical properties throughout. Examples: salt water, gravy, whipped cream Heterogeneous matter: matter that has parts with different properties. Examples: granite, soil, potpourri, cookies LIQUID MIXTURES Miscible vs. Immiscible • Miscible Mixture: liquids that mix evenly, forming a homogeneous solution. Example: Soda mixed with Koolaid • Immiscible Mixture: liquids that DO NOT mix evenly, forming a heterogeneous mixture. Example: Oil mixed with Water EXAMPLES • What type of matter are each of the following… ? SAND SAND Heterogeneous mixture Salt (NaCl) Salt (NaCl) Pure Substance: COMPOUND Air Air • Homogeneous mixture of: Many gases make up mixture, but it looks like it is all one gas. Nitrogen, N2 78.08% Oxygen, O2 20.95% Argon, Ar 0.93% Carbon dioxide, CO2 0.033% Neon, Ne 0.0018% Helium, He 0.00052% Methane, CH4 0.0002% Krypton, Kr 0.00011% Nitrogen(I) oxide, N2O 0.00005% Hydrogen, H2 0.00005% Xenon, Xe 0.0000087% Ozone, O3 0.000001% Gold Gold Pure Substance: ELEMENT: Au Bronze Bronze Homogeneous mixture of copper and tin (alloy: mixture of metals) Salad Dressing: Salad Dressing: Heterogeneous Mixture Conservation • Law of Conservation of Mass: Mass cannot be created or destroyed. • Law of Conservation of Energy: Energy cannot be created or destroyed; it may only change from one form to another. Matter and Energy MUST be conserved is the LAW! Virtually everything is made up of atoms. From the very large... To the very small... We are all made of atoms…and only atoms. This includes you and me! Giggitty, Giggitty, Goo! Currently we have about 117 kinds of atoms. In the natural world there exists 92 different kinds of atoms. The others have been artificially produced in laboratories. The Elements Song We call each kind of atom an element, and give it a specific name and symbol. Copper Gold Cu Au Periodic Table Abundance of the elements, by weight The Earth’s interior is rich in iron Sand is made of Silicon & Oxygen The ocean waters are made of oxygen & hydrogen Atoms are made up of protons, neutrons, and Of electrons. course real atoms don’t look anything like this you imbecile! Protons and neutrons are found in the nucleus of atoms -- roughly at the center Electrons travel around the nucleus. Different kinds of atoms, or elements, are different because they have different numbers of protons. It would be sweet if atoms really were this huge! We list the elements by their atomic numbers the number of protons they have. Hydrogen, number 1 Helium, number 2 In several cases the atomic weights are in parentheses. This indicates that these elements have no stable isotopes; that is, they are radioactive. The value enclosed in parentheses and used for the atomic weight is the atomic mass number of the most stable known isotope, as indicated by the longest half-life. Physical Properties • Physical properties: characteristics that can be observed without changing the identity of the substance. • Examples: – – – – – – mass volume color shape texture density Physical Changes • Physical change: a change in the physical form or properties of a substance that occurs without a change in composition. • Examples: – – – – melting freezing grinding dissolving Chemical Properties • Chemical property: describes a substance’s ability to change into a different substance. • Examples: – flammability – reactivity Chemical Changes • Chemical change: occurs when a substance changes composition by forming one or more new substances. (bonds are broken and bonds are formed) • Example: – HCl + NaOH NaCl + H2O Indications of a Chemical Change… Heat in Endothermic (feels cold) Heat out Exothermic (feels hot) Gas is given off (fizzing or bubbles) Color Change New Substance is Formed Evaporation is a physical change Breaking is a physical change. Boiling is a change of state, and therefore a physical change! Rusting is a Chemical Change Burning is a Chemical Change Kinetic Theory • All matter is made of atoms and molecules that act like tiny particles. • These tiny particles are always in motion. The higher the temp., the faster the particles move. • At the same temp., more massive (heavier) particles move slower than less massive (lighter) particles. (inertia) SOLIDS • Definite shape? • YES • Definite volume? • YES • Molecules in a solid are tightly packed and constantly vibrating. Eureka: Molecules in Solids LIQUIDS • Definite shape? • NO • Definite volume? • YES • Some liquids flow more easily than others. The resistance of a liquid to flow is called viscosity. – Honey has a high viscosity compared to water. Eureka: Molecules in Liquids GASES • Definite shape? • NO • Definite volume? • NO • The particles in a gas are spread very far apart, but can be compressed by pumping them into a restricted volume. Eureka: Molecules in Gases Phase Changes (Changes of State) • Changes in phase are examples of physical changes. • • • • • • Melting: solid liquid Freezing: liquid solid Vaporization: liquid gas Condensation: gas liquid Sublimation: solid gas Deposition: gas solid Changes of State GAS Vaporization Sublimation Condensation Deposition Melting LIQUID Melting Freezing SOLID Energy Transfers • ENERGY is the ability to change or move matter. • Energy is ABSORBED when substances melt or evaporate. – NOTE: our bodies cool down when our sweat evaporates. • Energy is RELEASED when substances freeze or condense. Melting • The change of state from solid to liquid. • Energy (heat) is absorbed by the substance that is melting. Freezing • The change of state from liquid to solid. Opposite of melting. • Energy (heat) is released by the substance undergoing freezing. Evaporation • The change of state at the surface of a liquid as it passes to a vapor. This results from the random motion of molecules that occasionally escape from the liquid surface. – Energy (heat) is absorbed by the liquid – Can happen at any temperature Condensation • The change of state from gas to liquid. The opposite of evaporation. – Energy (heat) is released by the gas to become a liquid. Boiling • Change from state from a liquid to a gas. • Occurs throughout the liquid. – boiling point/temperature is determined by pressure – Energy (heat) is absorbed by the liquid for it to boil and produce gas. *Boiling & freezing points depend on the pressure. Water at normal pressure (1 atm): • For water at normal (every day) pressures: • Melting/freezing point: 0 oC (32oF) • Condensing/boiling point: 100 oC (212oF) 100°C 0°C *Boiling & freezing points depend on the pressure. Change the pressure Change the Boiling Point