Chapter 9 Lecture Outline Prepared by
advertisement

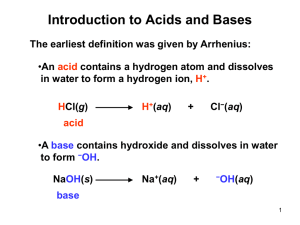
Chapter 9 Lecture Outline Prepared by Andrea D. Leonard University of Louisiana at Lafayette 1 Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 9.1 Introduction to Acids and Bases The Brønsted–Lowry definition: •A Brønsted–Lowry acid is a proton (H+) donor. •A Brønsted–Lowry base is a proton (H+) acceptor. This proton is donated. HCl(g) + H2O(l) H3O+(aq) + Cl−(aq) •HCl is a Brønsted–Lowry acid because it donates a proton to the solvent water. •H2O is a Brønsted–Lowry base because it accepts a proton from HCl. 4 9.1 Introduction to Acids and Bases A. Brønsted–Lowry Acids •A Brønsted–Lowry acid must contain a hydrogen atom. •Common Brønsted–Lowry acids (HA): HCl hydrochloric acid HBr hydrobromic acid HNO3 nitric acid H2SO4 sulfuric acid H H C O C acidic H atom O H H acetic acid 5 9.1 Introduction to Acids and Bases A. Brønsted–Lowry Acids •A monoprotic acid contains one acidic proton. HCl •A diprotic acid contains two acidic protons. H2SO4 •A triprotic acid contains three acidic protons. H3PO4 •A Brønsted–Lowry acid may be neutral or it may carry a net positive or negative charge. HCl, H3O+, HSO4− 6 9.1 Introduction to Acids and Bases B. Brønsted–Lowry Bases •A Brønsted–Lowry base is a proton acceptor, so it must be able to form a bond to a proton. •A base must contain a lone pair of electrons that can be used to form a new bond to the proton. This e− pair forms a new bond to a H from H2O. H N H + H Brønsted–Lowry base H2O(l) + H H N H + OH− (aq) H 7 9.1 Introduction to Acids and Bases B. Brønsted–Lowry Bases •Common Brønsted–Lowry Bases (B ): Lone pairs make these neutral compounds bases. NH3 ammonia H2O water The OH− is the base in each metal salt. NaOH sodium hydroxide KOH potassium hydroxide Mg(OH)2 magnesium hydroxide Ca(OH)2 calcium hydroxide 8 9.2 The Reaction of a Brønsted–Lowry Acid with a Brønsted–Lowry Base This e− pair stays on A. H A acid This e− pair forms a new bond to H+. gain of H+ + B base A − + H B+ loss of H+ 9 9.2 The Reaction of a Brønsted–Lowry Acid with a Brønsted–Lowry Base gain of H+ H A acid + B base A − + H B+ conjugate conjugate acid base loss of H+ •The product formed by loss of a proton from an acid is called its conjugate base. •The product formed by gain of a proton by a base is called its conjugate acid. 10 9.2 The Reaction of a Brønsted–Lowry Acid with a Brønsted–Lowry Base gain of H+ H Br acid + H2O base Br− + H3O+ conjugate conjugate acid base loss of H+ •HBr and Br− are a conjugate acid–base pair. •H2O and H3O+ are a conjugate acid–base pair. •The net charge must be the same on both sides of the equation. 11 9.2 The Reaction of a Brønsted–Lowry Acid with a Brønsted–Lowry Base Amphoteric compound: A compound that contains both a hydrogen atom and a lone pair of e−; it can be either an acid or a base. + H H O H add H+ H2O as a base H O H H2O as an acid H O H conjugate acid remove H+ − H O conjugate base 13 9.3 Acid and Base Strength A. Relating Acid and Base Strength •When a covalent acid dissolves in water, the proton transfer that forms H3O+ is called dissociation. •When a strong acid dissolves in water, 100% of the acid dissociates into ions. HCl(g) + H2O(l) H3O+(aq) + Cl−(aq) •A single reaction arrow can be used, because the product is greatly favored at equilibrium. •Common strong acids are HI, HBr, HCl, H2SO4, and HNO3. 14 9.3 Acid and Base Strength A. Relating Acid and Base Strength •When a weak acid dissolves in water, only a small fraction of the acid dissociates into ions. •Unequal reaction arrows may be used, because the reactants are usually favored at equilibrium. CH3COOH(l) + H2O(l) H3O+(aq) + CH3COO−(aq) •Common weak acids are H3PO4, HF, H2CO3, and HCN. 15 9.3 Acid and Base Strength A. Relating Acid and Base Strength A strong acid, HCl, is completely dissociated into H3O+(aq) and Cl−(aq). A weak acid, CH3COOH, contains mostly undissociated acid. 16 9.3 Acid and Base Strength A. Relating Acid and Base Strength •A strong acid readily donates a proton, forming a weak conjugate base. HCl strong acid Cl− weak conjugate base •A strong base readily accepts a proton, forming a weak conjugate acid. OH− strong base H2O weak conjugate acid 19 9.3 Acid and Base Strength A. Relating Acid and Base Strength 9.2 ----- 20 9.3 Acid and Base Strength B. Using Acid Strength to Predict the Direction of Equilibrium •A Brønsted–Lowry acid–base reaction represents an equilibrium. H A acid + B base A − + H B+ conjugate conjugate acid base •The position of the equilibrium depends upon the strengths of the acids and bases. •The stronger acid reacts with the stronger base to form the weaker acid and the weaker base. 21 9.3 Acid and Base Strength B. Using Acid Strength to Predict the Direction of Equilibrium •When the stronger acid and base are the reactants on the left side, the reaction readily occurs and the reaction proceeds to the right. H A + B stronger stronger acid base A − + weaker base H B+ weaker acid •A larger forward arrow means that products are favored. 22 9.3 Acid and Base Strength B. Using Acid Strength to Predict the Direction of Equilibrium •If an acid–base reaction would form the stronger acid and base, equilibrium favors the reactants and little product forms. H A weaker acid + B weaker base A − + stronger base H B+ stronger acid •A larger reverse arrow means that reactants are favored. 23 9.3 Acid and Base Strength HOW TO Predict the Direction of Equilibrium in an Acid–Base Reaction Are the reactants or products favored in Example the following acid–base reaction? gain of H+ HCN(g) acid + −OH(aq) base −CN(aq) + H2O(l) conjugate conjugate acid base loss of H+ Step [1] Identify the acid in the reactants and the conjugate acid in the products. 24 9.3 Acid and Base Strength HOW TO Predict the Direction of Equilibrium in an Acid–Base Reaction Step [2] Determine the relative strength of the acid and the conjugate acid. •From Table 9.2, HCN is a stronger acid than H2O. Step [3] HCN(g) stronger acid Equilibrium favors the formation of the weaker acid. + −OH(aq) −CN(aq) + H2O(l) weaker acid Products are favored. 25 9.4 Equilibrium and Acid Dissociation Constants For the reaction where an acid (HA) dissolves in water, HA(g) + H2O(l) H3O+(aq) + A − (aq) the following equilibrium constant can be written: K = [H3O+][ A −] [HA][H2O] 26 9.4 Equilibrium and Acid Dissociation Constants •Multiplying both sides by [H2O] forms a new (more useful) constant, called the acid dissociation constant, Ka. Ka = K[H2O] = [H3O+][ A −] [HA] acid dissociation constant •The stronger the acid, the larger the Ka value. •Equilibrium favors formation of the weaker acid, the acid with the smaller Ka value. 27 9.4 Equilibrium and Acid Dissociation Constants 28 9.5 Dissociation of Water Water can behave as both a Brønsted–Lowry acid and a Brønsted–Lowry base. Thus, two water molecules can react together in an acid–base reaction: loss of H+ H O acid H + H O base H + H − O H conjugate base + H O H conjugate acid gain of H+ 29 9.5 Dissociation of Water •From the reaction of two water molecules, the following equilibrium constant expression can be written: K = [H3O+][OH−] [H2O]2 •Multiplying both sides by [H2O]2 yields Kw, the ion-product constant for water. Kw = ion-product constant [H3O+][OH−] 30 9.5 Dissociation of Water •Experimentally it can be shown that: [H3O+] = [OH−] = 1.0 x 10−7 M at 25 oC Kw = [H3O+] [OH−] Kw = (1.0 x 10−7) x (1.0 x 10−7) Kw = 1.0 x 10−14 •Kw is a constant, 1.0 x 10−14, for all aqueous solutions at 25 oC. 31 9.5 Dissociation of Water If the [H3O+] in a cup of coffee is 1.0 x 10−5 M, then the [OH−] can be calculated as follows: [OH−] = Kw [H3O+] 1.0 x 10−14 = = −5 1.0 x 10 1.0 x 10−9 M In this cup of coffee, therefore, [H3O+] > [OH–], and the solution is acidic overall. 33 9.5 Dissociation of Water 34 9.6 The pH Scale A. Calculating pH The lower the pH, the higher the concentration of H3O+: •Acidic solution: [H3O+] > 1 x 10−7 •Neutral solution: [H3O+] = 1 x 10−7 pH = 7 •Basic solution: [H3O+] < 1 x 10−7 pH < 7 pH > 7 35 9.6 The pH Scale A. Calculating pH 36 9.6 The pH Scale B. Calculating pH Using a Calculator •A logarithm has the same number of places after the decimal as there are digits in the original number. •Example: If [H3O+] = 1.2 x 10–5 M for a solution, what is its pH? pH = –log [H3O+] pH = –log (1.2 x 10–5) 2 digits pH = –(–4.92) 2 decimal places pH = 4.92 The solution is acidic because the pH < 7. 37 9.6 The pH Scale B. Calculating pH Using a Calculator If the pH of a solution is 8.50, what is the [H3O+]? pH = −log [H3O+] 8.50 = −log [H3O+] −8.50 = log [H3O+] antilog (−8.50 ) = [H3O+] 2 decimal places [H3O+] = 3.2 x 10−9 M 2 digits The solution is basic because [H3O+] > 1 x 10–7 M. 38 9.6 The pH Scale 39 9.7 Common Acid–Base Reactions A. Reaction of Acids with Hydroxide Bases Neutralization reaction: An acid-base reaction that produces a salt and water as products. HA(aq) + MOH(aq) base acid H OH(l) + MA(aq) water salt •The acid HA donates a proton (H+) to the OH− base to form H2O. •The anion A− from the acid combines with the cation M+ from the base to form the salt MA. 40 9.7 Common Acid–Base Reactions HOW TO Draw a Balanced Equation for a Neutralization Reaction Between HA and MOH Example Write a balanced equation for the reaction of Mg(OH)2 with HCl. Step [1] Identify the acid and base in the reactants and draw H2O as one product. HCl(aq) + Mg(OH)2(aq) base acid H2O(l) + salt water 41 9.7 Common Acid–Base Reactions HOW TO Draw a Balanced Equation for a Neutralization Reaction between HA and MOH Step [2] Determine the structure of the salt. •The salt is formed from the parts of the acid and base that are not used to form H2O. HCl Mg(OH)2 H+ Cl− reacts to used to form H2O form salt Mg2+ 2 OH− used to react to form salt form water Mg2+ and Cl− combine to form MgCl2. 42 9.7 Common Acid–Base Reactions HOW TO Draw a Balanced Equation for a Neutralization Reaction between HA and MOH Step [3] Balance the equation. Place a 2 to balance O and H. 2 HCl(aq) + Mg(OH)2(aq) base acid 2 H2O(l) + MgCl2 water salt Place a 2 to balance Cl. 43 9.7 Common Acid–Base Reactions A. Reaction of Acids with Hydroxide Bases A net ionic equation contains only the species involved in a reaction. HCl(aq) + NaOH(aq) H—OH(l) + NaCl(aq) •Written as individual ions: H+(aq) + Cl−(aq) + Na+(aq) + OH− (aq) H—OH(l) + Na+(aq) + Cl−(aq) •Omit the spectator ions, Na+ and Cl–. •What remains is the net ionic equation: H+(aq) + OH− (aq) H—OH(l) 44 9.7 Common Acid–Base Reactions B. Reaction of Acids with Bicarbonate Bases •A bicarbonate base, HCO3−, reacts with one H+ to form carbonic acid, H2CO3. H+(aq) + HCO3−(aq) H2CO3(aq) H2O(l) + CO2(g) •Carbonic acid then decomposes into H2O and CO2. •For example: HCl(aq) + NaHCO3(aq) NaCl(aq) + H2CO3(aq) H2O(l) + CO2(g) 45 9.7 Common Acid–Base Reactions B. Reaction of Acids with Bicarbonate Bases •A carbonate base, CO32–, reacts with two H+ to form carbonic acid, H2CO3. 2 H+(aq) + CO32–(aq) H2CO3(aq) H2O(l) + CO2(g) •For example: 2 HCl(aq) + Na2CO3(aq) 2 NaCl(aq) + H2CO3(aq) H2O(l) + CO2(g) 46 9.8 The Acidity and Basicity of Salt Solutions A salt can form an acidic, basic, or neutral solution depending on whether its cation and anion are derived from a strong or weak acid and base. For the salt M+A−: •The cation M+ comes from the base. •The anion A− comes from the acid HA. 47 9.8 The Acidity and Basicity of Salt Solutions NaCl Na+ from NaOH strong base Cl− from HCl strong acid A salt derived from a strong acid and strong base forms a neutral solution (pH = 7). 48 9.8 The Acidity and Basicity of Salt Solutions NaHCO3 Na+ from NaOH strong base HCO3− from H2CO3 weak acid A salt derived from a strong base and a weak acid forms a basic solution (pH > 7). 49 9.8 The Acidity and Basicity of Salt Solutions NH4Cl NH4+ from NH3 weak base Cl− from HCl strong acid A salt derived from a weak base and a strong acid forms an acidic solution (pH < 7). 50 9.8 The Acidity and Basicity of Salt Solutions Thus, the ion derived from the stronger acid or base determines whether the solution is acidic or basic. 51 9.9 Titration •To determine the concentration of an acid or base in a solution, we carry out a titration. •If we want to know the concentration of an acid solution, a base of known concentration is added slowly until the acid is neutralized. •When the acid is neutralized: # of moles of acid = # of moles of base •This is called the end point of the titration. 52 9.9 Titration 53 9.9 Titration Determining an unknown molarity from titration data requires three operations: mole–mole conversion factor Moles of base M (mol/L) conversion factor [1] Volume of base [2] Moles of acid [3] M (mol/L) conversion factor Volume of acid 54 9.9 Titration HOW TO Determine the Molarity of an Acid Solution from Titration Example What is the molarity of an HCl solution if 22.5 mL of a 0.100 M NaOH solution are needed to titrate a 25.0 mL sample of the acid? volume of base (NaOH) 22.5 mL conc. of base (NaOH) 0.100 M volume of acid (HCl) 25.0 mL conc. of acid (HCl) ? 55 9.9 Titration HOW TO Determine the Molarity of an Acid Solution from Titration Step [1] Determine the number of moles of base used to neutralize the acid. Volume of base 22.5 mL NaOH x M (mol/L) conversion factor 1L x 0.100 mol NaOH 1000 mL 1L = 0.00225 mol NaOH Moles of base 56 9.9 Titration HOW TO Determine the Molarity of an Acid Solution from Titration Step [2] Determine the number of moles of acid that react from the balanced chemical equation. HCl(aq) + NaOH(aq) H2O(l) + NaCl(aq) mole–mole conversion Moles of Moles of factor base acid 0.00225 mol NaOH x 1 mol HCl = 0.00225 mol HCl 1 mol NaOH 57 9.9 Titration HOW TO Determine the Molarity of an Acid Solution from Titration Step [3] M = = Determine the molarity of the acid from the number of moles and the known volume. mol L = 0.00225 mol HCl x 25.0 mL solution 0.0900 M HCl 1000 mL 1L mL—L conversion factor 3 sig. fig. Answer 58 9.10 Buffers A buffer is a solution whose pH changes very little when acid or base is added. Most buffers are solutions composed of roughly equal amounts of: •A weak acid •The salt of its conjugate base The buffer resists change in pH because •Added base, OH−, reacts with the weak acid •Added acid, H3O+, reacts with the conjugate base 59 9.10 Buffers A. General Characteristics of a Buffer If an acid is added to the following buffer equilibrium, then the excess acid reacts with the conjugate base, so the overall pH does not change much. 60 9.10 Buffers A. General Characteristics of a Buffer If a base is added to the following buffer equilibrium, then the excess base reacts with the conjugate acid, so the overall pH does not change much. 61 9.10 Buffers 62 9.10 Buffers B. Calculating the pH of a Buffer •The effective pH range of a buffer depends on its Ka. H3O+(aq) + A −(aq) HA(aq) + H2O(l) Ka = [H3O+][ A −] [HA] •Rearranging this expression to solve for [H3O+]: [H3 O+] = determines the buffer pH Ka x [HA] [ A −] 63 9.11 Focus on the Human Body Buffers in the Blood •Normal blood pH is between 7.35 and 7.45. •The principal buffer in the blood is carbonic acid/ bicarbonate (H2CO3/HCO3−). CO2(g) + H2O(l) H2CO3(aq) H2O H3O+(aq) + HCO3−(aq) •CO2 is constantly produced by metabolic processes in the body. •The amount of CO2 is related to the pH of the blood. 64 9.11 Focus on the Human Body Buffers in the Blood Respiratory acidosis results when the body fails to eliminate enough CO2, due to lung disease or failure. 65 9.11 Focus on the Human Body Buffers in the Blood Respiratory alkalosis is caused by hyperventilating; very little CO2 is produced by the body. 66